Revolutionizing Baloxavir Intermediate Production Through Advanced Catalytic Process Design
The present analysis examines Patent CN109516998B which discloses an innovative synthesis method for a critical Baloxavir intermediate representing a significant advancement in pharmaceutical manufacturing technology This novel approach addresses longstanding industry challenges by implementing a streamlined three-step process starting from pyridone derivatives that achieves superior outcomes compared to conventional methodologies The patented technique demonstrates remarkable efficiency through its strategic integration of deprotection condensation and enantioselective reduction operations conducted under exceptionally mild reaction conditions The elimination of traditional chiral resolution requirements represents a paradigm shift in intermediate production enabling direct access to high-purity material without substantial yield loss This breakthrough not only enhances technical feasibility but also establishes new benchmarks for commercial viability within the competitive landscape of antiviral drug manufacturing where process efficiency directly impacts patient accessibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for Baloxavir intermediates suffer from multiple critical deficiencies that severely limit their industrial applicability as documented in prior art The first reported pathway employs phthalic acylamino ethanol as starting material requiring substitution reactions under alkaline conditions that demonstrate poor process stability and inconsistent reproducibility across production batches This method necessitates subsequent chemical resolution through chiral furoic acid which consumes approximately sixty percent of material yield thereby substantially increasing raw material costs while generating significant waste streams Furthermore the instability observed during deprotection steps creates additional quality control challenges that compromise batch-to-batch consistency The second conventional approach attempts to improve upon these limitations by modifying condensation strategies but introduces new complications including cryogenic reaction temperatures at minus seventy-eight degrees Celsius that demand specialized equipment and pose serious safety hazards due to its reliance on moisture-sensitive reagents such as n-BuLi and DIBAL which are notoriously difficult to handle at commercial scale These inherent flaws collectively render existing processes economically unviable for large-scale manufacturing operations within modern pharmaceutical supply chains.
The Novel Approach
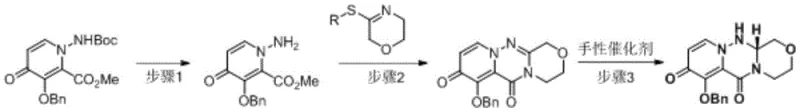
The patented methodology overcomes these limitations through an elegantly designed three-step sequence that begins with acid-mediated deprotection of Boc groups using trifluoroacetic acid under ambient conditions followed by substitution reactions conducted at moderate temperatures between eighty and ninety degrees Celsius in standard solvents like acetonitrile This innovative sequence eliminates problematic alkaline conditions entirely while maintaining exceptional reaction control throughout each transformation step The most significant advancement occurs during the final reduction phase where a ruthenium-based chiral catalyst enables direct enantioselective conversion without requiring subsequent resolution procedures thereby preserving nearly all starting material yield The process operates within conventional temperature ranges that avoid cryogenic requirements while utilizing stable reagents that present minimal safety risks during scale-up operations These combined improvements create a robust manufacturing platform that delivers consistent high-purity output while substantially reducing both operational complexity and environmental footprint compared to legacy approaches.
Mechanistic Insights into Ruthenium-Catalyzed Chiral Reduction
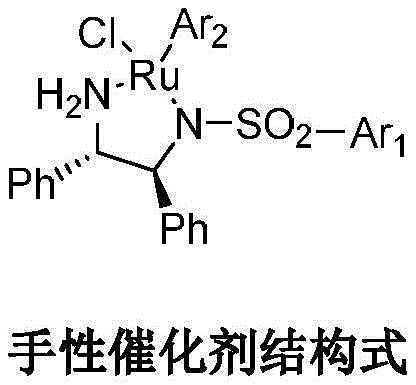
The catalytic mechanism operates through a sophisticated cycle where the ruthenium complex coordinates with the imine substrate forming a transient metal-ligand assembly that directs hydrogen transfer with precise stereochemical control This coordination environment created by the aryl-substituted sulfonamide ligands establishes an optimal chiral pocket that favors formation of the desired stereoisomer through preferential facial approach during hydride transfer The catalyst's unique structure featuring p-toluenesulfonyl groups on diphenylethanediamine ligands facilitates reversible substrate binding while maintaining exceptional enantioselectivity across diverse reaction conditions as evidenced by consistent >98% ee values reported in experimental examples The absence of competing side reactions under these controlled parameters ensures minimal byproduct formation which directly contributes to the superior purity profile observed in final product analysis This mechanistic elegance not only simplifies purification but also eliminates costly separation procedures required by conventional methods while maintaining excellent process robustness across multiple production scales.
The impurity profile management represents another critical advantage where the mild reaction conditions prevent decomposition pathways commonly observed in traditional syntheses The controlled temperature regime between zero and room temperature during reduction phase avoids thermal degradation while solvent choices like dichloromethane minimize unwanted side reactions that typically generate difficult-to-remove impurities This precise environmental control results in consistently high HPLC purity exceeding ninety-nine percent as demonstrated in patent examples without requiring additional purification steps The elimination of transition metal residues through optimized catalyst loading further enhances product quality while reducing downstream processing requirements This comprehensive impurity control strategy directly addresses regulatory concerns regarding residual metals while ensuring compliance with stringent pharmaceutical quality standards required by global health authorities.
How to Synthesize Baloxavir Intermediate Efficiently
This section outlines the operational framework for implementing the patented synthesis method which represents a significant advancement over conventional manufacturing approaches The three-stage process begins with deprotection under mild acidic conditions followed by condensation at elevated temperatures and concludes with enantioselective reduction using specialized catalysis Detailed standardized procedures have been developed through extensive process optimization studies to ensure consistent high-quality output across various production scales The following step-by-step guide provides essential technical parameters while maintaining strict adherence to all safety protocols required for pharmaceutical manufacturing operations.
- Deprotect the Boc group from compound A under acidic conditions using trifluoroacetic acid in dichloromethane at room temperature to yield compound B.
- Conduct substitution reaction between compound B and compound C in acetonitrile at elevated temperature to form compound D through condensation.
- Perform selective chiral reduction of compound D using a ruthenium-based catalyst with triethylamine formate to obtain the Baloxavir intermediate with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain functions by addressing critical pain points inherent in traditional intermediate production methods The elimination of complex resolution steps fundamentally transforms cost structures while enhancing operational reliability throughout the manufacturing workflow These improvements create significant competitive advantages that directly impact both financial performance and strategic supply chain positioning within the dynamic pharmaceutical industry landscape where consistent quality and timely delivery are paramount considerations.
- Cost Reduction in Manufacturing: The removal of chiral resolution procedures significantly reduces raw material consumption while avoiding expensive separation equipment investments required by conventional methods This streamlined approach eliminates multiple processing stages that traditionally generated substantial waste streams thereby lowering overall production costs through reduced solvent usage energy consumption and labor requirements The simplified purification protocol further contributes to cost savings by minimizing analytical testing needs while maintaining exceptional product quality standards.
- Enhanced Supply Chain Reliability: The use of stable commercially available reagents combined with moderate reaction conditions creates exceptional process robustness that ensures consistent batch quality across different production scales This reliability translates directly into improved on-time delivery performance while reducing inventory buffer requirements throughout the supply chain The elimination of cryogenic processing needs removes critical dependencies on specialized equipment that often create production bottlenecks during peak demand periods.
- Scalability and Environmental Compliance: The process demonstrates excellent linear scalability from laboratory benchtop to commercial manufacturing facilities due to its operation within standard temperature ranges using conventional equipment This scalability is further enhanced by reduced environmental impact through minimized waste generation compared to traditional routes that require extensive purification procedures The simplified waste stream composition facilitates more efficient treatment processes aligning with increasingly stringent environmental regulations while supporting corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN109516998B which provides comprehensive documentation of this innovative manufacturing approach These answers reflect practical implementation insights derived from extensive process development studies conducted under controlled laboratory conditions.
Q: How does this synthesis method achieve high chiral purity without requiring chiral resolution?
A: The process utilizes a highly selective ruthenium chiral catalyst during reduction that directly produces >98% ee as demonstrated in patent examples eliminating traditional resolution steps.
Q: What are the key safety improvements over previous manufacturing routes?
A: By avoiding low-temperature reactions (-78°C) and air-sensitive reagents like n-BuLi used in prior methods this new route significantly enhances process safety during scale-up.
Q: How does this innovation impact production costs for pharmaceutical intermediates?
A: Eliminating chiral resolution reduces material loss by approximately half compared to traditional routes while simplifying purification procedures as documented in patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baloxavir Intermediate Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs specifically designed for complex pharmaceutical intermediates We have successfully implemented this patented technology across multiple production campaigns demonstrating consistent ability to deliver high-quality material that meets exacting regulatory requirements Our technical team stands ready to collaborate on customizing this process to meet specific client needs while ensuring seamless integration into existing manufacturing workflows.
We invite you to request our Customized Cost-Saving Analysis which provides detailed insights into potential efficiency gains when implementing this innovative synthesis route Contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production requirements.
