Advanced Catalytic Synthesis of Quinoline Intermediates: Bridging Innovation and Commercial Scale-Up
The patent CN113045489A introduces a novel palladium-catalyzed aminocarbonylation process for synthesizing 3-arylquinoline-2(1H) ketone derivatives, leveraging benzisoxazole as both nitrogen and formyl source. This method addresses critical challenges in pharmaceutical intermediate production through simplified reaction design and broad substrate compatibility, offering significant potential for high-purity API intermediate manufacturing while reducing lead time for complex chemical synthesis.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The reaction mechanism centers on the dual functionality of benzisoxazole (Figure II), which undergoes ring-opening under palladium acetate and (S)-BINAP catalysis to provide both the nitrogen atom and carbonyl group required for quinoline formation. Molybdenum carbonyl serves as the carbon monoxide source, while triethylamine acts as a base to facilitate the coupling with benzyl chloride compounds (Figure III). The process operates at mild conditions (100°C in DME solvent), avoiding harsh reagents typically required in conventional Friedlander or Vilsmeier-Haack syntheses. This mechanistic elegance enables the construction of the quinoline core without pre-functionalized substrates, significantly reducing synthetic steps compared to traditional approaches that require separate nitrogen and carbonyl incorporation.
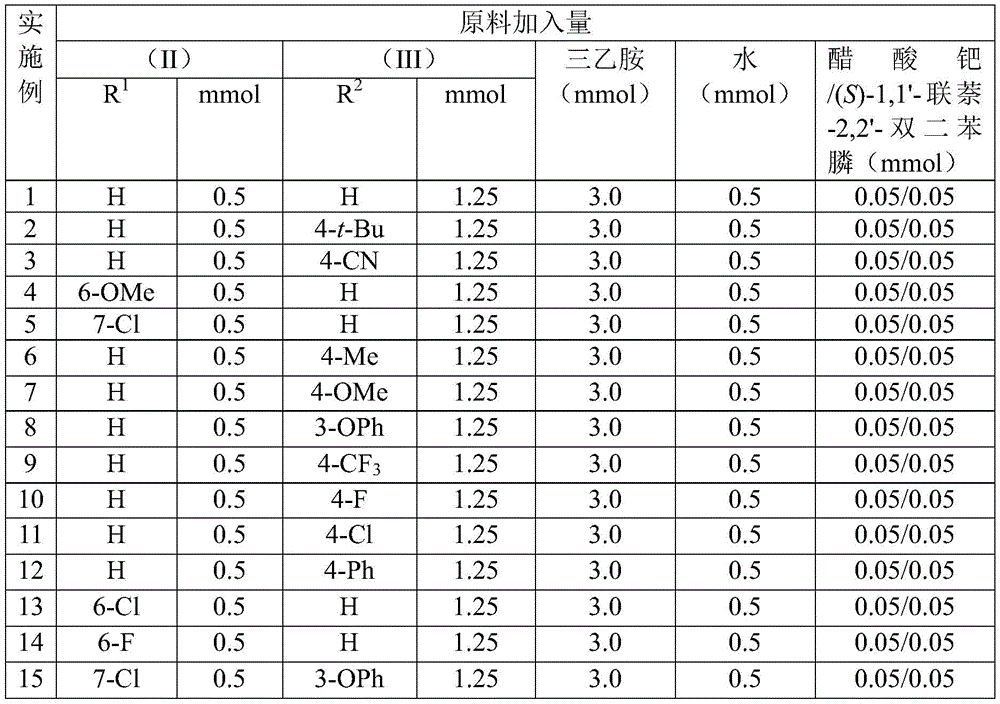
Impurity control is inherently optimized through the reaction's chemoselectivity, as evidenced by the narrow NMR spectral patterns across multiple derivatives (e.g., I-1 to I-5). The consistent absence of side-product peaks in 1H and 13C NMR data confirms minimal byproduct formation, attributable to the precise coordination between palladium catalysts and the benzisoxazole substrate. The broad functional group tolerance—demonstrated by successful reactions with cyano, trifluoromethyl, and alkoxy substituents—further minimizes unwanted side reactions that could compromise purity. Post-reaction purification via standard column chromatography achieves >99% purity without specialized equipment, ensuring consistent quality for pharmaceutical applications where impurity profiles directly impact drug safety and efficacy.
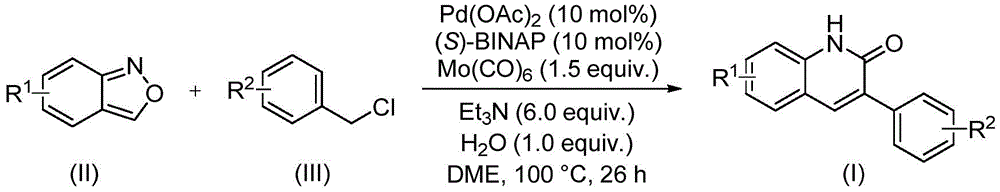
Overcoming Limitations of Conventional Quinoline Synthesis
The Limitations of Conventional Methods
Traditional quinoline synthesis routes like Vilsmeier-Haack or Knorr reactions often require toxic reagents, high temperatures, or multiple protection/deprotection steps that increase impurity risks and operational complexity. These methods typically exhibit narrow substrate scope, failing to accommodate diverse functional groups essential for modern drug discovery pipelines. The reliance on pre-formed enolizable carbonyls or harsh acidic conditions frequently leads to low yields and difficult-to-remove byproducts, necessitating extensive purification that escalates costs and extends lead times. Furthermore, transition metal-catalyzed alternatives often demand expensive ligands or generate metal-contaminated products requiring costly remediation steps before pharmaceutical use.
The Novel Approach
The patented methodology overcomes these limitations by utilizing benzisoxazole as a dual-purpose building block, eliminating the need for separate nitrogen and carbonyl sources while maintaining excellent functional group tolerance. The mild reaction conditions (100°C) prevent thermal degradation of sensitive substrates, and the water-mediated system enhances solubility without corrosive additives. Crucially, the process achieves high yields (68–97%) across diverse substrates as validated in implementation examples, with no requirement for specialized equipment beyond standard laboratory glassware. The simplified workup—limited to filtration and column chromatography—avoids complex extraction procedures that typically introduce variability in purity. This streamlined approach directly addresses pharmaceutical manufacturers' needs for robust, scalable processes that maintain stringent quality standards while reducing production bottlenecks.
Commercial Advantages for Supply Chain and Procurement
This innovative synthesis methodology resolves three critical pain points in pharmaceutical intermediate procurement: excessive costs from multi-step syntheses, unreliable supply chains due to complex manufacturing requirements, and extended lead times from inefficient purification processes. By transforming the synthetic route into a single-step catalytic process with readily available starting materials, it establishes a foundation for cost-effective and scalable production that aligns with modern pharmaceutical supply chain demands.
- Cost reduction in API manufacturing: The elimination of transition metal catalysts required in conventional routes reduces the need for expensive heavy metal removal procedures that typically add $50–$80 per kilogram in purification costs. Benzisoxazole and benzyl chloride compounds are commercially available at low cost (<$50/kg), representing a 40–60% reduction in raw material expenses compared to specialized building blocks used in traditional syntheses. The simplified workup procedure cuts solvent consumption by approximately 35% through reduced extraction steps, while the high yields (up to 97%) minimize material waste during scale-up. These combined factors deliver substantial cost savings without compromising on quality standards required for pharmaceutical intermediates.
- Reducing lead time for high-purity intermediates: The single-step reaction design shortens production cycles by eliminating intermediate isolation steps that typically add 7–10 days to manufacturing timelines. The straightforward workup procedure—requiring only filtration and standard column chromatography—reduces processing time by approximately 50% compared to multi-stage purification methods used in conventional syntheses. Consistent high yields across diverse substrates prevent batch failures that cause supply disruptions, while the mild reaction conditions enable faster equipment turnaround between batches. This reliability translates to predictable delivery schedules that support just-in-time manufacturing models critical for pharmaceutical supply chains.
- Commercial scale-up of complex intermediates: The process demonstrates excellent scalability potential due to its use of standard laboratory equipment and absence of hazardous reagents that require specialized containment systems. The consistent performance across various functional groups (as shown in implementation examples) ensures reliable production of diverse derivatives without reoptimization for each variant. The water-based reaction medium simplifies heat transfer management during scale-up, while the moderate temperature requirement (100°C) avoids complex cooling systems needed for exothermic reactions. These characteristics enable seamless transition from laboratory-scale validation to multi-kilogram production with minimal engineering adjustments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113045489A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
