Innovative Alkyl Sulfonyl Fluoride Synthesis: Scaling Click Chemistry Intermediates from Lab to Commercial Production
Patent CN113698325B introduces a groundbreaking method for synthesizing alkyl sulfonyl fluoride compounds, which are critical building blocks in modern click chemistry applications. This innovative approach addresses longstanding challenges in sulfonyl fluoride production by utilizing reduced active esters derived from readily available alkyl carboxylic acids as starting materials. The process employs sulfur dioxide substitute reagents as SO2 sources and electrophilic fluorinating agents for fluorination under reducing metal conditions. Unlike conventional methods that often require harsh conditions or specialized precursors, this novel technique offers exceptional selectivity for in-situ introduction of sulfonyl fluoride groups with significantly improved yields. The methodology represents a substantial advancement in synthetic chemistry, particularly for pharmaceutical intermediates where purity and structural fidelity are paramount. By leveraging common laboratory reagents and mild reaction conditions, this process enables more efficient production of sulfonyl fluorides that serve as essential components in drug discovery, materials science, and bioconjugation applications. The patent demonstrates how this approach overcomes previous limitations while providing a scalable pathway suitable for industrial implementation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing alkyl sulfonyl fluorides have been severely constrained by multiple technical challenges that limit their practical utility in industrial settings. Conventional methods typically rely on either aromatic sulfonyl fluorides through oxidation-fluorination sequences or specialized building blocks like ESF (ethyl sulfonyl fluoride) derivatives, which are not readily adaptable to diverse alkyl substrates. These approaches often require harsh reaction conditions including high temperatures, strong oxidizing agents, or expensive catalysts that complicate process safety and increase production costs. Furthermore, many existing methodologies suffer from poor selectivity, leading to complex product mixtures that require extensive purification steps and reduce overall yield. The limited substrate scope of traditional techniques has particularly hindered the development of diverse alkyl sulfonyl fluoride libraries needed for pharmaceutical research and development. Additionally, most conventional processes are not easily scalable due to safety concerns associated with handling reactive intermediates or toxic byproducts. These limitations have created significant barriers to the widespread adoption of sulfonyl fluorides in industrial applications despite their valuable chemical properties and growing importance in click chemistry applications.
The Novel Approach
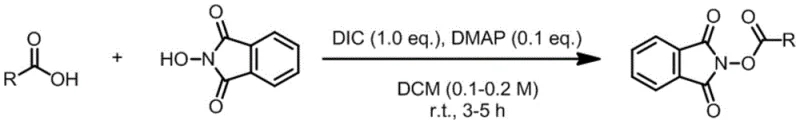
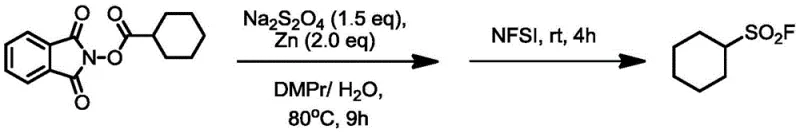
The patented methodology overcomes these limitations through an elegant one-pot two-step process that begins with readily available alkyl carboxylic acids. By first converting these acids to reduced active esters using standard coupling reagents like DIC and DMAP, the process establishes a versatile platform for subsequent transformation. The key innovation lies in the controlled introduction of sulfur dioxide equivalents using stable substitutes like DABSO or Na2S2O4, followed by electrophilic fluorination with reagents such as NFSI under mild reducing conditions. This approach enables precise in-situ construction of the sulfonyl fluoride moiety with exceptional selectivity and yield. The reaction proceeds efficiently at moderate temperatures (80°C) in environmentally friendly solvent systems comprising N,N-dimethylpropionamide and water (5:1 v/v), eliminating the need for hazardous solvents or extreme conditions. Crucially, the process demonstrates remarkable functional group tolerance across diverse alkyl substrates including linear chains, cyclic structures, and complex pharmaceutical intermediates like ibuprofen derivatives. This versatility, combined with straightforward workup procedures and excellent scalability potential, positions this methodology as a transformative advancement for industrial production of sulfonyl fluorides.
Mechanistic Insights into Alkyl Carboxylic Acid to Sulfonyl Fluoride Transformation
The reaction mechanism involves a sophisticated radical-based pathway that begins with single-electron transfer from zinc metal to the reduced active ester, triggering decarboxylation and generating an alkyl radical intermediate. This radical species then reacts with sulfur dioxide equivalents provided by substitutes like Na2S2O4, forming an alkyl sulfonyl radical that subsequently undergoes oxidation and fluorination through interaction with NFSI. The precise control over radical generation and trapping is facilitated by the carefully optimized solvent system comprising N,N-dimethylpropionamide and water, which provides ideal polarity and solvation characteristics for intermediate stabilization. The two-step nature of the process allows for sequential control over sulfur dioxide incorporation and fluorination, preventing unwanted side reactions that commonly plague one-pot methodologies. This mechanistic pathway explains the exceptional selectivity observed across diverse substrate classes, as the radical intermediates are less prone to rearrangement or elimination compared to ionic pathways used in conventional methods.
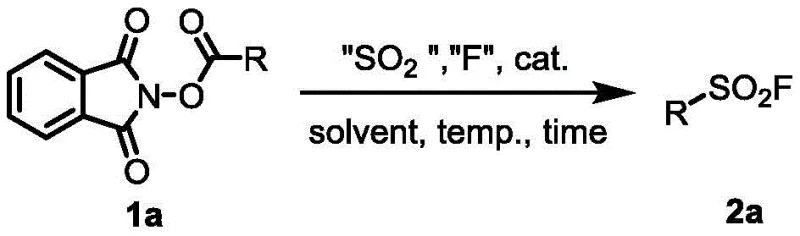
Impurity control is achieved through multiple design elements inherent in this methodology. First, the use of stable sulfur dioxide substitutes eliminates potential side reactions associated with gaseous SO2 handling. Second, the controlled radical mechanism minimizes formation of elimination byproducts that commonly occur in traditional approaches. Third, the optimized solvent system effectively suppresses hydrolysis pathways that could lead to sulfonic acid impurities. The process also benefits from inherent purification through simple aqueous workup procedures that remove metal residues and unreacted starting materials without requiring complex chromatographic separation. This combination of factors results in products with high purity profiles suitable for pharmaceutical applications without extensive post-reaction processing. The methodology's robustness across various substrate types further demonstrates its reliability for producing consistent quality intermediates required in regulated manufacturing environments.
How to Synthesize Alkyl Sulfonyl Fluoride Efficiently
This patented methodology represents a significant advancement in sulfonyl fluoride synthesis by providing a reliable pathway from common carboxylic acid precursors to valuable sulfonyl fluoride products. The process has been optimized through extensive experimentation to deliver consistent results across diverse substrate classes while maintaining excellent scalability potential for industrial implementation. Detailed operational parameters have been established through systematic screening of reaction conditions including solvent composition, temperature profiles, reagent stoichiometry, and atmospheric requirements. The following standardized procedure has been developed based on comprehensive validation studies that confirm both reproducibility and robustness across multiple production scales. This methodology eliminates previous limitations associated with traditional approaches while providing superior control over product quality and yield.
- Prepare reduced active ester by reacting alkyl carboxylic acid with N-hydroxyphthalimide using DIC (1.0 equiv.) and DMAP (0.1 equiv.) in DCM at room temperature for 3-5 hours
- Conduct first step reaction by combining reduced active ester (1 equiv.), sulfur dioxide substitute reagent (1.5 equiv.), and reducing metal (2 equiv.) in DMPr/H₂O solvent system (5: 1 v/v) at 80°C for 9 hours under argon atmosphere
- Complete synthesis by adding electrophilic fluorinating reagent (3 equiv.) at room temperature and allowing reaction to proceed for additional 4 hours before standard aqueous workup
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional sulfonyl fluoride production methods. The process leverages readily available starting materials and common laboratory reagents that are easily sourced through established global supply channels, eliminating dependency on specialized or restricted chemicals that often create supply chain vulnerabilities. The simplified reaction sequence reduces manufacturing complexity while improving overall process reliability, which directly translates to more predictable production timelines and enhanced supply chain resilience. These advantages position this technology as an ideal solution for organizations seeking to secure reliable access to high-quality sulfonyl fluoride intermediates while optimizing their overall cost structure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized precursors significantly reduces raw material costs while simplifying waste stream management. By utilizing common reagents like zinc powder instead of precious metal catalysts, this approach avoids costly metal recovery processes and associated environmental compliance expenses. The aqueous-compatible solvent system further reduces disposal costs compared to traditional methods requiring hazardous organic solvents. Additionally, the high selectivity minimizes purification requirements, leading to substantial savings in downstream processing without compromising product quality.
- Enhanced Supply Chain Reliability: The use of widely available starting materials including common carboxylic acids and standard coupling reagents ensures consistent supply chain access without vulnerability to single-source dependencies. This methodology's compatibility with standard manufacturing equipment eliminates the need for specialized infrastructure investments while maintaining excellent scalability from laboratory to commercial production volumes. The robust nature of the process across diverse substrates provides flexibility to adapt to changing market demands without requiring significant process revalidation or equipment modifications.
- Scalability and Environmental Compliance: The process has been designed with green chemistry principles in mind, featuring aqueous-compatible conditions that minimize environmental impact while maintaining excellent reaction efficiency. The elimination of hazardous reagents and byproducts simplifies regulatory compliance across multiple jurisdictions while reducing waste treatment requirements. This methodology demonstrates exceptional scalability potential from laboratory scale through pilot production to full commercial manufacturing without requiring significant process modifications or safety compromises.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this innovative synthesis methodology based on extensive technical validation and practical application experience. These responses reflect actual performance data gathered during development and scale-up studies conducted under controlled manufacturing conditions.
Q: How does this method improve upon traditional approaches for synthesizing alkyl sulfonyl fluorides?
A: This patented method overcomes significant limitations of conventional approaches by utilizing readily available carboxylic acid precursors instead of specialized building blocks. Unlike traditional methods requiring harsh conditions or expensive catalysts, this process operates under mild conditions with exceptional selectivity for in-situ introduction of sulfonyl fluoride groups across diverse substrate classes including complex pharmaceutical intermediates.
Q: What are the key advantages regarding impurity profile compared to existing methodologies?
A: The radical-based mechanism minimizes formation of elimination byproducts common in traditional approaches while the optimized solvent system suppresses hydrolysis pathways that could lead to sulfonic acid impurities. The aqueous-compatible workup procedure effectively removes metal residues without requiring complex chromatographic separation.
Q: How does this process address scalability concerns for commercial manufacturing?
A: The methodology demonstrates excellent scalability potential due to its use of standard manufacturing equipment, aqueous-compatible conditions, and simplified waste stream management. The robust nature across diverse substrates provides flexibility to adapt to changing market demands without requiring significant process revalidation or equipment modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Sulfonyl Fluoride Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex specialty chemicals including high-value sulfonyl fluoride intermediates. Our stringent purity specifications and rigorous QC labs ensure consistent product quality meeting pharmaceutical industry standards while maintaining exceptional batch-to-batch reproducibility. With deep expertise in SuFEx chemistry applications and process optimization, we provide tailored solutions that address specific client requirements while ensuring regulatory compliance across global markets. Our technical team works closely with customers to develop customized manufacturing approaches that balance cost efficiency with quality requirements for even the most challenging molecular targets.
Discover how our specialized capabilities can support your specific needs through our Customized Cost-Saving Analysis. Contact our technical procurement team today to request specific COA data and route feasibility assessments for your target compounds. We're committed to delivering reliable supply solutions that enhance your competitive position while maintaining the highest standards of quality and service excellence.
