Advanced Rhodium-Catalyzed Aminocarbonylation for Scalable Acetamide Manufacturing
The chemical industry is currently witnessing a paradigm shift towards sustainable synthesis methodologies, driven by the urgent need to reduce environmental footprints while maintaining high efficiency in the production of critical intermediates. Patent CN112812032B introduces a groundbreaking preparation method for acetamide compounds that addresses these challenges through a novel rhodium-catalyzed aminocarbonylation strategy. This technology leverages dimethyl carbonate (DMC) not merely as a benign reaction medium but actively as a C1 building block, effectively replacing hazardous traditional methylating agents. For R&D directors and procurement specialists seeking reliable acetamide supplier partnerships, this patent represents a significant advancement in green chemistry, offering a pathway to high-purity pharmaceutical intermediates with simplified downstream processing and reduced waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acetamide derivatives and related carbonyl-containing compounds has relied heavily on classical methylation and carbonylation protocols that pose severe safety and environmental risks. Conventional methylating reagents such as diazomethane, dimethyl sulfate, and methyl iodide are notorious for their high toxicity, explosiveness, and corrosive nature, necessitating specialized handling equipment and rigorous safety protocols that inflate operational costs. Furthermore, traditional carbonylation processes, exemplified by the Monsanto acetic acid process, often require hydroiodic acid (HI) as a co-catalyst, which induces severe corrosion in reactor vessels and piping, leading to frequent maintenance downtime and potential equipment failure. Additionally, these legacy methods typically demand stoichiometric amounts of strong bases to drive the reaction, resulting in the generation of substantial quantities of inorganic salt byproducts that require complex and costly disposal procedures, thereby negatively impacting the overall atom economy and sustainability profile of the manufacturing process.
The Novel Approach
In stark contrast to these hazardous legacy techniques, the methodology disclosed in CN112812032B utilizes a sophisticated rhodium catalytic system that operates under significantly milder and safer conditions. By employing dimethyl carbonate as a dual-function reagent, this novel approach eliminates the need for toxic methyl halides and corrosive acid promoters, fundamentally altering the safety landscape of acetamide production. The reaction proceeds efficiently at 120°C over a 24-hour period, utilizing readily available nitro compounds as nitrogen sources, which are generally cheaper and more stable than their amine counterparts. This innovation not only streamlines the synthetic route by combining methylation and carbonylation into a single pot but also ensures that the byproducts, primarily methanol and carbon dioxide, are environmentally benign and can potentially be recycled, aligning perfectly with modern principles of green chemistry and circular economy manufacturing.
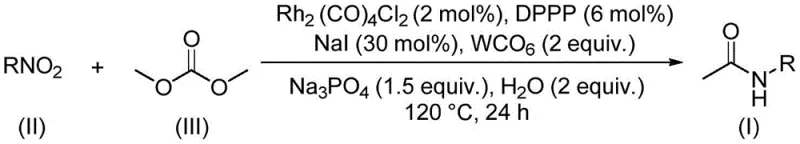
Mechanistic Insights into Rh-Catalyzed Aminocarbonylation
The core of this technological breakthrough lies in the intricate interplay between the dirhodium tetracarbonyl dichloride catalyst, the bidentate phosphine ligand 1,3-bis(diphenylphosphine)propane (DPPP), and the promoting effects of tungsten carbonyl and sodium iodide. Mechanistically, the rhodium center facilitates the reduction of the nitro group to an intermediate amine species in situ, which subsequently undergoes carbonylation with the dimethyl carbonate. The presence of sodium iodide is crucial as it likely generates reactive methyl iodide intermediates in catalytic amounts from the dimethyl carbonate, which then participate in the carbonylation cycle without the need for bulk hazardous reagents. The DPPP ligand stabilizes the rhodium species, preventing aggregation and ensuring high turnover numbers, while tungsten carbonyl may act as a secondary CO source or stabilizer, enhancing the overall robustness of the catalytic cycle against deactivation.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity acetamide compounds required in sensitive pharmaceutical applications. The mild reaction conditions (120°C) minimize thermal degradation of sensitive functional groups on the aromatic ring, such as halogens or trifluoromethyl groups, which might otherwise decompose under harsher acidic or basic conditions found in conventional methods. Furthermore, the absence of stoichiometric inorganic bases prevents the formation of difficult-to-remove salt impurities, simplifying the purification workflow. The high functional group tolerance observed across various substrates, including electron-rich and electron-deficient aromatics, suggests that the catalytic cycle is highly selective for the desired aminocarbonylation pathway, effectively suppressing side reactions such as over-methylation or homocoupling, thus delivering products with superior purity profiles suitable for direct use in drug synthesis.
How to Synthesize Acetamide Compounds Efficiently
Implementing this synthesis route requires precise control over reagent ratios and reaction parameters to maximize yield and reproducibility. The patent outlines a straightforward protocol where the catalyst system is combined with the nitro substrate and dimethyl carbonate in a sealed vessel, ensuring that the volatile components remain contained under the elevated temperature and pressure generated during the reaction. The standard procedure involves heating the mixture to 120°C for 24 hours, followed by a simple workup involving filtration and silica gel treatment. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below which encapsulates the critical steps for successful execution.
- Combine dirhodium tetracarbonyl dichloride, DPPP ligand, tungsten carbonyl, sodium phosphate, sodium iodide, water, nitro compound substrate, and dimethyl carbonate in a sealed vessel.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion of the nitro group to the acetamide functionality.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the high-purity acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits regarding cost stability and supply continuity. The shift away from regulated, hazardous reagents like methyl iodide reduces the regulatory burden and insurance costs associated with storing and transporting dangerous chemicals, while the use of commodity-grade dimethyl carbonate ensures a stable and predictable raw material supply chain unaffected by the volatility of specialty reagent markets. Moreover, the simplified post-treatment process, which avoids complex aqueous workups for salt removal, significantly reduces solvent consumption and waste disposal volumes, leading to substantial operational expenditure savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous methylating agents directly lowers the bill of materials, while the reduced need for corrosion-resistant reactor linings decreases capital expenditure requirements for plant infrastructure. By avoiding the generation of stoichiometric inorganic salts, the facility saves significantly on waste treatment fees and environmental compliance costs, creating a leaner and more profitable manufacturing model that enhances competitiveness in the global fine chemical market.
- Enhanced Supply Chain Reliability: Since the key starting materials, including nitro compounds and dimethyl carbonate, are widely produced commodities with multiple global suppliers, the risk of supply disruption is minimized compared to processes relying on single-source specialty catalysts or restricted reagents. This diversification of the supply base ensures consistent production schedules and reliable delivery timelines for downstream customers, mitigating the risks associated with geopolitical instability or logistical bottlenecks that often plague the chemical industry.
- Scalability and Environmental Compliance: The robust nature of the catalytic system allows for seamless scale-up from laboratory benchtop to multi-ton commercial production without significant re-optimization, facilitating rapid time-to-market for new drug candidates. Additionally, the green chemistry credentials of using a biodegradable solvent and generating benign byproducts simplify the permitting process for new manufacturing lines and align with the increasingly stringent environmental, social, and governance (ESG) mandates imposed by multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology for industrial partners seeking to optimize their acetamide production capabilities.
Q: What are the primary advantages of using dimethyl carbonate in this synthesis?
A: Dimethyl carbonate serves a dual role as both a green solvent and the C1 carbon source, eliminating the need for toxic methylating agents like methyl iodide and reducing inorganic salt waste.
Q: Does this catalytic system tolerate diverse functional groups?
A: Yes, the rhodium-catalyzed system demonstrates broad substrate scope, successfully accommodating substituents such as halogens, trifluoromethyl groups, alkoxy groups, and heterocycles without significant yield loss.
Q: Is the catalyst loading economically viable for large-scale production?
A: The process utilizes low catalyst loading (2 mol% Rh complex), and the starting materials including nitro compounds and dimethyl carbonate are commercially available and inexpensive, supporting cost-effective scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed aminocarbonylation technology for the next generation of pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of acetamide compound delivered meets the exacting standards required for API synthesis and advanced material applications.
We invite forward-thinking organizations to collaborate with us to leverage this innovative synthesis route for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of acetamide intermediates that will strengthen your product pipeline and enhance your market position.
