Advanced Synthesis of Alkyl Sulfonyl Fluorides for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of Alkyl Sulfonyl Fluorides for Scalable Pharmaceutical Manufacturing
The landscape of sulfur-fluorine exchange (SuFEx) click chemistry has been revolutionized by the ability to rapidly construct complex molecular architectures with high fidelity. A pivotal advancement in this domain is detailed in Chinese Patent CN113698325A, which discloses a highly efficient method for preparing alkyl sulfonyl fluorides directly from alkyl carboxylic acids. This technology addresses a critical bottleneck in modern medicinal chemistry, where the installation of the sulfonyl fluoride motif onto aliphatic chains has historically been challenging due to the lack of robust, generalizable protocols. By leveraging reductive active esters derived from ubiquitous carboxylic acids, this process enables the in-situ generation of alkyl radicals that capture sulfur dioxide equivalents before undergoing electrophilic fluorination. For global procurement teams and R&D directors seeking reliable alkyl sulfonyl fluoride suppliers, this patent represents a paradigm shift towards more accessible and cost-effective supply chains for these high-value intermediates.
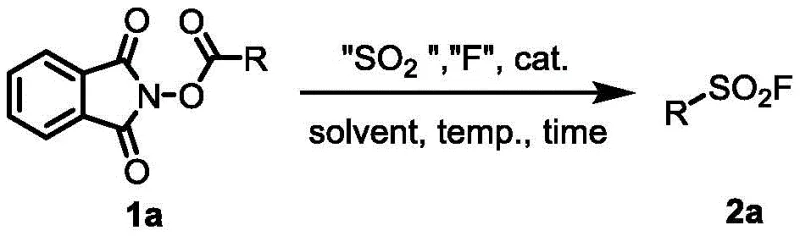
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkyl sulfonyl fluorides has relied heavily on the functionalization of alkyl halides or the oxidative fluorination of thiol precursors, both of which present significant logistical and chemical hurdles. The reliance on alkyl halides often necessitates harsh reaction conditions that can compromise sensitive functional groups elsewhere in the molecule, leading to complex impurity profiles that are difficult to purge during downstream processing. Furthermore, the direct introduction of the sulfonyl fluoride group onto non-activated aliphatic systems typically requires multi-step sequences involving unstable intermediates, which drastically increases the overall cost of goods and extends lead times for material delivery. These conventional pathways frequently suffer from poor atom economy and generate substantial hazardous waste, creating environmental compliance burdens that modern sustainable manufacturing initiatives strive to eliminate. Consequently, the scarcity of efficient methods has limited the widespread adoption of alkyl sulfonyl fluorides in drug discovery campaigns compared to their aromatic counterparts.
The Novel Approach
The methodology outlined in the patent data introduces a streamlined, one-pot two-step strategy that bypasses these historical limitations by utilizing N-hydroxyphthalimide (NHPI) esters as versatile radical precursors. This approach capitalizes on the ease of preparing NHPI esters from commercially available carboxylic acids, effectively turning a vast library of simple building blocks into potential sulfonyl fluoride candidates. The core innovation lies in the use of sodium dithionite as a sulfur dioxide source combined with a reducing metal, which facilitates the gentle generation of alkyl radicals under relatively mild thermal conditions. This radical species is then trapped by the sulfur source and subsequently fluorinated using an electrophilic fluorinating agent like NFSI. The result is a direct, modular transformation that preserves the integrity of the carbon skeleton while installing the valuable SO2F handle with high selectivity. This novel route not only simplifies the synthetic workflow but also significantly enhances the feasibility of producing diverse alkyl sulfonyl fluorides on a commercial scale.
Mechanistic Insights into Reductive Decarboxylative Sulfonyl Fluorination
The mechanistic pathway of this transformation is a sophisticated interplay of single-electron transfer (SET) processes and radical trapping events that ensure high conversion rates. Initially, the NHPI ester undergoes reduction, likely mediated by the zinc powder and sodium dithionite system, triggering a decarboxylation event that releases carbon dioxide and generates a reactive alkyl radical. This radical intermediate is highly transient but is efficiently captured by the sulfur dioxide surrogate present in the reaction mixture, forming an alkylsulfonyl radical species. The presence of water in the solvent system, specifically in a mixture with N,N-dimethylpropionamide (DMPr), appears to play a crucial role in stabilizing the ionic species and facilitating the solubility of the inorganic reagents. Following the sulfur insertion, the addition of the electrophilic fluorinating reagent, such as N-fluorobisbenzenesulfonamide (NFSI), completes the cycle by transferring a fluorine atom to the sulfur center. This sequence avoids the formation of stable sulfinate salts that might require separate activation steps, thereby condensing the synthesis into a single operational unit.
From an impurity control perspective, the mildness of the radical generation step is paramount for maintaining product purity, especially when dealing with complex pharmaceutical intermediates. Unlike strong oxidants or aggressive nucleophiles used in traditional methods, the reductive conditions employed here are compatible with a wide range of functional groups, including ethers, esters, and aromatic rings. The patent data highlights that the reaction can tolerate various substituents on the alkyl chain, suggesting that side reactions such as beta-hydride elimination or over-oxidation are minimized. Furthermore, the use of zinc powder as a stoichiometric reductant helps to scavenge any oxidative byproducts that could degrade the sensitive S-F bond. The careful optimization of the solvent ratio, particularly the 5:1 volume ratio of DMPr to water, ensures that the reaction medium remains homogeneous enough for efficient mass transfer while preventing the hydrolysis of the final sulfonyl fluoride product. This balance is critical for achieving the reported high yields and clean reaction profiles observed in the experimental examples.
How to Synthesize Alkyl Sulfonyl Fluoride Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize waste, making it an ideal candidate for standardized manufacturing protocols. The process begins with the preparation of the NHPI ester, which serves as the stable, isolable precursor for the radical reaction. Once the ester is secured, the subsequent one-pot transformation involves heating the mixture with the sulfur source and reductant, followed by a cooling phase for fluorination. This operational simplicity allows for easy adaptation in standard glass-lined reactors or stainless steel vessels commonly found in pilot plants. The following guide outlines the critical stages of this procedure, emphasizing the specific conditions that have been validated to produce high-quality alkyl sulfonyl fluorides consistently.
- Preparation of Reductive Active Ester: React alkyl carboxylic acid with N-hydroxyphthalimide (NHPI) using DIC and DMAP in DCM at room temperature.
- Radical Generation and SO2 Insertion: Treat the NHPI ester with Na2S2O4 and Zinc powder in DMPr/Water at 80°C under argon for 9 hours.
- Electrophilic Fluorination: Cool to room temperature, add NFSI, and stir for 4 hours to yield the final alkyl sulfonyl fluoride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits that extend beyond mere chemical novelty, directly impacting the bottom line through cost reduction in pharmaceutical intermediates manufacturing. The primary economic driver is the utilization of alkyl carboxylic acids as starting materials, which are among the most abundant and inexpensive feedstocks in the chemical industry. By avoiding the need for specialized sulfur-containing precursors or expensive transition metal catalysts, the raw material costs are significantly lowered, allowing for more competitive pricing structures in long-term supply agreements. Additionally, the elimination of noble metals from the process removes the necessity for costly and time-consuming heavy metal scavenging steps, which are often required to meet stringent regulatory limits for API production. This simplification of the purification train not only reduces solvent consumption but also shortens the overall production cycle time, enhancing the agility of the supply chain to respond to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals such as sodium dithionite and zinc powder, which are available in bulk quantities at low prices. The avoidance of cryogenic conditions or high-pressure equipment further reduces capital expenditure and operational energy costs, making the process inherently scalable without proportional increases in overhead. Moreover, the high atom economy of the decarboxylative approach means that less waste is generated per kilogram of product, lowering disposal fees and environmental levies. These factors combine to create a manufacturing profile that is resilient to fluctuations in raw material markets, ensuring stable pricing for downstream customers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity, as the process is less susceptible to minor variations in temperature or reagent quality compared to sensitive catalytic cycles. The patent data indicates that the reaction can proceed even under non-inert atmospheres, albeit with lower yields, suggesting a degree of forgiveness that is valuable in large-scale operations where perfect exclusion of air is difficult. This tolerance reduces the risk of batch failures and reworks, which are common causes of supply disruptions. Furthermore, the broad substrate scope means that a single manufacturing line can be adapted to produce a variety of alkyl sulfonyl fluorides by simply changing the carboxylic acid feedstock, providing flexibility to manage inventory and fulfill diverse customer orders efficiently.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to tonnage levels is facilitated by the use of common organic solvents like DMPr and water, which are easier to handle and recycle than exotic fluorinated solvents. The absence of toxic gases like sulfur dioxide in its gaseous form, replaced by solid surrogates, greatly improves workplace safety and simplifies regulatory compliance regarding emissions. The aqueous workup described in the examples allows for straightforward separation of organic products from inorganic salts, minimizing the volume of organic waste streams. This alignment with green chemistry principles not only satisfies corporate sustainability goals but also future-proofs the manufacturing process against tightening environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this alkyl sulfonyl fluoride synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction performance and applicability. Understanding these details is essential for R&D teams evaluating this route for their specific project needs and for procurement specialists assessing the feasibility of sourcing these materials.
Q: What are the primary advantages of this sulfonyl fluoride synthesis method?
A: This method utilizes cheap and readily available alkyl carboxylic acids as starting materials, avoiding the need for pre-functionalized sulfur precursors. It operates under mild conditions with high chemoselectivity and achieves significant yields suitable for scale-up.
Q: Is the reaction sensitive to air or moisture?
A: While optimal yields are achieved under inert gas protection (argon), experimental data indicates the reaction can proceed even in air, albeit with reduced efficiency. The use of a DMPr/Water solvent system enhances stability and solubility.
Q: What is the substrate scope for this transformation?
A: The protocol demonstrates broad compatibility with primary, secondary, and tertiary alkyl groups, including cycloalkyls like cyclohexyl and adamantane, as well as arylmethyl derivatives such as ibuprofen precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Sulfonyl Fluoride Supplier
As the demand for sulfur-fluoride containing molecules continues to surge in drug discovery and material science, having a partner capable of delivering high-purity alkyl sulfonyl fluorides is essential for maintaining your development timeline. NINGBO INNO PHARMCHEM leverages deep expertise in process chemistry to translate innovative academic protocols like CN113698325A into robust commercial manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of alkyl sulfonyl fluoride meets the exacting standards required for pharmaceutical applications. We understand the critical nature of these intermediates in SuFEx click chemistry and are committed to supporting your research with materials of the highest quality.
We invite you to engage with our technical team to explore how this advanced synthesis method can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to this carboxylic acid-based route can optimize your budget without compromising on quality. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Contact us today to discuss your requirements for high-purity alkyl sulfonyl fluorides and let us demonstrate our capability as your trusted partner in fine chemical manufacturing.
