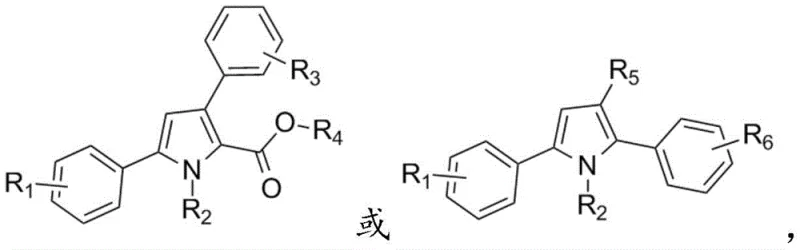
Advanced Synthesis of Polysubstituted Pyrrole Derivatives for Pharmaceutical Applications
Advanced Synthesis of Polysubstituted Pyrrole Derivatives for Pharmaceutical Applications
The field of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and material science industries, particularly regarding the construction of nitrogen-containing heterocycles. A significant breakthrough in this domain is detailed in patent CN110804007B, which discloses a highly efficient preparation method for polysubstituted pyrrole derivatives. Pyrrole rings are ubiquitous structural motifs found in vital biological molecules such as heme, chlorophyll, and vitamin B12, making their synthetic accessibility a priority for R&D teams globally. This innovative technology leverages a sophisticated cascade reaction involving carbon-carbon insertion, cyclization, and [1,5] migration to construct the pyrrole core with exceptional precision. By utilizing either diazo compounds or phenylhydrazone compounds reacting with amino-substituted unsaturated carbonyl compounds, this method overcomes many historical limitations associated with heterocycle synthesis. The result is a series of polysubstituted pyrrole derivatives obtained with high efficiency and yield, offering a robust platform for developing new active pharmaceutical ingredients (APIs) and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole derivatives has relied heavily on classical methodologies such as the Knorr, Paal-Knorr, and Hantzsch syntheses. While these methods established the foundation of heterocyclic chemistry, they often suffer from significant drawbacks when applied to modern drug discovery pipelines. Traditional routes frequently necessitate harsh reaction conditions, including strong acids or high temperatures, which can degrade sensitive functional groups present in complex drug candidates. Furthermore, the starting materials for these conventional methods are often complex to prepare, requiring multiple protection and deprotection steps that drastically reduce overall atom economy. From a supply chain perspective, the reliance on specialized precursors can lead to bottlenecks in raw material availability, increasing lead times and costs. Additionally, many transition-metal catalyzed modern methods reported in recent decades still struggle with the complexity of raw material preparation and the need for ideal synthesis strategies that minimize environmental pollution while maximizing yield.
The Novel Approach
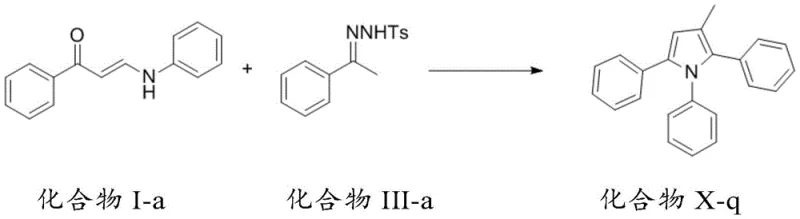
In stark contrast to these legacy methods, the technology described in CN110804007B introduces a streamlined strategy that prioritizes simplicity and efficiency. This novel approach utilizes readily available starting materials that can be obtained through simple reactions, such as the condensation of acetophenone derivatives with amines. The core innovation lies in the tandem reaction sequence that constructs the pyrrole ring in a single operational step from these simple precursors. As illustrated in the reaction scheme below, the method allows for the use of phenylhydrazone compounds as an alternative to diazo species, expanding the substrate scope and safety profile of the synthesis. This flexibility ensures that manufacturers can adapt the process based on the specific availability and cost of local raw materials, thereby enhancing supply chain resilience. The mild reaction conditions, often ranging from room temperature to moderate heating, significantly reduce energy consumption and safety risks associated with high-pressure or high-temperature reactors.
Mechanistic Insights into Silver-Catalyzed Cascade Cyclization
The mechanistic pathway of this synthesis is a testament to modern organometallic chemistry, involving a intricate sequence of carbon-carbon insertion and cyclization events. The reaction typically initiates with the activation of the diazo compound by a metal catalyst, such as silver trifluoromethanesulfonate, generating a reactive metal-carbene intermediate. This electrophilic species then undergoes insertion into the electron-rich double bond of the amino-substituted unsaturated carbonyl compound. Following this initial C-C bond formation, the system undergoes a spontaneous cyclization driven by the nucleophilic attack of the nitrogen atom onto the activated carbonyl or imine functionality. Crucially, the process involves a [1,5] migration cascade that rearranges the molecular skeleton to finalize the aromatic pyrrole system. This cascade nature minimizes the formation of stable intermediates that would otherwise require isolation, thereby reducing solvent usage and processing time. The ability to tune the electronic properties of the substrates through various R-group substitutions allows chemists to finely control the reactivity and regioselectivity of the cyclization.
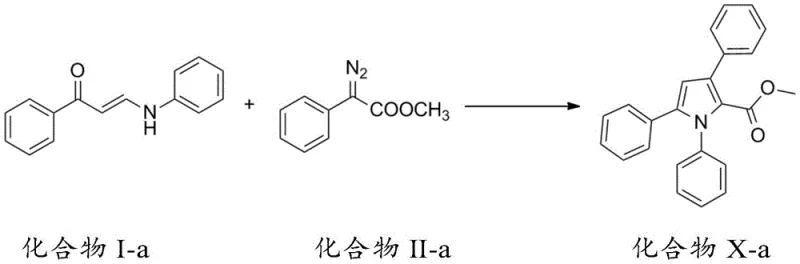
Impurity control is inherently built into this mechanism due to the high specificity of the metal-catalyzed carbene transfer. Unlike radical-based cyclizations which can lead to polymeric byproducts, this ionic or concerted pathway favors the formation of the five-membered heterocycle with high fidelity. The use of mild solvents like dichloromethane or toluene further aids in maintaining a clean reaction profile, facilitating easier downstream purification. For R&D directors concerned with impurity profiles, this method offers a distinct advantage as the major byproducts are typically unreacted starting materials which are easily removed via standard column chromatography or recrystallization. The robustness of the catalytic cycle ensures that even with diverse substituents on the phenyl rings, the reaction proceeds with consistent yields, typically ranging from 70% to over 90% in optimized examples. This predictability is essential for scaling up processes where batch-to-batch consistency is a regulatory requirement.
How to Synthesize Polysubstituted Pyrrole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the addition rates and catalyst loading to maximize the cascade efficiency. The process generally begins with the preparation of the amino-substituted enone precursor, followed by the slow addition of the diazo or hydrazone component to the catalytic mixture. This controlled addition is critical to managing the exothermicity of the carbene formation and preventing dimerization of the diazo species. The patent provides extensive experimental data demonstrating the reproducibility of this method across a wide range of substrates, confirming its utility for generating diverse chemical libraries. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the optimal conditions identified in the patent examples.
- Prepare the amino-substituted unsaturated carbonyl compound (Compound I) by reacting acetophenone derivatives with N,N-dimethylformamide methylal and subsequently with an amine.
- Synthesize the diazo compound (Compound II) via diazotization of the corresponding ester using 4-acetamidobenzenesulfonyl azide and a base like DBU.
- React Compound I and Compound II in dichloromethane with a silver catalyst (e.g., AgOTf) at room temperature, adding the diazo compound dropwise to ensure controlled cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology translates into tangible strategic benefits regarding cost stability and operational flexibility. The primary driver of cost efficiency here is the utilization of commodity chemicals as starting materials. Acetophenone, aniline derivatives, and simple esters are produced on a massive global scale, ensuring a stable supply base that is less susceptible to the volatility seen with exotic heterocyclic building blocks. By eliminating the need for multi-step precursor synthesis, the overall manufacturing timeline is drastically shortened, allowing for faster response to market demands. Furthermore, the mild reaction conditions reduce the burden on facility infrastructure, as specialized high-pressure or cryogenic equipment is not required, lowering both capital expenditure and maintenance costs.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the high atom economy and the avoidance of expensive protecting group chemistry. Since the raw materials are cheap and easy to obtain, the direct material cost per kilogram of the final pyrrole derivative is substantially lower compared to traditional methods. Additionally, the high yields reported (often exceeding 80-90%) mean that less raw material is wasted, directly improving the gross margin of the production process. The elimination of complex purification steps further reduces the consumption of solvents and silica gel, contributing to substantial cost savings in consumables and waste disposal.
- Enhanced Supply Chain Reliability: Relying on widely available feedstocks mitigates the risk of supply disruptions that can plague the pharmaceutical industry. The flexibility to use either diazo compounds or phenylhydrazones provides a contingency plan; if one class of reagent faces supply constraints, the process can be adapted to use the alternative without redesigning the entire synthetic route. This redundancy is invaluable for maintaining continuous production schedules. Moreover, the scalability of the reaction from gram to kilogram scales has been validated, ensuring that the transition from R&D to commercial manufacturing is seamless and predictable.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The reaction operates at or near ambient pressure and moderate temperatures, reducing energy consumption and the carbon footprint of the manufacturing process. The solvents used, such as toluene or dichloromethane, are standard industrial solvents with well-established recovery and recycling protocols. The high efficiency of the reaction minimizes the generation of hazardous waste streams, simplifying compliance with increasingly stringent environmental regulations. This makes the process not only commercially attractive but also sustainable for long-term operation.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational requirements. These insights are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these nuances is critical for assessing the feasibility of integrating this chemistry into existing production lines or new drug development programs.
Q: What are the primary advantages of this pyrrole synthesis method over traditional Knorr synthesis?
A: Unlike traditional Knorr or Paal-Knorr syntheses which often require harsh acidic conditions and produce significant waste, this novel cascade method operates under mild temperatures (0-60°C) and utilizes readily available starting materials, resulting in higher atom economy and simpler purification processes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the preparation method is suitable for large-scale production due to its mild reaction conditions, environmental friendliness, and the use of cheap, easily obtainable raw materials that do not require complex pre-functionalization.
Q: What types of catalysts are compatible with this cyclization reaction?
A: The process demonstrates broad catalyst compatibility, effectively utilizing various metal catalysts including palladium acetate, rhodium acetate dimer, and notably silver salts such as silver trifluoromethanesulfonate, silver carbonate, and silver nitrate at loadings of 3mol% to 10mol%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development. Our team of expert chemists has extensively analyzed this patent technology and possesses the capability to implement these advanced cascade reactions at scale. We offer comprehensive CDMO services that cover everything from route optimization to commercial manufacturing. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of polysubstituted pyrrole derivatives meets the highest international standards for impurity profiles and physical properties.
We invite potential partners to leverage our technical expertise to accelerate their drug discovery timelines. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that identifies specific opportunities to optimize your supply chain using this novel synthesis method. We encourage you to contact our technical procurement team to request specific COA data for relevant derivatives and to discuss route feasibility assessments tailored to your project's unique requirements. Let us help you transform this innovative chemistry into a commercial reality.
