Revolutionizing Acetamide Production: A Green Rh-Catalyzed Route for Commercial Scale-Up
Revolutionizing Acetamide Production: A Green Rh-Catalyzed Route for Commercial Scale-Up
The landscape of organic synthesis is constantly evolving towards more sustainable and efficient methodologies, particularly in the production of high-value intermediates for the pharmaceutical and agrochemical sectors. A groundbreaking development detailed in patent CN112812032A introduces a novel preparation method for acetamide compounds that leverages the unique reactivity of dimethyl carbonate (DMC) in conjunction with a sophisticated rhodium catalytic system. This technology represents a paradigm shift from traditional methylation and carbonylation strategies, offering a pathway that is not only chemically elegant but also industrially robust. By utilizing nitro compounds as nitrogen sources and DMC as a dual-purpose reagent and solvent, this method addresses critical pain points regarding toxicity, waste generation, and operational complexity that have long plagued the synthesis of acetamide derivatives. For industry leaders seeking reliable acetamide intermediate suppliers, this patent provides the foundational IP for a next-generation manufacturing process that aligns perfectly with modern green chemistry principles while maintaining high reaction efficiency and broad substrate scope.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of acetamide frameworks and related carbonyl-containing compounds has relied heavily on reagents that pose significant safety and environmental hazards. Conventional methylating agents such as diazomethane, dimethyl sulfate, and methyl iodide are frequently employed, yet they suffer from severe drawbacks including explosiveness, high toxicity, corrosiveness, and exorbitant costs. Furthermore, these traditional pathways often necessitate the use of stoichiometric amounts of strong bases to drive the reaction to completion, which inevitably leads to the generation of substantial quantities of useless inorganic salts that require costly and energy-intensive disposal procedures. In the context of carbonylation reactions, processes like the Monsanto acetic acid synthesis utilize hydroiodic acid (HI) as a cocatalyst, which induces severe corrosion on reactor equipment, thereby increasing maintenance costs and risking supply chain interruptions due to equipment failure. These legacy methods create a bottleneck for procurement managers and supply chain heads who are increasingly pressured to reduce the environmental footprint and total cost of ownership for chemical manufacturing processes without compromising on purity or yield.
The Novel Approach
In stark contrast to these hazardous legacy methods, the invention disclosed in CN112812032A presents a transformative approach that utilizes dimethyl carbonate as both a C1 source and a green solvent, effectively eliminating the need for toxic methylating agents and stoichiometric bases. This rhodium-catalyzed aminocarbonylation reaction operates under relatively mild conditions, typically at 120°C for 24 hours, and demonstrates exceptional compatibility with a wide range of functional groups including halogens, alkoxy groups, and trifluoromethyl moieties. The use of DMC is particularly advantageous as it is a low-toxicity, biodegradable compound that can be derived from atmospheric carbon dioxide, thus closing the carbon loop and enhancing the sustainability profile of the synthesis. The reaction system employs a catalytic amount of dirhodium tetracarbonyl dichloride alongside tungsten carbonyl and sodium iodide promoters, ensuring high atom economy and minimizing heavy metal waste. This novel route not only simplifies the operational workflow by combining the solvent and reactant roles into a single component but also facilitates easier downstream processing, making it an ideal candidate for the commercial scale-up of complex organic intermediates.
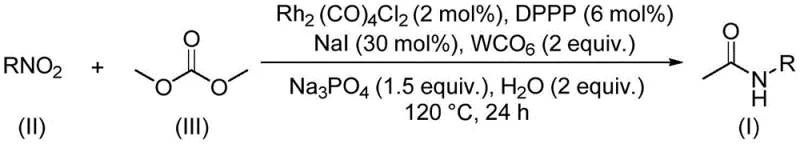
Mechanistic Insights into Rh-Catalyzed Aminocarbonylation
The mechanistic underpinning of this transformation involves a sophisticated interplay between the rhodium catalyst, the tungsten carbonyl promoter, and the nitro substrate within the dimethyl carbonate medium. The reaction initiates with the activation of the nitro compound, likely undergoing a reduction sequence facilitated by the metal carbonyl species and the iodide promoter to generate an amine intermediate in situ. Subsequently, the rhodium center coordinates with the carbonyl species derived from the decomposition or activation of dimethyl carbonate, forming an acyl-metal complex. This complex then undergoes nucleophilic attack by the in situ generated amine, leading to the formation of the acetamide bond and regeneration of the catalytic species. The presence of tungsten carbonyl (W(CO)6) is crucial as it serves as a solid source of carbon monoxide or facilitates the transfer of carbonyl groups, stabilizing the active rhodium species and preventing catalyst deactivation. The addition of water and sodium phosphate plays a pivotal role in buffering the reaction environment and assisting in the hydrolysis or activation steps required for the turnover of the catalytic cycle. This intricate balance of reagents ensures that the reaction proceeds with high selectivity, minimizing the formation of side products such as ureas or over-methylated species, which is a common challenge in amine carbonylation reactions.
From an impurity control perspective, the mild nature of this catalytic system offers significant advantages over harsh acidic or basic conditions found in conventional methods. The use of DMC as a solvent creates a homogeneous reaction environment that promotes efficient mass transfer and heat distribution, reducing the likelihood of localized hot spots that can lead to thermal degradation of sensitive functional groups. The specific ligand system, involving 1,3-bis(diphenylphosphino)propane (DPPP), provides the necessary steric and electronic environment around the rhodium center to favor the desired aminocarbonylation pathway over competing reactions. Furthermore, the absence of strong corrosive acids like HI means that the reactor walls remain intact, preventing metal leaching into the product stream which could otherwise complicate purification and fail stringent heavy metal specifications required for pharmaceutical intermediates. The result is a crude product profile that is significantly cleaner, reducing the burden on downstream purification units and allowing for higher overall recovery rates of the target high-purity acetamide compounds.
How to Synthesize Acetamide Compounds Efficiently
The practical implementation of this synthesis route is straightforward and designed for reproducibility in both laboratory and pilot plant settings. The procedure involves charging a sealed reaction vessel with the specific nitro compound substrate, dimethyl carbonate, and the catalytic system comprising Rh2(CO)4Cl2, DPPP, W(CO)6, NaI, Na3PO4, and water. The mixture is then heated to 120°C and stirred for approximately 24 hours to ensure complete conversion. Post-reaction, the workup is remarkably simple, involving filtration to remove insoluble salts and catalyst residues, followed by standard column chromatography or crystallization techniques to isolate the pure product. This streamlined protocol minimizes unit operations and reduces the time required for batch turnover, directly impacting manufacturing throughput. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Charge a reaction vessel with Rh2(CO)4Cl2 catalyst, DPPP ligand, W(CO)6 promoter, NaI, Na3PO4 base, water, the specific nitro compound substrate, and dimethyl carbonate solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion of the nitro group to the acetamide functionality.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the high-purity acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology offers compelling strategic advantages that extend beyond mere chemical efficiency. The primary driver for cost reduction lies in the substitution of expensive and hazardous reagents with dimethyl carbonate, which is not only cheaper but also acts as the reaction solvent, thereby reducing the volume of auxiliary chemicals required. The elimination of stoichiometric bases and the associated inorganic salt waste drastically lowers waste disposal costs and environmental compliance burdens, contributing to substantial cost savings in manufacturing overheads. Additionally, the use of readily available nitro compounds as starting materials ensures a stable and diversified supply base, mitigating the risks associated with sourcing specialized or controlled reagents like diazomethane. The robustness of the catalytic system allows for consistent batch-to-batch quality, reducing the incidence of failed batches and the associated financial losses. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the dual functionality of dimethyl carbonate, which removes the need for separate solvents and methylating agents, significantly simplifying the bill of materials. By avoiding the use of corrosive cocatalysts like hydroiodic acid, the lifespan of reactor equipment is extended, reducing capital expenditure on maintenance and replacement. The high atom economy of the reaction ensures that a larger proportion of raw material mass is converted into the final product, maximizing yield per kilogram of input and lowering the effective cost per unit of the high-purity acetamide compounds produced.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key raw materials, including nitro compounds and dimethyl carbonate, are commodity chemicals with established global supply chains. This reduces dependency on single-source suppliers for exotic reagents and minimizes the risk of production stoppages due to raw material shortages. The mild reaction conditions (120°C) are compatible with standard stainless steel reactors found in most multipurpose chemical plants, meaning that existing infrastructure can be utilized without the need for specialized lined vessels, further accelerating the time to market for new products.
- Scalability and Environmental Compliance: The green chemistry credentials of this method facilitate easier regulatory approval and environmental permitting, as the process generates minimal hazardous waste and utilizes a biodegradable solvent. The simplicity of the post-treatment workup, which avoids complex extraction or neutralization steps, allows for seamless scaling from kilogram to multi-ton production volumes. This scalability ensures that the supply chain can respond flexibly to fluctuating market demands, providing a secure and sustainable source of critical intermediates for the pharmaceutical and agrochemical industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and scope of the technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios and for procurement teams assessing the long-term viability of the supply chain.
Q: What are the primary advantages of using dimethyl carbonate in this synthesis?
A: Dimethyl carbonate serves a dual role as both a green solvent and a C1 source, eliminating the need for toxic methylating agents like methyl iodide and reducing inorganic salt waste generation significantly.
Q: What is the functional group tolerance of this rhodium-catalyzed method?
A: The method exhibits excellent tolerance for various substituents including alkoxy, trifluoromethyl, halogens (F, Cl, Br), methyl, thiomethyl, and acetyl groups on aryl rings, as well as alkyl chains.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of commercially available starting materials, moderate reaction temperatures (120°C), and simplified post-treatment procedures makes this route highly scalable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the rhodium-catalyzed aminocarbonylation technology described in CN112812032A and are fully equipped to leverage this innovation for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive catalytic reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the highest international standards. We understand that consistency and quality are paramount in the supply of pharmaceutical intermediates, and our integrated approach guarantees that every batch of acetamide compounds delivered meets the exacting requirements of your drug development programs.
We invite you to collaborate with us to unlock the full commercial potential of this green synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality targets, demonstrating how this technology can optimize your manufacturing economics. We encourage you to contact our technical procurement team today to request specific COA data for our acetamide portfolio and to discuss route feasibility assessments for your custom projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable acetamide intermediate supplier committed to driving innovation, sustainability, and cost efficiency in fine chemical manufacturing.
