Advanced One-Pot Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
Advanced One-Pot Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharmaceutical Applications
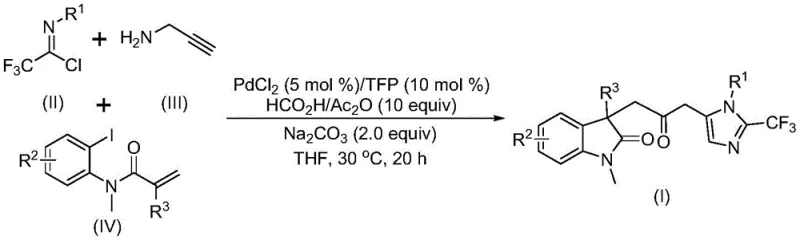
The pharmaceutical and fine chemical industries are constantly seeking more efficient, safer, and cost-effective routes to complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift from traditional step-wise couplings to a streamlined, one-pot palladium-catalyzed cascade reaction. By integrating trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, the process constructs indolinone-imidazole frameworks with high atom economy. For R&D directors and procurement managers, this innovation offers a compelling value proposition: it eliminates the need for hazardous carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility, thereby addressing critical safety and supply chain challenges in modern API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has relied on methodologies that are fraught with operational complexities and safety risks. Traditional approaches often involve the direct coupling of two separate heterocyclic substrates or oxidative cyclization reactions that require harsh conditions and multiple synthetic steps. A particularly significant bottleneck in conventional carbonylation chemistry is the reliance on exogenous carbon monoxide gas. Handling pressurized CO cylinders necessitates specialized infrastructure, rigorous safety protocols, and expensive gas detection systems, all of which inflate the capital expenditure and operational overhead for chemical manufacturers. Furthermore, multi-step syntheses inherently suffer from cumulative yield losses and increased waste generation, as each isolation and purification stage contributes to material loss and solvent consumption. These factors collectively hinder the economic viability of producing complex biheterocycles at a commercial scale, creating a pressing need for more integrated and benign synthetic strategies.
The Novel Approach
The methodology described in patent CN115353511A overcomes these historical barriers through an elegant multi-component design that merges bond formation and ring closure into a single operational sequence. Instead of introducing toxic CO gas, the process utilizes a mixture of formic acid and acetic anhydride to generate carbon monoxide in situ. This clever substitution not only mitigates severe safety hazards but also simplifies the reactor setup, allowing the reaction to proceed under standard atmospheric pressure conditions. The reaction is catalyzed by a palladium system, specifically palladium chloride paired with a trifurylphosphine ligand, which facilitates the intricate cascade of bond formations required to build the dual heterocyclic core. Operating at a remarkably mild temperature of 30°C, this approach preserves the integrity of sensitive functional groups that might degrade under the high thermal stress of older methods. The result is a robust, scalable protocol that delivers diversified substituted double heterocyclic compounds with trifluoromethyl and carbonyl functionalities in high yields.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide precursor. This is followed by an intramolecular Heck reaction, which generates a divalent alkyl-palladium intermediate and establishes the initial cyclic framework. Subsequently, the carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond, forming an acyl-palladium intermediate. This carbonylation step is the linchpin of the process, introducing the essential carbonyl bridge that links the two heterocyclic systems. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine forms a trifluoroacetamidine compound, which undergoes isomerization. The final stage involves the activation of this amidine by the acyl-palladium intermediate, triggering an intramolecular cyclization that locks the structure into the final carbonyl-bridged biheterocyclic product. This complex interplay of catalytic cycles ensures high regioselectivity and minimizes the formation of unwanted byproducts.
From an impurity control perspective, the choice of reagents and conditions plays a pivotal role in ensuring product purity. The use of sodium carbonate as a base helps neutralize acidic byproducts generated during the in situ CO formation, preventing acid-catalyzed degradation of the sensitive imidazole or indolinone rings. Furthermore, the specific ligand environment provided by trifurylphosphine stabilizes the palladium center, reducing the likelihood of palladium black precipitation which can lead to metal contamination in the final API. The mild reaction temperature of 30°C further suppresses thermal decomposition pathways that often plague high-temperature carbonylations. For quality control teams, this means a cleaner crude reaction profile, which simplifies downstream purification. The ability to tolerate diverse substituents on the aromatic rings—such as halogens, alkyl groups, and trifluoromethyl moieties—without compromising yield indicates a broad functional group tolerance, making this method highly versatile for generating libraries of analogs for drug discovery programs.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis in a laboratory or pilot plant setting, precise adherence to the molar ratios and solvent systems defined in the patent is essential for reproducibility. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, and the CO-generating mixture in an aprotic solvent like tetrahydrofuran (THF). The substrates are then introduced, and the mixture is stirred at the controlled temperature. Detailed standardized synthesis steps follow below to ensure consistent high-quality output.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits beyond mere chemical novelty. The elimination of toxic carbon monoxide gas from the supply chain removes a significant logistical and regulatory burden, streamlining the procurement of raw materials and reducing the need for specialized hazard mitigation infrastructure. This simplification directly correlates to lower operational expenditures and reduced insurance premiums associated with handling hazardous gases. Moreover, the one-pot nature of the reaction consolidates what would traditionally be multiple synthetic steps into a single unit operation. This consolidation drastically reduces solvent consumption, labor hours, and equipment occupancy time, leading to substantial cost savings in manufacturing. The high atom economy and excellent yields reported in the patent examples further enhance the overall process efficiency, ensuring that raw material costs are optimized and waste disposal volumes are minimized.
- Cost Reduction in Manufacturing: The replacement of gaseous carbon monoxide with liquid reagents (formic acid and acetic anhydride) fundamentally alters the cost structure of the synthesis. Liquid reagents are easier to store, transport, and meter accurately compared to compressed gases, which require cylinder rentals and specialized delivery logistics. Additionally, the mild reaction conditions (30°C) significantly reduce energy consumption for heating and cooling compared to processes requiring reflux or cryogenic temperatures. The high conversion rates observed mean that less starting material is wasted, directly improving the cost-per-kilogram metric for the final intermediate. By avoiding complex multi-step sequences, the process also reduces the cumulative loss of material, ensuring that the theoretical yield is much closer to the actual isolated yield, which is a critical factor in determining the commercial viability of high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including trifluoroethylimidoyl chloride, propargylamine, and various acrylamides, are commercially available and inexpensive. This availability ensures a stable supply chain that is not dependent on niche or custom-synthesized precursors that might face availability bottlenecks. The robustness of the reaction across a wide range of substrates means that supply chain disruptions for one specific derivative can often be mitigated by switching to alternative analogs without re-engineering the entire process. Furthermore, the scalability demonstrated in the patent, extending from milligram to gram scales with consistent performance, provides confidence for seamless transition to multi-kilogram or ton-scale production. This reliability is paramount for maintaining continuous manufacturing schedules and meeting the strict delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a greener alternative to traditional carbonylations. The in situ generation of CO minimizes the risk of accidental release into the workplace, aligning with increasingly stringent occupational safety regulations. The use of THF as a solvent, while requiring proper recovery systems, is well-established in the industry, and the reduced reaction time (12-20 hours) allows for faster batch turnover, increasing the throughput of existing manufacturing assets. The simplified post-treatment procedure, involving filtration and column chromatography, avoids complex aqueous workups that generate large volumes of wastewater. This reduction in waste generation not only lowers disposal costs but also enhances the sustainability profile of the manufacturing site, a key metric for modern corporate social responsibility goals and for satisfying the green chemistry criteria of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this multi-component synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide accurate guidance for potential adopters.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, significantly reducing workplace hazards and regulatory compliance burdens.
Q: What represents the optimal temperature range for this palladium-catalyzed cascade reaction?
A: The process operates efficiently at a mild temperature of 30°C. This low thermal requirement not only saves energy costs but also ensures compatibility with heat-sensitive functional groups often found in complex pharmaceutical intermediates.
Q: Can this multi-component reaction be scaled for industrial production?
A: Yes, the patent explicitly demonstrates the feasibility of scaling the reaction to the gram level with high efficiency. The use of commercially available starting materials and simple post-treatment procedures supports robust commercial scale-up potential.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to grow in the development of next-generation therapeutics, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced catalytic technologies like the palladium-catalyzed carbonylation described in CN115353511A to deliver superior intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications, minimizing the risk of downstream processing issues.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a reliable, cost-effective, and safe supply of these critical building blocks for your drug development pipeline.
