Scalable Production of Chiral Oxazolidinones via Novel Lithium-Salt Catalyzed Reduction
Scalable Production of Chiral Oxazolidinones via Novel Lithium-Salt Catalyzed Reduction
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for synthesizing high-value chiral intermediates. A recent technological breakthrough documented in patent CN113620897A introduces a highly efficient preparation method for oxazolidinone compounds, a class of molecules critical for antibiotic development and asymmetric synthesis. This innovation specifically addresses the long-standing challenges associated with the reduction of aromatic amino acids to amino alcohols, a pivotal step in constructing the oxazolidinone core. By leveraging a novel lithium-salt promoted sodium borohydride reduction system, this process eliminates the need for hazardous borane complexes or expensive lithium aluminum hydride reagents. For R&D directors and procurement managers alike, this represents a significant leap forward in process safety and economic viability, offering a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxazolidinones has relied on several established reduction protocols, each carrying distinct disadvantages that hinder large-scale industrial adoption. One common approach involves the direct reduction of amino acids using borane complexes. As illustrated in the reaction scheme below, while effective on a small scale, this method poses severe safety risks due to the in-situ generation of hydrogen gas, which leads to violent temperature fluctuations that are difficult to control in large reactors. Furthermore, borane solutions in tetrahydrofuran (THF) typically possess low concentrations, necessitating the use of vast volumes of solvent and driving up both material and waste disposal costs significantly.
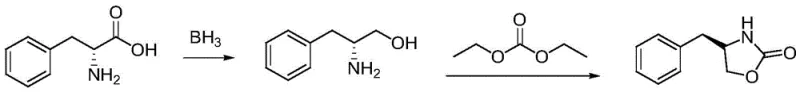
Alternative methods utilizing sodium borohydride activated by iodine have also been explored, yet they suffer from poor yields due to the high water solubility of the resulting amino alcohol products, complicating isolation and purification. Additionally, processes employing lithium aluminum hydride (LiAlH4), often in conjunction with Lewis acids like benzyl chloride, achieve high yields but are economically prohibitive for bulk manufacturing due to the high cost of the reductant and the stringent safety measures required for handling such reactive species. These conventional limitations create substantial bottlenecks in the supply chain for reliable pharmaceutical intermediate suppliers, necessitating a safer and more economical alternative.
The Novel Approach
The methodology disclosed in patent CN113620897A fundamentally reimagines the reduction step by introducing a catalytic amount of lithium salt into a sodium borohydride system utilizing an ethanol-water solvent mixture. This strategic modification allows for the complete dissolution of the reductant and the dissociation of lithium ions, which actively participate in the reaction mechanism to lower the activation energy. Unlike the hazardous borane route or the costly LiAlH4 pathway, this new approach operates under mild conditions with stable thermal profiles. The result is a streamlined three-step process—esterification, reduction, and cyclization—that delivers high-purity oxazolidinones with drastically simplified post-treatment procedures, making it ideally suited for cost reduction in API manufacturing.
Mechanistic Insights into Lithium-Salt Promoted Reduction
The core innovation of this technology lies in the synergistic interaction between sodium borohydride and the added lithium salt within a polar protic solvent system. In traditional sodium borohydride reductions, steric hindrance around the carbonyl group of the ester intermediate often necessitates a large excess of the reductant, typically ranging from 3 to 10 equivalents, to drive the reaction to completion. However, the introduction of lithium salts, such as lithium chloride or lithium bromide, alters the solvation environment. In the ethanol-water mixture, the lithium salt dissociates to form free lithium ions which coordinate with the carbonyl oxygen, effectively activating the substrate and reducing the steric barrier for the incoming hydride ion.
This mechanistic enhancement allows the reaction to proceed efficiently with only 2 equivalents of sodium borohydride, a substantial reduction in reagent consumption compared to standard protocols. Furthermore, the choice of ethanol-water as the solvent ensures that the lithium borohydride species formed in situ remains soluble and active, preventing the precipitation issues seen in other systems. From an impurity control perspective, the mild nature of this reduction minimizes side reactions such as over-reduction or racemization, ensuring that the chiral integrity of the starting aromatic amino acid is preserved throughout the transformation. This level of control is paramount for producing high-purity OLED material precursors or pharmaceutical intermediates where enantiomeric excess is a critical quality attribute.
How to Synthesize Oxazolidinone Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for generating 4-substituted-2-oxazolidinones, such as (S)-4-phenyl-2-oxazolidinone or (S)-4-benzyl-2-oxazolidinone. The process begins with the esterification of the aromatic amino acid using thionyl chloride in methanol, followed by the critical lithium-promoted reduction step, and concludes with a cyclization reaction using diethyl carbonate. This sequence avoids the use of phosgene or its toxic analogues, aligning with modern green chemistry principles. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and workup procedures required to achieve optimal yields and purity, are provided in the technical guide below.
- Perform esterification of aromatic amino acids using methanol and thionyl chloride at 50-60°C.
- Execute reduction using sodium borohydride and a catalytic lithium salt in an ethanol-water solvent system at low temperature.
- Complete cyclization with diethyl carbonate and sodium methoxide in toluene at 100°C to form the final oxazolidinone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling economic and operational benefits that extend beyond simple reagent substitution. The primary advantage stems from the drastic optimization of raw material usage, specifically the reduction of sodium borohydride equivalents. By lowering the stoichiometric requirement from the industry standard of 3-10 equivalents down to merely 2 equivalents, the direct material cost for the reduction step is significantly curtailed. This efficiency gain is compounded by the elimination of expensive reagents like lithium aluminum hydride and the avoidance of specialized solvents like anhydrous THF, leading to substantial cost savings in the overall manufacturing budget without compromising product quality.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the minimized consumption of reducing agents and the use of commodity chemicals. Sodium borohydride and lithium chloride are widely available and inexpensive compared to borane-THF complexes or LiAlH4. Furthermore, the ability to use ethanol-water mixtures instead of strictly anhydrous organic solvents reduces solvent procurement and recycling costs. The simplified workup procedure, which avoids complex extraction sequences often required to remove metal residues from LiAlH4 reactions, further lowers labor and utility expenses, resulting in a leaner, more cost-effective production model.
- Enhanced Supply Chain Reliability: Adopting this method mitigates supply chain risks associated with hazardous or specialized reagents. Borane solutions often have limited shelf lives and require strict temperature-controlled logistics, whereas sodium borohydride and lithium salts are stable, non-hazardous solids that are easily sourced from multiple global suppliers. This diversification of the supply base ensures continuity of supply even during market fluctuations. Additionally, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by safety incidents or equipment failures related to exothermic runaways, guaranteeing consistent delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The safety profile of this process makes it inherently more scalable than traditional borane reductions. The absence of hydrogen gas generation eliminates the need for specialized venting systems and explosion-proof infrastructure, facilitating easier technology transfer from pilot plant to commercial scale. From an environmental standpoint, the use of ethanol and water reduces the volatile organic compound (VOC) load of the process waste streams. The reduced stoichiometric excess of reagents also translates to less chemical waste requiring treatment, simplifying compliance with increasingly stringent environmental regulations and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazolidinone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industrial standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: How does the lithium salt improve the reduction efficiency?
A: The lithium salt dissociates in the ethanol-water solvent to form free ions, which effectively reduces steric hindrance during the hydride transfer, allowing the sodium borohydride dosage to be lowered to just 2 equivalents.
Q: Why is this method safer than traditional borane reduction?
A: Unlike borane reduction which generates hazardous hydrogen gas and causes violent temperature spikes, this sodium borohydride-based method operates under mild conditions with stable thermal profiles suitable for industrial reactors.
Q: What are the cost benefits of this new process?
A: By reducing the requirement for sodium borohydride from the typical 3-10 equivalents down to only 2 equivalents and avoiding expensive reagents like lithium aluminum hydride, the overall material cost is significantly reduced.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the lithium-salt promoted reduction are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of oxazolidinone compound meets the exacting standards required for API synthesis and chiral auxiliary applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to request specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
