Advanced Copper-Catalyzed Synthesis of Indolo[3,2-c]quinoline Derivatives for Commercial Pharmaceutical Applications
Advanced Copper-Catalyzed Synthesis of Indolo[3,2-c]quinoline Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic compounds that exhibit potent biological activities, particularly in the realms of antimalarial and anticancer therapeutics. Patent CN114621220B discloses a groundbreaking methodology for the synthesis of indolo[3,2-c]quinoline compounds containing benzamide moieties, which are recognized for their significant potential as protein kinase DYRK1A inhibitors and DNA intercalators. This technical insight report analyzes the novel copper-catalyzed oxidative cyclization strategy detailed in the patent, highlighting its superiority over conventional methods that rely on expensive transition metals and hazardous reagents. The disclosed process utilizes readily available 2-(2-aminophenyl)indole derivatives as starting materials, employing cheap copper salts as catalysts under an air atmosphere at moderate temperatures ranging from 70-90°C. ![General molecular structure of the indolo[3,2-c]quinoline compound with variable R1 substituents](/insights/img/indolo-quinoline-synthesis-copper-catalysis-pharma-supplier-20260303141047-01.png) This approach not only streamlines the synthetic pathway but also enhances the economic viability of producing these high-value pharmaceutical intermediates, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
This approach not only streamlines the synthetic pathway but also enhances the economic viability of producing these high-value pharmaceutical intermediates, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolo[3,2-c]quinoline scaffold has been fraught with significant synthetic challenges that hinder large-scale commercial adoption. Traditional literature methods predominantly rely on palladium-catalyzed insertion reactions involving isocyanides, gold-catalyzed cyclization of acyclic alkynes, or aza-Wittig reactions utilizing isocyanates. These legacy processes suffer from severe drawbacks, including the difficult availability and high toxicity of isocyanide starting materials, the prohibitive cost of palladium and gold catalysts, and the necessity for harsh reaction conditions that often lead to low yields and narrow substrate applicability. Furthermore, the requirement for inert atmospheres and specialized equipment to handle sensitive reagents increases the operational complexity and capital expenditure required for manufacturing. The presence of heavy metal residues from precious metal catalysts also necessitates rigorous and costly purification steps to meet stringent regulatory standards for active pharmaceutical ingredients, thereby creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the invention described in CN114621220B introduces a highly efficient and environmentally benign synthetic strategy that leverages earth-abundant copper catalysis. The core innovation lies in the direct oxidative cyclization of 2-(2-aminophenyl)indole derivatives using molecular oxygen from air as the terminal oxidant, effectively bypassing the need for toxic isocyanides or expensive noble metals. ![Reaction scheme showing copper-catalyzed oxidative cyclization of 2-(2-aminophenyl)indole to indolo[3,2-c]quinoline](/insights/img/indolo-quinoline-synthesis-copper-catalysis-pharma-supplier-20260303141047-02.webp) The process operates under mild thermal conditions between 70-90°C in common organic solvents such as ethanol or dimethyl sulfoxide, demonstrating exceptional functional group tolerance for substituents like fluorine, chlorine, and methyl groups. This novel approach drastically simplifies the reaction setup by eliminating the need for gloveboxes or Schlenk lines, as the reaction proceeds smoothly under an open air atmosphere. The use of inexpensive copper salts like copper bromide, copper chloride, or copper acetate at low molar loadings (1:0.1-0.3 ratio) ensures that the cost of goods sold is significantly reduced, while the broad substrate scope allows for the rapid generation of diverse analog libraries for drug discovery efforts without the need for process re-optimization.
The process operates under mild thermal conditions between 70-90°C in common organic solvents such as ethanol or dimethyl sulfoxide, demonstrating exceptional functional group tolerance for substituents like fluorine, chlorine, and methyl groups. This novel approach drastically simplifies the reaction setup by eliminating the need for gloveboxes or Schlenk lines, as the reaction proceeds smoothly under an open air atmosphere. The use of inexpensive copper salts like copper bromide, copper chloride, or copper acetate at low molar loadings (1:0.1-0.3 ratio) ensures that the cost of goods sold is significantly reduced, while the broad substrate scope allows for the rapid generation of diverse analog libraries for drug discovery efforts without the need for process re-optimization.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation involves a sophisticated copper-mediated C-H functionalization and subsequent intramolecular cyclization that constructs the quinoline ring system with high regioselectivity. The copper catalyst likely activates the amino group of the 2-(2-aminophenyl)indole precursor, facilitating a single-electron transfer process that generates a reactive radical intermediate capable of attacking the adjacent aromatic ring. Molecular oxygen from the air serves as the stoichiometric oxidant to regenerate the active copper species and drive the aromatization of the newly formed ring, resulting in the stable indolo[3,2-c]quinoline core. This mechanism avoids the formation of unstable intermediates often seen in isocyanide chemistry, leading to a cleaner reaction profile with fewer side products. The choice of solvent plays a critical role in stabilizing the transition states and solubilizing the polar intermediates, with polar protic solvents like ethanol proving particularly effective in promoting the reaction kinetics while maintaining catalyst stability throughout the 10 to 14 hour reaction window.
From an impurity control perspective, the high selectivity of this copper-catalyzed system is paramount for ensuring the quality of the final pharmaceutical intermediate. The reaction conditions are finely tuned to minimize over-oxidation or polymerization of the indole starting material, which are common degradation pathways in oxidative heterocycle synthesis. The use of mild bases like sodium carbonate during the workup phase effectively neutralizes acidic byproducts and removes residual copper species, preventing metal contamination in the final product. Furthermore, the purification protocol involving silica gel column chromatography with a petroleum ether and ethyl acetate gradient allows for the precise separation of the target benzamide-containing indoloquinoline from any unreacted starting materials or minor regioisomers. This robust control over the impurity profile is essential for meeting the rigorous specifications required by R&D directors for preclinical and clinical candidate materials, ensuring that the synthetic route is viable for GMP manufacturing.
How to Synthesize N-(2-(11H-indolo[3,2-c]quinolin-6-yl)phenyl)-2-aminobenzamide Efficiently
The synthesis of the target indolo[3,2-c]quinoline compound is achieved through a streamlined two-stage process that begins with the preparation of the key 2-(2-aminophenyl)indole intermediate followed by the copper-catalyzed cyclization step. The initial stage involves the condensation of o-aminoacetophenone with phenylhydrazine in the presence of acetic acid to form a hydrazone intermediate, which subsequently undergoes acid-catalyzed cyclization using methanesulfonic acid and phosphorus pentoxide to yield the indole precursor. 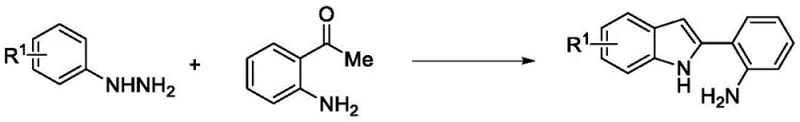 This precursor is then subjected to the oxidative cyclization conditions described previously, utilizing copper bromide in ethanol under air heating. The detailed standardized synthetic steps, including specific reagent quantities, temperature profiles, and purification parameters, are outlined in the guide below to ensure reproducibility and safety for laboratory and pilot plant operations.
This precursor is then subjected to the oxidative cyclization conditions described previously, utilizing copper bromide in ethanol under air heating. The detailed standardized synthetic steps, including specific reagent quantities, temperature profiles, and purification parameters, are outlined in the guide below to ensure reproducibility and safety for laboratory and pilot plant operations.
- Prepare 2-(2-aminophenyl)indole derivatives by reacting o-aminoacetophenone with phenylhydrazine derivatives in ethanol/acetic acid, followed by cyclization using methanesulfonic acid and phosphorus pentoxide.
- Conduct the oxidative cyclization by reacting the indole precursor with a copper salt catalyst (e.g., CuBr2, CuCl2) in an organic solvent like ethanol at 70-90°C under an air atmosphere.
- Purify the crude reaction mixture by washing with sodium carbonate solution, extracting with ethyl acetate, and performing silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed synthesis route offers transformative benefits that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. The shift from precious metal catalysts to base metal copper represents a fundamental change in the cost structure of the synthesis, removing the volatility associated with palladium and gold pricing while eliminating the need for complex metal scavenging resins. The reliance on air as the oxidant further reduces the consumption of hazardous chemical oxidants, lowering both material costs and the expenses related to hazardous waste disposal. Additionally, the use of commodity chemicals like ethanol and ethyl acetate as solvents ensures that the supply chain is not dependent on specialized or restricted reagents, thereby enhancing supply security and reducing lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive palladium and gold catalysts, combined with the use of inexpensive copper salts and ambient air, results in substantial cost savings in raw material expenditures. The simplified workup procedure, which avoids complex metal removal steps, further reduces processing time and consumable costs, leading to a more economically efficient manufacturing process that improves overall profit margins for API production.
- Enhanced Supply Chain Reliability: The starting materials, including o-aminoacetophenone and phenylhydrazine derivatives, are commercially available bulk chemicals with stable supply chains, mitigating the risk of production delays caused by raw material shortages. The robustness of the reaction conditions, which tolerate a wide range of substrates and do not require strict inert atmospheres, ensures consistent batch-to-batch quality and reliability, allowing supply chain planners to forecast production schedules with greater confidence and accuracy.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of standard reactor equipment and the absence of highly exothermic or hazardous steps, facilitating a smooth transition from laboratory scale to multi-ton commercial production. The reduced generation of toxic waste and the avoidance of heavy metal contaminants align with increasingly stringent environmental regulations, minimizing the environmental footprint of the manufacturing process and reducing the regulatory burden associated with waste management and emissions compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology, providing clarity on its practical application and benefits for stakeholders involved in pharmaceutical development and manufacturing. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their pipelines.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium-catalyzed routes?
A: The copper-catalyzed method utilizes inexpensive copper salts and ambient air as the oxidant, eliminating the need for costly palladium catalysts, toxic isocyanide reagents, and inert gas protection, thereby significantly reducing raw material costs and operational complexity.
Q: What is the substrate scope for R1 substituents in this synthesis?
A: The synthesis demonstrates broad substrate tolerance, successfully accommodating electron-donating groups like methyl, electron-withdrawing groups like fluoro and chloro, as well as unsubstituted hydrogen at the R1 position, ensuring versatility for diverse drug discovery programs.
Q: How does this process address environmental and safety concerns in large-scale manufacturing?
A: By avoiding toxic isocyanides and precious metal residues, and utilizing air instead of pure oxygen or hazardous oxidants, the process minimizes hazardous waste generation and simplifies downstream purification, aligning with green chemistry principles for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[3,2-c]quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the copper-catalyzed route for indolo[3,2-c]quinoline synthesis and confirmed its potential for delivering high-quality intermediates with exceptional purity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials regardless of their project phase. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, meeting the exacting demands of international regulatory bodies for antimalarial and anticancer drug candidates.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next drug development program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our indolo[3,2-c]quinoline derivatives and to discuss route feasibility assessments that can accelerate your path to clinical trials and commercial launch.
