Advanced Palladium-Catalyzed Synthesis of 3-Arylquinoline Derivatives for Commercial-Scale Pharmaceutical Manufacturing
The recently granted Chinese patent CN113045489B introduces a transformative methodology for synthesizing 3-arylquinoline-2(1H) ketone derivatives, representing a significant advancement in heterocyclic compound manufacturing for the pharmaceutical sector. This innovative process leverages benzisoxazole as a dual-function reagent serving simultaneously as nitrogen source and formyl donor, eliminating the need for multiple synthetic steps traditionally required in quinolinone production. The methodology demonstrates exceptional substrate versatility across diverse functional groups while maintaining high reaction efficiency under mild conditions, addressing critical limitations in existing synthetic routes. By integrating these key improvements, the patent establishes a robust foundation for producing high-value pharmaceutical intermediates with enhanced purity profiles and operational simplicity, directly supporting the development of next-generation therapeutics requiring complex heterocyclic scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for quinolinone derivatives, including Vilsmeier Haack, Knorr, and Friedlander reactions, suffer from significant operational constraints that hinder their commercial viability for pharmaceutical manufacturing. These methods typically require harsh reaction conditions, multiple protection/deprotection steps, and generate complex impurity profiles that necessitate extensive purification processes, substantially increasing production costs and reducing overall yield. The reliance on stoichiometric reagents rather than catalytic systems creates substantial waste streams that conflict with modern green chemistry principles, while narrow functional group tolerance limits the structural diversity achievable through these routes. Furthermore, transition metal-catalyzed variants often demand expensive catalysts and intricate ligand systems that complicate scale-up procedures and introduce metal contamination risks requiring additional removal steps. These cumulative challenges result in extended production timelines and inconsistent quality that cannot meet the stringent requirements of modern pharmaceutical supply chains.
The Novel Approach
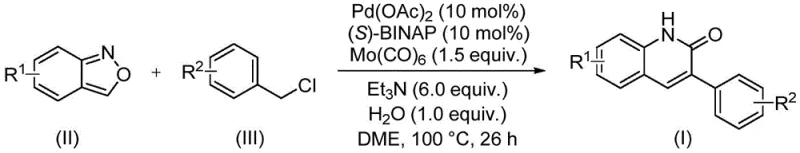
The patented methodology overcomes these limitations through an elegant palladium-catalyzed aminocarbonylation process that utilizes benzisoxazole as a dual-source reagent, fundamentally simplifying the synthetic pathway to 3-arylquinoline derivatives. By operating at a moderate temperature of 100°C with readily available catalysts including palladium acetate and (S)-BINAP, the process achieves high efficiency across a broad substrate scope without requiring specialized equipment or hazardous reagents. The integration of both nitrogen and formyl components within a single starting material eliminates intermediate isolation steps that typically introduce impurities in conventional methods, while the well-defined catalytic cycle ensures consistent product quality. This streamlined approach maintains compatibility with standard industrial processing equipment and demonstrates exceptional scalability from laboratory to commercial production volumes, directly addressing the critical pain points that have historically constrained quinolinone derivative manufacturing for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of benzyl chloride to the palladium(0) species generated in situ from palladium acetate and (S)-BINAP, forming an arylpalladium intermediate that subsequently coordinates with benzisoxazole. This key interaction triggers ring-opening of the isoxazole moiety, releasing the nitrogen component while simultaneously providing the formyl group through a unique decarbonylation pathway facilitated by Mo(CO)₆ as a carbonyl source. The resulting palladacycle undergoes intramolecular cyclization followed by reductive elimination to yield the target quinolinone structure while regenerating the active catalyst species. This sophisticated mechanism operates with remarkable efficiency due to the synergistic effects between the palladium catalyst system and the dual-function benzisoxazole reagent, which eliminates competing side reactions through its precisely controlled decomposition pathway under the specified reaction conditions of 100°C in DME solvent.
The process demonstrates exceptional impurity control through its integrated reaction design, where the dual-source nature of benzisoxazole prevents the formation of common byproducts associated with traditional quinolinone syntheses. By avoiding separate nitrogen and carbonyl sources, the methodology eliminates potential side reactions between incompatible reagents that typically generate difficult-to-remove impurities requiring additional purification steps. The well-defined catalytic cycle operates under mild conditions that minimize thermal degradation pathways, while the specific ligand system ((S)-BINAP) provides steric control that prevents undesired regioisomer formation. This inherent selectivity results in consistently high-purity products that meet pharmaceutical requirements without extensive post-reaction processing, directly addressing critical quality concerns for R&D directors focused on developing robust manufacturing processes for complex heterocyclic compounds.
How to Synthesize 3-Arylquinoline Derivatives Efficiently
This patented methodology represents a significant advancement in the synthesis of high-value quinolinone derivatives for pharmaceutical applications, offering a streamlined approach that addresses longstanding challenges in heterocyclic compound manufacturing. The process leverages innovative dual-source chemistry to simplify what was previously a multi-step sequence into a single efficient transformation with superior operational characteristics. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in the patent literature, ensuring consistent results across diverse substrate combinations while maintaining strict quality control parameters essential for pharmaceutical intermediate production. The following section outlines the precise implementation protocol for achieving optimal results when scaling this technology from laboratory to commercial manufacturing environments.
- Prepare reaction mixture with palladium acetate (0.1 mol%), (S)-BINAP (0.1 mol%), Mo(CO)₆ (1.5 equiv.), triethylamine (6.0 equiv.), and water (1.0 equiv.) in DME solvent
- Add benzisoxazole (II) and benzyl chloride compound (III) at molar ratio of 1: 2.5 with R¹/R² substituents selected for optimal yield
- Maintain reaction at 100°C for precisely 26 hours under inert atmosphere before standard workup and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial strategic benefits for procurement and supply chain management teams seeking reliable sources for complex pharmaceutical intermediates. By fundamentally rethinking the synthetic pathway through dual-source chemistry, the process eliminates multiple cost drivers while enhancing supply chain resilience through simplified material requirements and reduced processing complexity. The methodology directly addresses critical pain points in traditional quinolinone production that have historically created bottlenecks in pharmaceutical supply chains, offering procurement teams a more sustainable solution that aligns with modern manufacturing excellence principles while supporting continuous improvement initiatives across the value chain.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and reduction in purification requirements through integrated reaction design significantly lowers overall production costs without compromising quality standards. By utilizing benzisoxazole as a dual-function reagent, the process avoids costly intermediate isolation procedures while maintaining high efficiency across diverse substrates. The use of commercially available catalysts at low loadings combined with simplified workup procedures reduces both material expenses and processing time, creating substantial cost savings through operational streamlining rather than marginal efficiency improvements.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials with broad commercial sourcing options substantially improves raw material security while reducing vulnerability to single-source dependencies that plague traditional synthetic routes. The process demonstrates exceptional robustness across different supplier lots due to its tolerance for diverse functional groups and consistent performance under standard industrial conditions. This inherent flexibility enables procurement teams to maintain continuous production even during market fluctuations, while the simplified logistics profile reduces lead time variability for high-purity pharmaceutical intermediates through fewer processing steps and reduced quality control touchpoints.
- Scalability and Environmental Compliance: The methodology's compatibility with standard industrial equipment and straightforward scale-up profile enables seamless transition from laboratory to commercial production volumes while maintaining consistent quality parameters essential for pharmaceutical applications. The elimination of hazardous reagents and reduction in waste streams through integrated reaction design significantly improves environmental performance metrics without requiring specialized infrastructure investments. This green chemistry approach not only reduces regulatory compliance burdens but also supports corporate sustainability initiatives through inherently cleaner manufacturing processes that align with global environmental standards while maintaining commercial viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns raised by procurement teams evaluating this innovative manufacturing approach for pharmaceutical intermediate production. These responses are based on detailed analysis of the patented methodology's technical specifications and performance characteristics as documented in CN113045489B, providing factual information to support informed decision-making regarding implementation timelines, quality assurance protocols, and supply chain integration requirements for this advanced synthetic route.
Q: How does the dual-source benzisoxazole approach improve impurity profile compared to conventional methods?
A: The benzisoxazole serves as both nitrogen and formyl source in a single catalytic cycle, eliminating intermediate isolation steps that typically introduce impurities in traditional Vilsmeier Haack or Friedlander reactions. This integrated mechanism reduces byproduct formation and simplifies purification, consistently achieving high-purity pharmaceutical intermediates without additional metal scavenging.
Q: What supply chain advantages does this method offer for large-scale manufacturing?
A: The process utilizes commercially available, low-cost starting materials with broad functional group tolerance, significantly enhancing raw material security. The simplified workup procedure and elimination of transition metal removal steps reduce production bottlenecks, enabling reliable commercial scale-up of complex pharmaceutical intermediates while maintaining stringent quality specifications.
Q: How does the palladium-catalyzed system ensure scalability from lab to plant?
A: The well-defined catalytic system operates under consistent conditions (100°C, DME solvent) with predictable kinetics across diverse substrates. The absence of sensitive reagents and compatibility with standard industrial equipment allows seamless transition from 100 kgs to multi-ton production volumes while maintaining yield consistency and purity requirements for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline Derivatives Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds, ensuring seamless technology transfer from laboratory to full-scale manufacturing environments. Our rigorous QC labs implement stringent purity specifications through advanced analytical methodologies that guarantee consistent product quality meeting global pharmaceutical standards across all production volumes. As a trusted partner in specialty chemical manufacturing, we combine deep technical expertise in palladium-catalyzed transformations with comprehensive regulatory support to deliver reliable supply solutions for high-value intermediates requiring sophisticated synthetic approaches like this patented quinolinone methodology.
Leverage our technical procurement team's expertise to request specific COA data and route feasibility assessments tailored to your production requirements through our Customized Cost-Saving Analysis service. This dedicated support enables you to evaluate implementation timelines and quality assurance protocols while optimizing your supply chain strategy for complex pharmaceutical intermediates manufacturing.
