Revolutionizing Axial Chiral Indole-Naphthalene Production for Commercial Scale-Up
The landscape of asymmetric catalysis is undergoing a significant transformation with the emergence of efficient organocatalytic strategies for constructing complex chiral scaffolds. A pivotal development in this domain is documented in patent CN110452150B, which discloses a robust preparation method for axial chiral indole-naphthalene compounds. These structures are not merely academic curiosities but serve as critical building blocks for next-generation organic small molecule catalysts and high-value pharmaceutical intermediates. The innovation lies in the ability to construct the axial chiral indole-naphthalene structure in a single step from racemic raw materials, bypassing the multi-step resolutions often required in traditional synthesis. This breakthrough offers a compelling value proposition for R&D directors seeking streamlined pathways to high-purity chiral entities. By leveraging a chiral phosphoric acid catalyst system, the process achieves exceptional stereocontrol under remarkably mild conditions, setting a new benchmark for efficiency in fine chemical manufacturing.
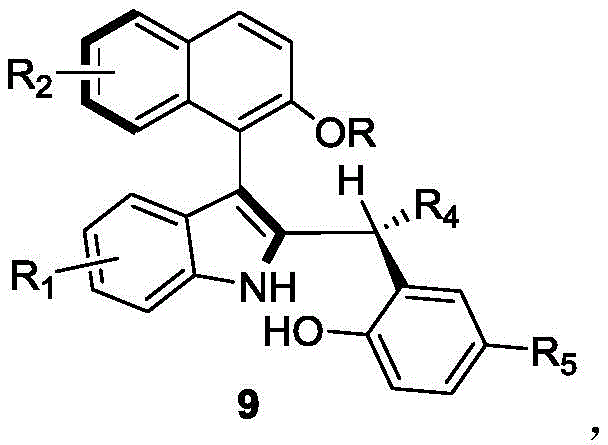
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral biaryl compounds, particularly those incorporating indole and naphthalene moieties, has been fraught with significant technical and economic challenges. Conventional methodologies predominantly rely on cross-coupling reactions between pre-functionalized indole and naphthalene rings, which often necessitate the use of expensive transition metal catalysts such as palladium or rhodium. These metal-catalyzed processes introduce severe downstream processing burdens, including the rigorous requirement for heavy metal removal to meet pharmaceutical regulatory standards. Furthermore, existing literature methods often suffer from limited substrate scope and harsh reaction conditions that compromise atom economy. The reliance on stoichiometric chiral auxiliaries or resolution of racemates further exacerbates cost inefficiencies, resulting in maximum theoretical yields of only 50% unless dynamic kinetic resolution is employed. For procurement managers, these factors translate into volatile supply chains and inflated raw material costs, creating a bottleneck for the commercial production of complex chiral intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN110452150B represents a paradigm shift towards sustainable and cost-effective organocatalysis. This novel approach utilizes a chiral phosphoric acid catalyst to facilitate an asymmetric addition reaction, effectively enabling the dynamic kinetic resolution of racemic starting materials. By employing a mixed solvent system of 1,1,2,2-tetrachloroethane and p-xylene alongside 5A molecular sieves, the reaction proceeds smoothly at temperatures ranging from 20°C to 30°C. This elimination of transition metals not only simplifies the purification workflow but also drastically reduces the environmental footprint associated with heavy metal waste disposal. The ability to generate diverse structural variants by modulating substituents on the indole and naphthalene rings provides unparalleled flexibility for medicinal chemists. For supply chain heads, this translates to a more resilient manufacturing process that is less dependent on scarce precious metals and more adaptable to large-scale production requirements.
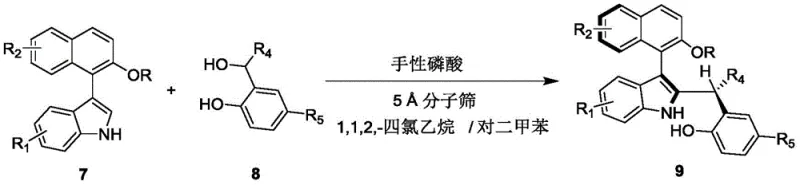
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
The core of this technological advancement lies in the precise mechanistic operation of the chiral phosphoric acid catalyst, specifically derivatives featuring a spiro skeleton structure. The catalyst functions through a dual hydrogen-bonding activation mode, where the acidic proton of the phosphoric acid moiety activates the electrophilic species while the phosphoryl oxygen coordinates with the nucleophile. This bifunctional activation creates a highly organized chiral environment within the transition state, effectively differentiating between the enantiotopic faces of the reacting molecules. The steric bulk provided by the anthracenyl groups on the catalyst backbone plays a crucial role in shielding one face of the substrate, thereby enforcing the formation of the desired axial chirality with high fidelity. This level of stereochemical control is essential for R&D directors who require consistent optical purity to ensure the biological efficacy of downstream drug candidates. The mechanism avoids the formation of unstable intermediates common in metal-catalyzed cycles, leading to a cleaner reaction profile and reduced formation of side products.
Impurity control is inherently built into this catalytic system due to the high enantioselectivity and diastereoselectivity observed during the bond-forming event. The use of 5A molecular sieves serves a dual purpose: it acts as a water scavenger to prevent catalyst deactivation and shifts the equilibrium towards product formation by removing water generated during the condensation process. This ensures that the reaction proceeds to completion without the accumulation of hydrolytic byproducts that could complicate downstream purification. The resulting products exhibit enantiomer ratios (er) as high as 98:2, demonstrating the robustness of the chiral induction. For quality assurance teams, this inherent purity reduces the burden on analytical testing and minimizes the risk of batch rejection. The structural diversity achievable through this mechanism allows for the rapid generation of analog libraries, accelerating the lead optimization phase in drug discovery programs while maintaining strict control over the impurity profile.
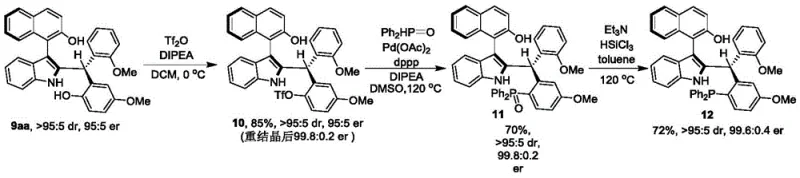
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
Implementing this synthesis route requires careful attention to solvent composition and catalyst loading to maximize yield and optical purity. The process begins with the preparation of the reaction mixture, where the molar ratio of the indole-naphthalene precursor to the phenol derivative is optimized between 1:1 and 1:3. The choice of solvent is critical, with a volume ratio of 1,1,2,2-tetrachloroethane to p-xylene preferably maintained at 1:4 to ensure optimal solubility and reaction kinetics. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are provided in the structured guide below to ensure reproducibility across different manufacturing sites.
- Prepare the reaction mixture by combining compound of formula 7 and compound of formula 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene.
- Add 5A molecular sieves and a chiral phosphoric acid catalyst (specifically a spiro skeleton derivative) to the mixture under stirring.
- Maintain the reaction temperature between 20°C to 30°C until completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this organocatalytic platform offers substantial advantages that directly address the pain points of modern chemical procurement and supply chain management. The elimination of transition metal catalysts removes a significant cost driver associated with both the purchase of precious metals and the specialized resins required for their removal. This simplification of the bill of materials leads to a more predictable cost structure, allowing procurement managers to negotiate better terms with raw material suppliers. Furthermore, the mild reaction conditions reduce energy consumption and lower the safety risks associated with high-pressure or high-temperature operations, contributing to overall operational expenditure savings. The robustness of the reaction across various substrates ensures that supply chain disruptions for specific starting materials can be mitigated by switching to alternative analogs without revalidating the entire process.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with organocatalysts fundamentally alters the cost dynamics of producing high-purity pharmaceutical intermediates. By removing the need for palladium or rhodium complexes, manufacturers avoid the volatility of precious metal markets and the capital expenditure associated with metal scavenging technologies. Additionally, the high atom economy of this addition reaction minimizes waste generation, reducing the costs associated with waste treatment and disposal. The simplified workup procedure, involving basic filtration and concentration, reduces labor hours and solvent usage compared to complex extraction protocols. These cumulative efficiencies result in significant cost savings that can be passed down the supply chain, enhancing the competitiveness of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on economically available and stable raw materials strengthens the resilience of the supply chain against geopolitical or market fluctuations. Unlike specialized metal ligands that may have limited suppliers, the chiral phosphoric acid catalysts and organic substrates used in this process are accessible from multiple global sources. The mild operating conditions also mean that the reaction can be performed in a wider range of manufacturing facilities, including those without specialized high-pressure infrastructure. This flexibility allows for diversified manufacturing strategies, reducing the risk of single-point failures in the supply network. For supply chain heads, this translates to improved on-time delivery performance and the ability to scale production rapidly in response to market demand without lengthy equipment upgrades.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exothermic hazards typically associated with metal-catalyzed couplings. The use of molecular sieves as a heterogeneous additive simplifies the separation process, making it amenable to continuous flow chemistry or large batch processing. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The solvent system, while requiring careful handling, is recyclable, further supporting sustainability goals. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the brand reputation of companies adopting this technology as responsible manufacturers committed to environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for adoption. Understanding these nuances is critical for making informed decisions about process integration and long-term sourcing strategies.
Q: What is the primary advantage of this synthesis method over traditional coupling reactions?
A: This method utilizes dynamic kinetic resolution of racemic compounds via organocatalysis, allowing for one-step construction of the axial chiral skeleton with high enantioselectivity, avoiding the need for complex resolution steps or expensive transition metals.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the reaction conditions are mild (20-30°C) and use economically available raw materials and solvents, making it highly suitable for commercial scale-up without requiring extreme pressure or temperature equipment.
Q: What is the optical purity achievable with this protocol?
A: The patent data indicates that the method yields products with high optical purity, with enantiomer ratios (er) reaching up to 98:2, which is critical for pharmaceutical applications requiring strict stereochemical control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in advancing the field of asymmetric synthesis. As a premier CDMO partner, we possess the technical expertise to translate complex laboratory protocols into robust commercial manufacturing processes. Our facilities are equipped to handle the specific solvent systems and purification requirements necessary for producing these high-value chiral intermediates. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain capable of delivering high-purity axial chiral indole-naphthalene compounds that meet the demanding requirements of modern drug development.
