Scalable Production of Soluble Polymer-Supported Chiral Oxazolidinones for Asymmetric Synthesis
Introduction to Advanced Soluble Polymer-Supported Chiral Technologies
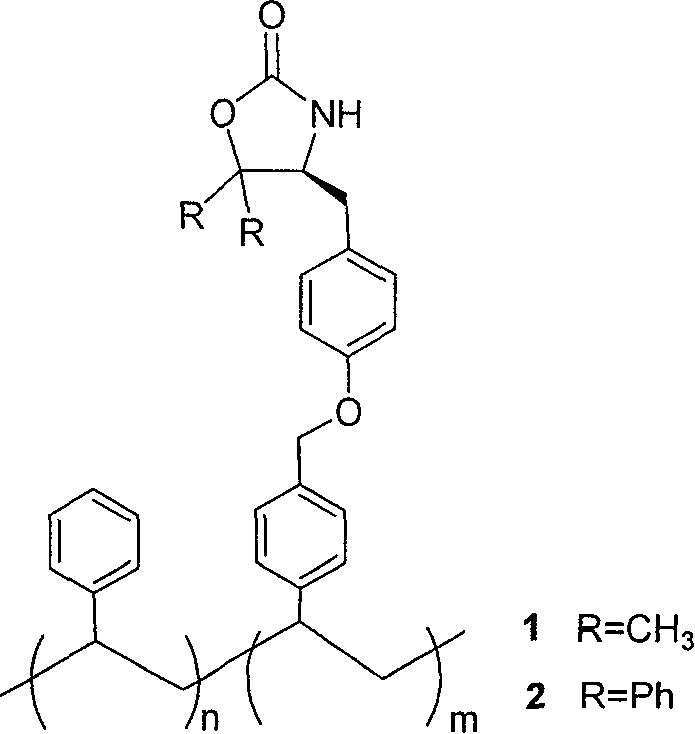
The landscape of asymmetric synthesis has evolved significantly since the pioneering work on solid-phase synthesis, yet challenges regarding reaction kinetics and purification persist. Patent CN100395270C introduces a sophisticated class of linear polystyrene supported (4S)-5,5-disubstituted oxazolidinones, specifically designed to bridge the gap between homogeneous and heterogeneous catalysis. By utilizing natural L-tyrosine as a chiral pool starting material, this technology offers a robust platform for inducing asymmetry in organic transformations while maintaining the ease of purification associated with polymer-supported reagents. The innovation lies in the solubility of the polymer backbone, which allows reactions to proceed under homogeneous conditions, thereby accelerating reaction rates and facilitating real-time monitoring.
Furthermore, the structural integrity of the oxazolidinone ring is reinforced through 5,5-disubstitution, addressing a critical failure point in traditional Evans auxiliaries where ring-opening often occurs during cleavage. This patent details the preparation of two distinct variants: the 5,5-dimethyl and 5,5-diphenyl derivatives, both anchored to a linear polystyrene chain. For R&D directors seeking reliable pharmaceutical intermediates supplier partnerships, understanding the mechanistic advantages of these soluble supports is crucial for optimizing chiral drug synthesis pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase organic synthesis (SPOS) typically relies on insoluble cross-linked resins, such as Merrifield resin, which impose significant mass transfer limitations on the reaction system. In these heterogeneous environments, reactants must diffuse into the polymer matrix to access the active sites, often resulting in prolonged reaction times and incomplete conversions that necessitate large excesses of reagents. Additionally, the insoluble nature of the support precludes the use of standard analytical techniques like thin-layer chromatography (TLC) or solution-phase NMR for monitoring reaction progress, forcing chemists to rely on indirect methods or cleave test samples. This lack of visibility increases the risk of batch failures and complicates the optimization of reaction parameters for complex asymmetric inductions.
The Novel Approach
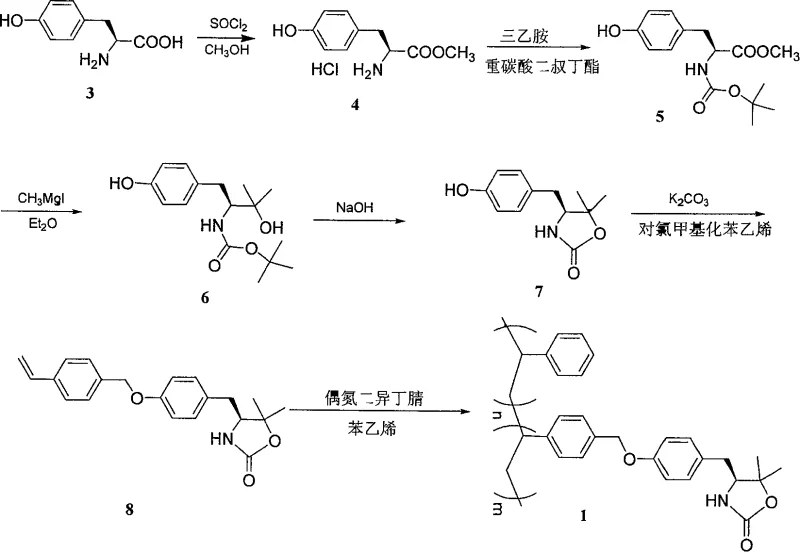
The methodology outlined in the patent circumvents these diffusion barriers by employing linear polystyrene, a soluble polymer support that renders the entire reaction mixture homogeneous. This transition to a soluble phase ensures that the chiral auxiliary is fully accessible to reagents, mimicking the kinetics of small-molecule solution chemistry while retaining the separation benefits of polymer support. The synthesis begins with the functionalization of L-tyrosine, proceeding through esterification and protection before the critical Grignard addition establishes the 5,5-substitution pattern. Subsequent cyclization and etherification with p-chloromethylstyrene install the polymerizable handle, culminating in a copolymerization with styrene to generate the final soluble reagent.
Mechanistic Insights into 5,5-Disubstituted Oxazolidinone Stability
The core innovation of this technology resides in the strategic substitution at the 5-position of the oxazolidinone ring, which fundamentally alters the stability profile of the chiral auxiliary during the cleavage phase. In conventional 5-unsubstituted oxazolidinones, the release of the chiral product often involves nucleophilic attack that can inadvertently open the oxazolidinone ring, destroying the auxiliary and complicating its recovery. By introducing bulky groups such as two methyl groups or two phenyl groups at the 5-position, the patent leverages steric hindrance to protect the carbonyl carbon from unwanted nucleophilic assault during product liberation. This structural reinforcement ensures that the chiral integrity of the auxiliary is preserved, allowing it to be recovered and potentially recycled for subsequent batches without significant degradation.
From a mechanistic standpoint, the synthesis utilizes a Grignard addition to an ester intermediate to generate a tertiary alcohol, which then undergoes intramolecular cyclization to form the oxazolidinone ring. The use of L-tyrosine ensures that the stereochemistry at the 4-position is fixed as (4S), providing a well-defined chiral environment for inducing asymmetry in downstream reactions. The soluble linear polystyrene backbone acts as a "liquid phase" anchor, ensuring that the steric bulk of the auxiliary does not impede the approach of electrophiles to the enolate formed during asymmetric alkylation or acylation. This balance between steric protection of the ring and accessibility of the reactive site is critical for achieving high diastereoselectivity in complex molecule synthesis.
How to Synthesize Linear Polystyrene Supported Oxazolidinone Efficiently
The preparation of these advanced chiral auxiliaries follows a logical sequence of functional group transformations designed to maximize yield and purity at each stage. Starting from the abundant natural amino acid L-tyrosine, the process involves initial esterification to protect the carboxylic acid, followed by amine protection to prevent side reactions during the Grignard step. The subsequent addition of organometallic reagents creates the quaternary center necessary for ring stability, which is then closed under basic conditions. Finally, the attachment of the vinyl group allows for controlled radical polymerization with styrene, tuning the molecular weight and solubility of the final product. Detailed standardized synthesis steps are provided in the guide below.
- Esterify L-Tyrosine with thionyl chloride in methanol, followed by Boc protection using di-tert-butyl dicarbonate.
- Perform Grignard addition using methyl iodide or bromobenzene to introduce 5,5-substituents, followed by base-catalyzed cyclization to form the oxazolidinone ring.
- Etherify the phenolic hydroxyl with p-chloromethylstyrene and copolymerize with styrene using AIBN initiator to yield the final soluble polymer support.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this soluble polymer-supported technology offers tangible benefits in terms of operational efficiency and cost management. The ability to perform reactions under homogeneous conditions significantly reduces the reaction time compared to traditional insoluble resins, leading to faster throughput and better utilization of reactor capacity. Furthermore, the simplified workup procedure, which often involves simple precipitation of the polymer to separate it from small molecule byproducts, eliminates the need for extensive chromatographic purification steps. This streamlining of the downstream processing directly translates to reduced solvent consumption and lower waste disposal costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic viability of chiral synthesis is often hindered by the high cost of chiral auxiliaries and the complexity of their recovery. This technology addresses this by designing an auxiliary that is robust against degradation, enabling multiple reuse cycles which drastically lowers the effective cost per kilogram of the final chiral product. Additionally, the use of commodity chemicals like L-tyrosine and styrene as starting materials ensures a stable and affordable supply chain for raw materials, mitigating the risk of price volatility associated with exotic chiral catalysts.
- Enhanced Supply Chain Reliability: Relying on natural amino acids as the chiral source provides a significant advantage in supply security, as L-tyrosine is produced on a massive industrial scale for the food and feed industries. This abundance ensures that production of the chiral auxiliary is not bottlenecked by the availability of rare earth metals or complex synthetic precursors. Moreover, the synthetic route is robust and scalable, avoiding sensitive conditions that might lead to batch-to-batch variability, thus ensuring a consistent supply of high-quality reagents for critical pharmaceutical campaigns.
- Scalability and Environmental Compliance: The transition from heterogeneous to homogeneous kinetics facilitates easier scale-up, as heat and mass transfer issues common in slurry reactors are minimized. The simplified purification strategy, relying on precipitation rather than column chromatography, significantly reduces the volume of organic solvents required for isolation. This reduction in solvent intensity not only lowers operational costs but also simplifies compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of soluble polymer-supported chiral auxiliaries in industrial settings. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on performance and handling. Understanding these nuances is essential for integrating this technology into existing manufacturing workflows effectively.
Q: What is the primary advantage of using linear polystyrene support over insoluble resins?
A: Linear polystyrene allows for homogeneous reaction conditions, significantly improving reaction kinetics and enabling standard solution-phase monitoring techniques like TLC and NMR, which are difficult with insoluble solid-phase synthesis.
Q: Why are 5,5-disubstituted oxazolidinones preferred in this design?
A: The 5,5-dimethyl or 5,5-diphenyl substitution provides steric bulk that prevents unwanted ring-opening side reactions when the chiral product is cleaved from the auxiliary, ensuring higher integrity of the recovered reagent.
Q: Can the chiral auxiliary be recovered and reused?
A: Yes, the design specifically facilitates the recovery of the chiral auxiliary after the asymmetric induction step, allowing for multiple cycles of use which drastically reduces the effective cost per batch of chiral product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral auxiliaries play in the efficient synthesis of complex active pharmaceutical ingredients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our polymer-supported reagents meets the exacting standards required for GMP-compliant drug substance production.
We invite you to collaborate with us to leverage this advanced technology for your next chiral synthesis project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific process needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our soluble oxazolidinone solutions can enhance your production efficiency and reduce overall manufacturing costs.
