Advanced Pd/Cu Catalyzed Synthesis of Chiral Tetrahydroquinoline Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds, particularly those possessing multiple chiral centers which are critical for biological activity. Patent CN116354930A introduces a groundbreaking methodology for the synthesis of chiral substituted tetrahydroquinolines, a privileged structure found in numerous bioactive natural products and drug candidates exhibiting anticancer, antiviral, and antidiabetic properties. This innovation leverages a synergistic Palladium and Copper catalytic system to facilitate the coupling of amino acid derivatives with ortho-haloaryl halides. Unlike traditional approaches that often suffer from harsh conditions or poor stereocontrol, this technique operates under relatively mild thermal conditions while maintaining exceptional functional group tolerance. For R&D directors and process chemists, this represents a significant leap forward in accessing diverse chemical space for drug discovery programs. The ability to construct the 1,2,3,4-tetrahydroquinoline core in a single step from abundant amino acid precursors not only streamlines the synthetic route but also inherently embeds chirality into the molecular framework, addressing a persistent challenge in asymmetric synthesis.
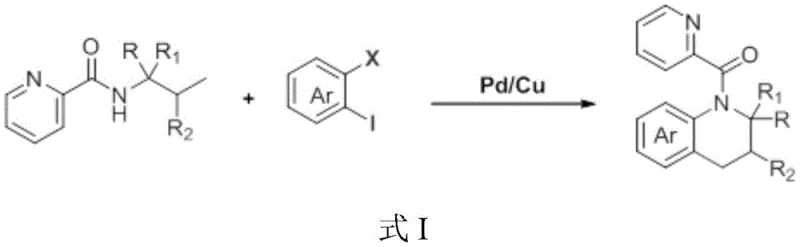
Historically, the construction of the tetrahydroquinoline skeleton has relied heavily on three predominant strategies, each carrying distinct limitations that hinder efficient commercial manufacturing. The first approach involves the direct reduction of quinolines, which typically necessitates the use of noble metal catalysts under high-pressure hydrogenation conditions, posing significant safety risks and equipment costs for large-scale operations. The second method, reductive intramolecular cyclization, often requires palladium on carbon and similar hydrogenation setups, suffering from similar safety and scalability constraints. The third strategy, intermolecular cycloaddition between aromatic imines and olefins, frequently encounters issues with regioselectivity and atom economy, leading to complex purification processes and lower overall yields. These conventional methods often struggle with substrate applicability, particularly when introducing diverse functional groups or specific stereochemical configurations required for modern API development. Consequently, there has been an urgent demand within the fine chemical sector for a safer, more step-economical, and versatile synthetic protocol that can bypass these historical bottlenecks while delivering high-purity intermediates.
The novel approach detailed in the patent data fundamentally shifts the paradigm by utilizing amino acid derivatives as the primary chiral building blocks. This strategy capitalizes on the inherent chirality of naturally occurring or easily accessible amino acids, transferring this stereochemical information directly to the tetrahydroquinoline product through a cascade cyclization process. The reaction employs a dual-metal catalytic system comprising Pd(OAc)2 and CuI, supported by Ag2CO3 as an additive and NaOAc as a base, all dissolved in a mixed solvent system of toluene and tert-amyl alcohol. This specific combination of reagents facilitates a smooth C-N bond formation and subsequent ring closure without the need for external hydrogen gas or extreme pressures. The operational simplicity is further enhanced by the use of standard heating conditions, typically around 140°C, which are easily manageable in standard glass-lined reactors used in commercial plants. This method effectively transforms simple, commercially available starting materials into complex, high-value heterocyclic intermediates with remarkable efficiency.
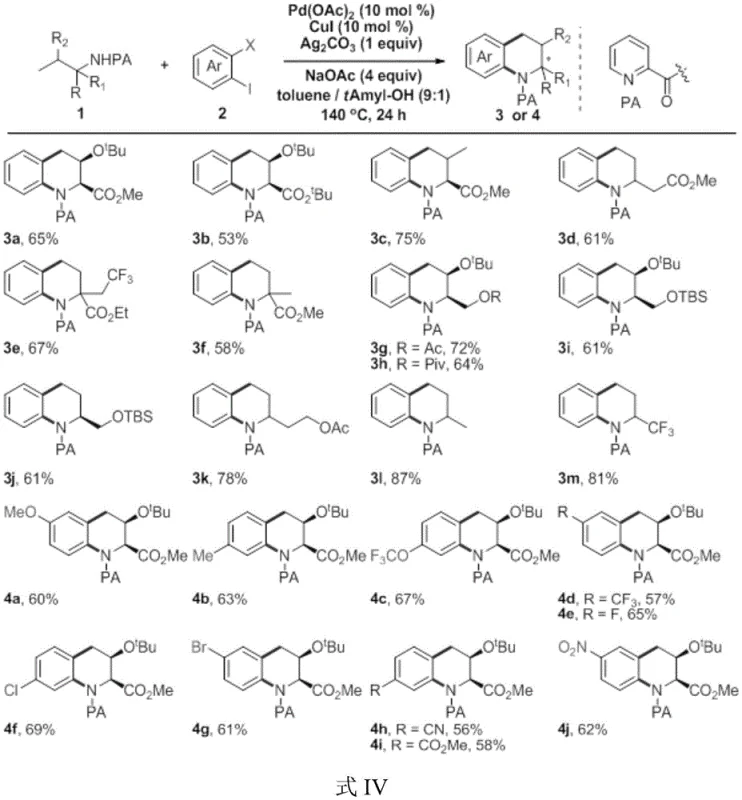
Mechanistically, the success of this transformation relies on the intricate synergy between the palladium and copper species. The palladium catalyst likely initiates the cycle through oxidative addition into the carbon-halogen bond of the ortho-haloaryl halide, generating a reactive organopalladium intermediate. Simultaneously, the copper species may assist in activating the amide nitrogen or facilitating the transmetallation step, ensuring rapid turnover and minimizing side reactions. The presence of silver carbonate acts as a halide scavenger, driving the equilibrium forward and preventing catalyst deactivation. From an impurity control perspective, this mechanism is highly advantageous because it avoids the formation of over-reduced byproducts often seen in hydrogenation methods. The specificity of the C-H activation and cyclization steps ensures that the final product profile is clean, significantly reducing the burden on downstream purification units. For quality control teams, this translates to a more predictable impurity profile, making it easier to establish robust specifications for GMP manufacturing. The retention of stereochemistry from the amino acid precursor is preserved throughout the catalytic cycle, ensuring that the optical purity of the final tetrahydroquinoline derivative remains high, which is critical for regulatory approval of chiral drugs.
How to Synthesize Chiral Tetrahydroquinoline Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The process begins with the precise weighing of the catalytic components, specifically Pd(OAc)2 and CuI at 10 mol% loading each, along with one equivalent of Ag2CO3 and four equivalents of NaOAc. These solids are introduced into a dry reaction vessel equipped with magnetic stirring to ensure homogeneous suspension. The substrate mixture, consisting of the amino acid-derived amide and the substituted ortho-bromoiodobenzene, is dissolved in a 9:1 volume ratio of toluene to tert-amyl alcohol. This specific solvent ratio is crucial for solubility and reaction kinetics. Once the mixture is injected into the reactor, the system is sealed and heated to 140°C for approximately 24 hours. Post-reaction workup involves standard extraction and column chromatography techniques to isolate the pure tetrahydroquinoline product. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Prepare the catalytic system by mixing Pd(OAc)2, CuI, Ag2CO3, and NaOAc in a dry reaction vessel.
- Dissolve the amide substrate and substituted ortho-bromoiodobenzene in a toluene and tert-amyl alcohol mixture.
- Heat the reaction mixture to 140°C for 24 hours, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic benefits that directly impact the bottom line and operational resilience. The shift away from high-pressure hydrogenation and noble metal catalysts like platinum or rhodium significantly reduces the capital expenditure required for specialized reactor infrastructure. By utilizing earth-abundant copper alongside palladium, the overall catalyst cost is optimized, and the reliance on scarce precious metals is mitigated. Furthermore, the use of amino acid derivatives as starting materials leverages a well-established global supply chain for bulk chemicals, ensuring consistent availability and price stability compared to exotic heterocyclic precursors. This stability is vital for long-term supply agreements and risk management strategies, allowing procurement managers to forecast costs with greater accuracy and avoid volatility associated with niche reagents.
- Cost Reduction in Manufacturing: The elimination of high-pressure hydrogenation equipment and the reduction in catalyst loading contribute to a leaner manufacturing cost structure. By avoiding the need for expensive hydrogen gas infrastructure and the associated safety protocols, facilities can operate with lower overheads. Additionally, the one-step nature of the synthesis reduces the number of unit operations, saving on labor, energy, and solvent consumption. The high atom economy of the reaction means less waste generation, which subsequently lowers waste disposal costs. These cumulative efficiencies result in a more competitive cost of goods sold (COGS), enabling pharmaceutical companies to price their final therapies more aggressively or improve their profit margins without compromising quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, tert-amyl alcohol, and sodium acetate ensures that the supply chain is robust against disruptions. Unlike specialized reagents that may have single-source suppliers, the inputs for this process are widely available from multiple global vendors. This diversification minimizes the risk of production stoppages due to raw material shortages. Moreover, the mild reaction conditions reduce the logistical hazards associated with transporting and storing hazardous materials, simplifying compliance with transportation regulations. For supply chain heads, this translates to shorter lead times and a more agile response to market demand fluctuations, ensuring continuous production flow even during periods of global supply constraint.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, moving seamlessly from gram-scale discovery to multi-ton commercial production. The solvent system used is compatible with standard recovery and recycling protocols, aligning with green chemistry principles and reducing the environmental footprint. The absence of heavy metal waste streams typical of other catalytic methods simplifies effluent treatment and regulatory compliance. As environmental regulations become increasingly stringent, adopting a synthesis route that minimizes toxic waste and energy consumption provides a significant competitive advantage. This sustainability profile not only meets current regulatory requirements but also future-proofs the manufacturing process against evolving environmental standards, securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetrahydroquinoline synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for stakeholders. Understanding these nuances is essential for making informed decisions about technology adoption and process integration. The answers provided reflect the robust data supporting the efficacy and versatility of this catalytic system.
Q: What are the primary advantages of this Pd/Cu catalyzed method over traditional quinoline reduction?
A: This method utilizes readily available amino acid derivatives as chiral sources, avoiding the need for high-pressure hydrogenation and expensive noble metal catalysts typically required for direct quinoline reduction, thereby enhancing operational safety and atom economy.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process employs robust reaction conditions with common organic solvents like toluene and tert-amyl alcohol, and avoids sensitive reagents, making it highly adaptable for commercial scale-up from kilogram to multi-ton quantities.
Q: How does this method ensure high stereochemical purity?
A: By using chiral amino acid derivatives as the starting substrates, the chirality is effectively transferred to the tetrahydroquinoline skeleton during the cyclization process, ensuring high enantiomeric excess without requiring complex resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd/Cu catalyzed synthesis route for the next generation of pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this chemistry, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for clinical and commercial applications. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our integrated approach ensures full traceability and compliance throughout the manufacturing lifecycle.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data for related intermediates and comprehensive route feasibility assessments. Let us help you accelerate your timeline to market with a reliable, scalable, and cost-effective supply of high-quality tetrahydroquinoline derivatives.
