Scalable Copper-Catalyzed Synthesis of Indolo[3,2-c]quinoline Intermediates for Global Pharma
Scalable Copper-Catalyzed Synthesis of Indolo[3,2-c]quinoline Intermediates for Global Pharma
The pharmaceutical landscape is constantly evolving, driven by the urgent need for novel therapeutic agents capable of addressing complex diseases such as malaria and various forms of cancer. In this context, patent CN114621220B introduces a groundbreaking synthetic methodology for producing indolo[3,2-c]quinoline compounds containing benzamide groups, which have demonstrated exceptional potential as antimalarial and anticancer candidates. This technology represents a significant leap forward in medicinal chemistry, offering a robust pathway to access these biologically active scaffolds with high efficiency and selectivity. By leveraging a copper-catalyzed oxidative cyclization strategy, the invention circumvents the limitations of prior art, providing a sustainable and economically viable route for the production of high-purity pharmaceutical intermediates. For R&D directors and procurement specialists alike, understanding the nuances of this patented process is crucial for securing a reliable supply chain for next-generation therapeutics targeting protein kinase DYRK1A and DNA replication mechanisms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indolo[3,2-c]quinoline derivatives has been fraught with significant challenges that hinder their widespread adoption in drug discovery pipelines. Traditional literature methods predominantly rely on palladium-catalyzed insertion reactions of isocyanides or gold-catalyzed cyclization of acyclic alkynes, which necessitate the use of exorbitantly expensive precious metal catalysts that drastically inflate the cost of goods sold (COGS). Furthermore, these conventional pathways often involve harsh reaction conditions, narrow substrate scopes, and the utilization of toxic or difficult-to-handle reagents like isocyanides, posing severe safety and environmental risks in a manufacturing setting. The low yields and complex purification requirements associated with these legacy methods further exacerbate the problem, creating bottlenecks in the supply of critical intermediates and limiting the ability of pharmaceutical companies to scale up production for clinical trials or commercial launch without incurring prohibitive expenses.
The Novel Approach
In stark contrast, the innovative method disclosed in patent CN114621220B utilizes a cost-effective copper salt catalyst and molecular oxygen from air as the terminal oxidant, fundamentally reshaping the economic and operational feasibility of producing these valuable heterocycles. This approach employs readily available 2-(2-aminophenyl)indole derivatives as starting materials, which react smoothly in common organic solvents such as ethanol or dimethyl sulfoxide at moderate temperatures ranging from 70°C to 90°C. The transition to a base-metal catalytic system not only eliminates the dependency on scarce precious metals but also simplifies the downstream processing by reducing the burden of heavy metal removal, a critical quality attribute for API manufacturing. By enabling a broader application range of substrates and achieving high catalytic efficiency under environmentally benign conditions, this novel synthesis route offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous standards for purity and structural integrity.
![General reaction scheme for copper-catalyzed synthesis of indolo[3,2-c]quinoline compounds](/insights/img/indolo-quinoline-synthesis-copper-catalyst-pharma-supplier-20260303052906-02.webp)
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the intricate mechanism of copper-catalyzed oxidative C-H functionalization, which facilitates the construction of the quinoline ring system with remarkable precision. The reaction likely proceeds through a radical or organometallic pathway where the copper species activates the C-H bond adjacent to the nitrogen atom, promoting an intramolecular cyclization that forms the new C-N bond essential for the indoloquinoline scaffold. The use of air as the oxidant suggests a catalytic cycle where the copper center is regenerated by molecular oxygen, ensuring that the reaction remains atom-economical and generates water as the primary byproduct, thereby aligning with green chemistry principles. This mechanistic elegance allows for the tolerance of various functional groups, including halogens and methyl substituents, as evidenced by the successful synthesis of fluoro-, chloro-, and methyl-substituted derivatives without compromising yield or selectivity. For process chemists, understanding this mechanism is vital for optimizing reaction parameters and troubleshooting potential impurities that may arise from over-oxidation or incomplete cyclization.
Impurity control is another critical aspect where this method excels, primarily due to the mild reaction conditions and the high specificity of the copper catalyst. Unlike harsh acidic or basic conditions used in older methods that can lead to decomposition or polymerization of sensitive intermediates, the neutral to slightly acidic environment provided by the copper salt in alcohol solvents preserves the integrity of the benzamide moiety. The purification protocol, involving a simple wash with sodium carbonate followed by extraction and silica gel chromatography, effectively removes residual catalyst and unreacted starting materials, ensuring the final product meets stringent purity specifications required for biological testing. The ability to consistently produce high-quality material with minimal side products reduces the risk of batch failures and ensures a stable supply of intermediates for downstream drug development activities, ultimately accelerating the timeline from benchtop discovery to clinical evaluation.
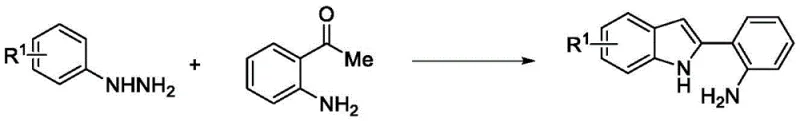
How to Synthesize Indolo[3,2-c]quinoline Efficiently
The synthesis of these bioactive compounds is streamlined into a logical two-stage process that begins with the preparation of the key indole precursor and culminates in the copper-catalyzed oxidative cyclization. The initial step involves a Fischer indole synthesis variant where o-aminoacetophenone reacts with phenylhydrazine derivatives under reflux in ethanol, followed by an acid-mediated cyclization using methanesulfonic acid and phosphorus pentoxide to yield the 2-(2-aminophenyl)indole scaffold. This precursor is then subjected to the oxidative coupling conditions in the presence of a copper catalyst and air, transforming it into the final indolo[3,2-c]quinoline derivative. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and purification techniques, are outlined in the comprehensive guide below to ensure reproducibility and scalability for industrial applications.
- Prepare the 2-(2-aminophenyl)indole precursor by reacting o-aminoacetophenone with phenylhydrazine derivatives in ethanol with acetic acid, followed by acid-mediated cyclization using methanesulfonic acid and phosphorus pentoxide.
- Combine the indole precursor with a copper salt catalyst (such as copper bromide or chloride) in an organic solvent like ethanol or DMSO within a sealed reaction vessel.
- Heat the mixture to 70-90°C under an air atmosphere for 10-14 hours to facilitate oxidative cyclization, then purify the resulting crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed synthesis route presents a transformative opportunity to optimize the cost structure and reliability of the pharmaceutical supply chain. By shifting away from precious metal catalysts like palladium and gold, which are subject to volatile market prices and geopolitical supply risks, manufacturers can achieve substantial cost savings and stabilize their raw material sourcing strategies. The use of air as an oxidant further eliminates the need for purchasing and storing hazardous stoichiometric oxidants, reducing both material costs and the regulatory burden associated with handling dangerous chemicals. Additionally, the simplified workup and purification procedures minimize solvent consumption and waste generation, leading to lower disposal costs and a reduced environmental footprint, which is increasingly important for meeting corporate sustainability goals and regulatory compliance standards in the global pharmaceutical industry.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or gold catalysts with abundant copper salts significantly lowers the direct material costs associated with the synthesis of these complex heterocycles. Furthermore, the elimination of specialized reagents like isocyanides and the use of commodity solvents such as ethanol contribute to a leaner manufacturing process that maximizes resource efficiency. This economic advantage allows pharmaceutical companies to allocate more budget towards R&D and clinical trials while maintaining competitive pricing for the final therapeutic products, ensuring long-term viability in a crowded market.
- Enhanced Supply Chain Reliability: The reliance on widely available and commercially mature reagents ensures a robust and resilient supply chain that is less susceptible to disruptions caused by shortages of exotic chemicals. The simplicity of the reaction setup, which does not require specialized high-pressure equipment or inert atmosphere gloveboxes for the oxidation step, facilitates easier technology transfer between manufacturing sites and contract development and manufacturing organizations (CDMOs). This flexibility enhances the overall agility of the supply network, enabling faster response times to market demands and reducing the lead time for high-purity pharmaceutical intermediates needed for urgent drug development programs.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of green oxidants make this process inherently safer and easier to scale from laboratory to commercial production volumes without encountering the thermal runaway risks often associated with traditional oxidation methods. The reduced generation of toxic waste streams simplifies effluent treatment and helps manufacturers comply with increasingly stringent environmental regulations regarding heavy metal discharge and solvent emissions. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate reputation of pharmaceutical companies committed to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of indolo[3,2-c]quinoline compounds, providing clarity for stakeholders evaluating this technology for their pipeline. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers in the pharmaceutical sector.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium routes?
A: The primary advantages include the substitution of expensive precious metal catalysts (Pd/Au) with abundant and low-cost copper salts, the use of molecular oxygen (air) as a green oxidant instead of hazardous stoichiometric oxidants, and milder reaction conditions that improve substrate tolerance and reduce energy consumption.
Q: What is the biological significance of the synthesized indolo[3,2-c]quinoline compounds?
A: These compounds exhibit potent biological activities, serving as high-efficiency inhibitors of protein kinase DYRK1A and potential DNA intercalators. They hold significant promise as candidate drugs for treating malaria and various forms of cancer due to their ability to inhibit DNA replication and transcription.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is highly suitable for scale-up due to the use of inexpensive reagents, simple operational procedures involving air oxidation, and straightforward purification protocols using standard silica gel chromatography, which collectively minimize production costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[3,2-c]quinoline Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications for complex heterocyclic intermediates, guaranteeing that every batch of indolo[3,2-c]quinoline derivatives delivered meets the highest industry standards for potency and safety. We understand the critical nature of your supply chain and are committed to providing consistent quality and reliable delivery schedules to support your drug development timelines.
We invite you to contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. By collaborating with us, you can gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about integrating this advanced synthetic methodology into your production strategy. Let us help you unlock the full potential of these promising antimalarial and anticancer candidates through our expertise in process optimization and commercial scale-up.
