Advanced Cobalt-Catalyzed Synthesis of 2-Alkoxy Indole Intermediates for Commercial Scale-Up
Advanced Cobalt-Catalyzed Synthesis of 2-Alkoxy Indole Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and cost-effective pathways to access complex heterocyclic scaffolds, particularly those found in bioactive molecules like the selective 5-HT4 receptor antagonists GR-125487 and SB-207266. A significant technological breakthrough in this domain is detailed in patent CN115772157A, which discloses a novel preparation method for 2-alkoxy indole compounds. This innovation represents a paradigm shift from traditional multi-step syntheses involving precious metals to a streamlined, earth-abundant cobalt-catalyzed C-H activation strategy. By leveraging transition metal catalysis, this method enables the direct functionalization of the indole core at the 2-position using simple alcohols as both solvents and reactants. For R&D directors and process chemists, this development offers a compelling alternative for constructing critical pharmaceutical intermediates with high atom economy and operational simplicity.
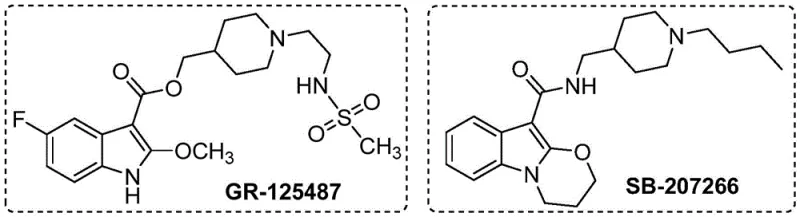
The strategic importance of 2-alkoxy indoles cannot be overstated, as they serve as pivotal building blocks for a wide array of therapeutic agents targeting central nervous system disorders and gastrointestinal conditions. The disclosed technology addresses the longstanding challenge of regioselective functionalization at the C2 position of the indole ring, a transformation that historically required harsh conditions or extensive protecting group manipulations. By establishing a reliable protocol that operates under moderate thermal conditions (90-110°C) and utilizes commercially accessible reagents, this patent lays the groundwork for a more sustainable supply chain. As a reliable pharmaceutical intermediate supplier, understanding such methodological advancements is crucial for maintaining competitiveness and offering clients cutting-edge synthetic solutions that balance performance with economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-alkoxy indole derivatives has been plagued by significant inefficiencies that hinder large-scale production. Traditional routes often rely on noble metal catalysts such as palladium or rhodium, which not only drive up the raw material costs but also introduce stringent requirements for metal residue removal in the final active pharmaceutical ingredient (API). Furthermore, conventional methodologies frequently necessitate pre-functionalized starting materials, such as halogenated indoles, adding extra synthetic steps and generating substantial chemical waste. These multi-step sequences result in lower overall yields and increased processing time, creating bottlenecks in the supply chain. Additionally, many older methods suffer from poor substrate tolerance, failing to accommodate diverse functional groups without decomposition or side reactions, thereby limiting the chemical space available for medicinal chemistry optimization.
The Novel Approach
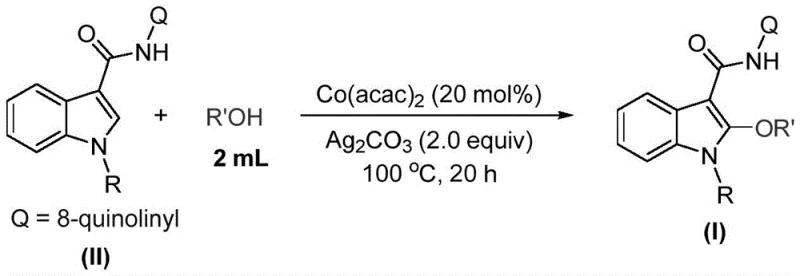
In stark contrast, the method described in patent CN115772157A introduces a direct C-H alkoxylation strategy that bypasses the need for pre-halogenation. By employing a cobalt catalyst system, specifically cobalt acetylacetonate (Co(acac)2), in conjunction with silver carbonate as a mild oxidant, the process achieves high efficiency with remarkable simplicity. The reaction proceeds smoothly in the alcohol solvent itself, eliminating the need for additional organic solvents and reducing the environmental footprint. This approach demonstrates exceptional substrate compatibility, successfully tolerating various substituents on the indole nitrogen (R groups ranging from alkyl to aryl) and accommodating different alcohol chain lengths (R' groups). The operational ease, characterized by a simple mix-and-heat procedure followed by standard filtration and purification, drastically reduces the technical barrier for implementation, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cobalt-Catalyzed C-H Alkoxylation
The mechanistic pathway underpinning this transformation involves a sophisticated interplay between the cobalt center and the indole substrate, driven by single-electron transfer (SET) processes. Initially, the cobalt(II) catalyst is oxidized by silver carbonate to generate a reactive cobalt(III) species, which subsequently coordinates with the indole compound. This coordination facilitates a single-electron transfer event, forming a radical cobalt(II) complex that is pivotal for the activation of the inert C-H bond at the 2-position of the indole ring. The radical intermediate is then re-oxidized by silver carbonate, stabilizing the system as a cobalt(III) complex with the C-H bond activated. This precise control over the oxidation state of the metal center ensures high regioselectivity, preventing unwanted functionalization at other positions on the aromatic ring.
Following the C-H activation step, the alcohol molecule enters the catalytic cycle, undergoing coordination with the cobalt(III) intermediate. This is followed by a migratory insertion step where the alkoxy group is transferred to the indole framework. The cycle concludes with a reductive elimination process that releases the desired 2-alkoxy indole product and regenerates the active cobalt species. Understanding this mechanism is vital for impurity control, as it highlights the critical role of the oxidant stoichiometry and reaction temperature in maintaining the catalytic turnover. The use of silver carbonate not only serves as the terminal oxidant but also helps in scavenging protons generated during the process, ensuring the reaction proceeds to completion with minimal byproduct formation. This mechanistic clarity allows process chemists to fine-tune reaction parameters to maximize yield and purity, essential for meeting the rigorous specifications of high-purity pharmaceutical intermediates.
How to Synthesize 2-Alkoxy Indole Efficiently
The practical execution of this synthesis is designed for scalability and reproducibility, utilizing standard laboratory equipment that translates easily to pilot plant reactors. The protocol involves charging a reaction vessel with the indole substrate, the cobalt catalyst, and the oxidant, followed by the addition of the alcohol solvent. The mixture is then heated to a controlled temperature range of 90-110°C for a duration of 16 to 24 hours. This extended reaction time ensures complete conversion of the starting material, which is critical for minimizing downstream purification burdens. The detailed standardized synthesis steps for implementing this protocol are outlined in the guide below.
- Charge a reaction vessel with cobalt acetylacetonate catalyst (20 mol%), the specific indole substrate, and silver carbonate oxidant (2.0 equivalents).
- Add the corresponding alcohol solvent (acting as both solvent and reactant, approx. 2.0 mL per 0.2 mmol substrate) to the mixture.
- Heat the reaction mixture to 100°C and stir for 16 to 24 hours until conversion is complete, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from precious metals to base metals like cobalt represents a significant opportunity for cost optimization, as the price volatility associated with palladium and rhodium is completely avoided. Moreover, the simplification of the synthetic route by eliminating pre-functionalization steps reduces the number of unit operations required, leading to shorter manufacturing cycles and lower labor costs. The use of alcohols as both reactants and solvents further simplifies the bill of materials, reducing the complexity of solvent recovery and waste management systems. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive cobalt acetylacetonate drastically lowers the direct material costs associated with catalysis. Additionally, the high atom economy of the direct C-H activation means less raw material is wasted in the form of leaving groups or byproducts, further enhancing the overall process economics. The simplified workup procedure, which primarily involves filtration and standard chromatography, reduces the consumption of silica gel and eluents, contributing to substantial cost savings in the purification stage.
- Enhanced Supply Chain Reliability: The starting materials for this process, including the indole derivatives and simple alcohols, are commodity chemicals that are readily available from multiple global suppliers. This abundance mitigates the risk of supply disruptions that often plague specialized reagents. Furthermore, the robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to maintain consistent inventory levels and meet tight delivery deadlines without compromising on quality or safety standards.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require high-pressure equipment or cryogenic temperatures, making the scale-up from gram to kilogram and ton scales straightforward and safe. The reduced reliance on hazardous halogenated solvents and the minimization of heavy metal waste align with increasingly stringent environmental regulations. This green chemistry profile not only simplifies regulatory compliance but also enhances the corporate sustainability image, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the method's capabilities and limitations for potential partners.
Q: What are the key advantages of this cobalt-catalyzed method over traditional palladium catalysis?
A: This method utilizes earth-abundant cobalt instead of expensive noble metals like palladium or rhodium, significantly reducing raw material costs. Furthermore, it achieves direct C-H activation at the 2-position without requiring pre-functionalized halogenated starting materials, streamlining the synthetic route.
Q: What is the substrate scope for the R and R' groups in this synthesis?
A: The process demonstrates excellent compatibility with various substituents. The R group on the nitrogen can be C1-C4 alkyl, aryl, or benzyl groups, while the R' group (from the alcohol) accommodates C1-C5 alkyl chains and even trifluoroethyl groups, allowing for diverse structural modifications.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method is scalable to gram-level production and beyond. The use of simple Schlenk tube conditions, commercially available catalysts, and straightforward workup procedures (filtration and column chromatography) makes it highly amenable to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkoxy Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115772157A can be seamlessly translated into robust manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch of 2-alkoxy indole compound we produce adheres to the highest industry standards, providing our clients with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your timeline and optimize your budget for the commercial scale-up of complex pharmaceutical intermediates.
