Advanced Manufacturing of Tetrahydroquinoline Derivatives Using In Situ Generated Lewis Acid Catalysts
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly 1,2,3,4-tetrahydroquinoline derivatives, which serve as critical scaffolds in numerous bioactive molecules. A significant technological breakthrough in this domain is detailed in patent CN102993094A, which discloses a novel synthetic strategy utilizing an in situ generated Lewis acid system. This innovation addresses long-standing challenges in the Povarov reaction, specifically the difficulty of employing aliphatic aldehydes and the reliance on expensive or hazardous catalysts. By leveraging a redox combination of stannous chloride (SnCl2) and an oxidant, the process generates the active tin tetrachloride (SnCl4) promoter directly within the reaction medium. This approach not only simplifies the operational workflow but also enhances the economic viability of producing high-purity tetrahydroquinoline derivatives, positioning it as a highly attractive route for reliable pharmaceutical intermediate supplier networks aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoline derivatives via the aza-Diels-Alder cycloaddition, commonly known as the Povarov reaction, has been plagued by significant technical and economic hurdles. Traditional protocols often rely on strong Lewis acids such as boron trifluoride etherate (BF3·OEt2) or expensive rare earth metal triflates like scandium triflate (Sc(OTf)3) and dysprosium triflate (Dy(OTf)3). The use of BF3·OEt2 presents severe safety and handling issues, as it hydrolyzes immediately upon exposure to atmospheric moisture, releasing toxic fluorochemical fumes and exhibiting high corrosivity that damages production equipment. Furthermore, rare earth catalysts, while effective, impose a heavy financial burden due to their exorbitant cost and the complexity associated with their recovery and recycling. Additionally, conventional methods struggle significantly when aliphatic aldehydes are employed as substrates; the resulting aliphatic imines are notoriously unstable under acidic conditions, prone to rapid hydrolysis or polymerization, which drastically limits the structural diversity of the accessible chemical space for drug discovery teams.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN102993094A introduces a paradigm shift by utilizing a cost-effective SnCl2 and oxidant combination to promote the reaction. This system ingeniously generates the active Lewis acid species in situ, thereby circumventing the need for handling hazardous anhydrous SnCl4 directly or purchasing prohibitively expensive rare earth salts. The method demonstrates exceptional versatility, successfully accommodating both aromatic and aliphatic aldehydes, which was previously a major bottleneck in the field. By stabilizing the reaction environment through this unique redox promotion, the process achieves high yields under mild conditions, typically ranging from -20°C to 110°C, using common organic solvents. This advancement not only broadens the substrate scope for R&D chemists but also offers a streamlined, safer, and more economically sustainable pathway for the commercial production of complex nitrogen heterocycles.
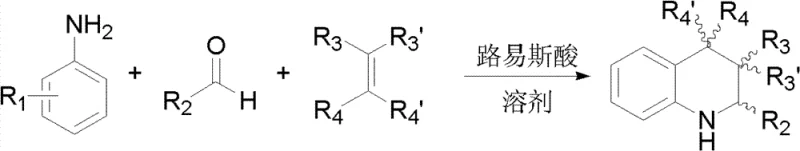
Mechanistic Insights into SnCl2/Oxidant Catalyzed Aza-Diels-Alder Cyclization
The core mechanistic advantage of this synthesis lies in the controlled in situ oxidation of tin(II) chloride to tin(IV) chloride, which acts as the potent Lewis acid catalyst driving the cycloaddition. In the reaction mixture, the SnCl2 reacts with the selected oxidant—such as FeCl3, CuCl2, KMnO4, or hydrogen peroxide—to generate SnCl4 directly within the solvent matrix. This in situ generation ensures that the active catalytic species is formed in the immediate presence of the substrates, minimizing decomposition pathways that might occur if pre-formed SnCl4 were added to a sensitive mixture. The generated SnCl4 coordinates with the carbonyl oxygen of the aldehyde and the nitrogen of the aniline, facilitating the formation of the N-aryl imine intermediate. Crucially, because this imine formation and subsequent cycloaddition occur in a one-pot sequence without isolation, the transient and unstable aliphatic imines are consumed rapidly by the electron-rich olefin before they can undergo hydrolysis or polymerization, thus preserving the integrity of the reaction pathway and maximizing atom economy.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-catalyzed routes. The mild nature of the redox-generated catalyst system reduces the incidence of side reactions such as oligomerization of the olefin or over-alkylation of the aniline, which are common when using super-acidic promoters. The specific choice of oxidant allows for fine-tuning the oxidative potential of the system; for instance, using FeCl3 as the oxidant creates a synergistic effect where iron species may also participate in Lewis acid catalysis, further enhancing reaction rates without introducing exotic contaminants. Furthermore, the reaction conditions allow for the use of diverse substituents on the aniline ring, including electron-withdrawing groups like nitro or halogen, and electron-donating groups like methoxy, without significant loss in efficiency. This robustness ensures a cleaner crude reaction profile, simplifying downstream purification processes such as column chromatography or crystallization, which is essential for meeting the stringent purity specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Tetrahydroquinoline Derivatives Efficiently
To implement this advanced synthesis route effectively, operators must adhere to precise stoichiometric ratios and temperature controls as outlined in the patent embodiments. The process begins with the preparation of the catalytic promoter system, where SnCl2 and the oxidant are dissolved in a suitable organic solvent such as dichloromethane, 1,2-dichloroethane, or toluene. It is critical to maintain the molar ratio of SnCl2 to oxidant between 1:0.2 and 1:5 to ensure complete conversion to the active SnCl4 species without excessive waste. Following the activation period, which can range from 1 to 24 hours depending on the temperature, the three components—aniline, aldehyde, and olefin—are introduced into the reaction vessel. The detailed standardized synthesis steps, including specific workup procedures and purification protocols to achieve optimal diastereoselectivity and yield, are provided in the guide below.
- Prepare the catalytic system by dissolving Lewis acid SnCl2 and a suitable oxidant (such as FeCl3, CuCl2, or AgNO3) in an organic solvent like dichloromethane or toluene.
- Stir the mixture at temperatures ranging from -20°C to 110°C for 1 to 24 hours to allow the in situ oxidation of SnCl2 to the active SnCl4 species.
- Add the aniline, aldehyde (aromatic or aliphatic), and olefin substrates to the reaction mixture, monitor progress via GC, and isolate the product after aqueous workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this SnCl2/oxidant promoted synthesis represents a strategic opportunity to optimize cost structures and enhance supply reliability. The shift away from rare earth catalysts and hazardous fluorine-based Lewis acids translates directly into reduced raw material expenditures and lower costs associated with specialized storage and handling infrastructure. By utilizing commodity chemicals like tin chloride and common oxidants, manufacturers can mitigate the risks associated with the price volatility of specialty catalysts, ensuring more stable long-term pricing for their clients. Furthermore, the ability to successfully utilize aliphatic aldehydes opens up new avenues for sourcing cheaper, readily available starting materials, thereby diversifying the supply base and reducing dependency on specific aromatic aldehyde suppliers who may face capacity constraints.
- Cost Reduction in Manufacturing: The elimination of expensive rare earth triflates such as scandium or dysprosium salts results in a drastic reduction in catalyst costs, which is a significant portion of the COGS for fine chemical synthesis. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, and the simplified workup procedures minimize solvent usage and waste disposal fees. The avoidance of highly corrosive reagents also extends the lifespan of reactor vessels and piping, lowering capital expenditure on equipment maintenance and replacement over time.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including SnCl2, FeCl3, and common organic solvents, are globally available commodities with robust supply chains, unlike niche catalysts that may suffer from geopolitical supply disruptions. The operational simplicity of the one-pot procedure reduces the risk of batch failures due to complex multi-step manipulations, ensuring consistent on-time delivery performance. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing, preventing costly delays in the broader pharmaceutical supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram to multi-kilogram scales without significant loss in yield or selectivity. The reduced toxicity and corrosivity of the reagent system simplify environmental, health, and safety (EHS) compliance, making it easier to obtain necessary permits for large-scale production. The generation of less hazardous waste streams compared to fluorine-based methods aligns with modern green chemistry principles, enhancing the sustainability profile of the manufacturing site and appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this route for potential adoption. Understanding these nuances is vital for assessing the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: Can this synthesis method utilize aliphatic aldehydes effectively?
A: Yes, unlike traditional Povarov reactions that struggle with aliphatic aldehydes due to imine instability, this SnCl2/oxidant system successfully facilitates the reaction with aliphatic aldehydes, significantly expanding substrate scope.
Q: What are the cost advantages of using the SnCl2/oxidant system over rare earth catalysts?
A: The system utilizes inexpensive and readily available tin salts and common oxidants instead of costly rare earth triflates like Sc(OTf)3 or Dy(OTf)3, leading to substantial reductions in raw material expenditure.
Q: Is the process scalable for industrial production of pharmaceutical intermediates?
A: Absolutely. The reaction operates under mild conditions with standard organic solvents and avoids highly corrosive reagents like BF3·OEt2, making it highly suitable for safe commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this SnCl2-mediated synthesis technology for the production of high-value nitrogen heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetrahydroquinoline derivative meets the exacting standards required by global pharmaceutical regulators. We are committed to leveraging our technical expertise to deliver consistent quality and supply continuity for your most critical projects.
We invite you to collaborate with us to explore how this cost-effective synthesis route can be tailored to your specific molecular targets. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel catalytic system for your specific application. Please contact us today to request specific COA data for our reference standards and to discuss comprehensive route feasibility assessments that will accelerate your development timelines and secure your supply chain.
