Scalable Cobalt-Catalyzed C-H Alkoxylation for High-Purity Pharmaceutical Intermediates
Scalable Cobalt-Catalyzed C-H Alkoxylation for High-Purity Pharmaceutical Intermediates
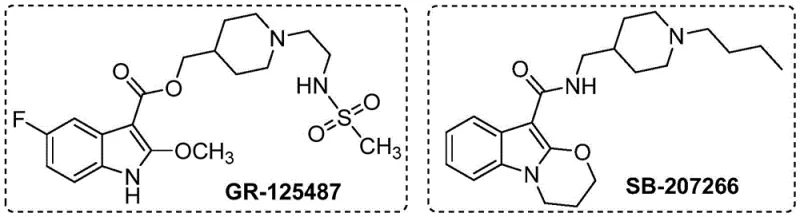
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and efficient synthetic routes. A significant breakthrough in this domain is detailed in patent CN115772157A, which introduces a novel preparation method for 2-alkoxy indole compounds. These structural motifs are not merely academic curiosities; they serve as the critical backbone for potent bioactive molecules, including selective 5-HT4 receptor antagonists such as GR-125487 and SB-207266. The ability to access these scaffolds efficiently is paramount for drug discovery teams aiming to accelerate lead optimization cycles.
This proprietary technology leverages a transition metal cobalt-catalyzed C-H activated alkoxylation reaction. Unlike traditional methodologies that often rely on expensive noble metals or cumbersome multi-step sequences, this approach utilizes earth-abundant cobalt to directly functionalize the indole core. For procurement managers and supply chain directors, this represents a tangible opportunity for cost reduction in pharmaceutical intermediate manufacturing. By shifting away from precious metal dependency, manufacturers can stabilize supply chains against volatile commodity markets while maintaining rigorous quality standards required for high-purity API intermediates.
The strategic value of this synthesis extends beyond mere cost savings; it addresses the critical bottleneck of scalability. The patent explicitly demonstrates that the method is operationally simple, requiring standard laboratory equipment and commercially available reagents. This ease of operation translates directly into reduced lead times for high-purity pharmaceutical intermediates, enabling faster transition from gram-scale discovery to kilogram-scale pilot production. As we delve deeper into the technical specifics, it becomes clear why this cobalt-catalyzed route is poised to become a preferred method for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-alkoxy indole compounds has been fraught with challenges that hinder efficient commercial production. Traditional pathways often necessitate multi-step sequences involving harsh reaction conditions, sensitive intermediates, and the use of stoichiometric amounts of expensive reagents. Furthermore, many established protocols rely heavily on noble metal catalysts such as palladium or rhodium. While effective on a small scale, these metals pose significant economic and environmental burdens when scaled up. The high cost of these catalysts, coupled with the stringent regulatory requirements for removing trace heavy metals from final drug substances, creates a complex and expensive purification burden.
Additionally, conventional methods frequently suffer from poor atom economy and limited substrate tolerance. The need for pre-functionalized starting materials or extensive protecting group strategies adds unnecessary steps, increasing waste generation and lowering overall yield. For a reliable pharmaceutical intermediate supplier, these inefficiencies translate into higher production costs and longer delivery timelines. The inability to directly install the alkoxy group via C-H activation means that chemists must often resort to circuitous routes that are neither green nor economically viable for large-scale commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
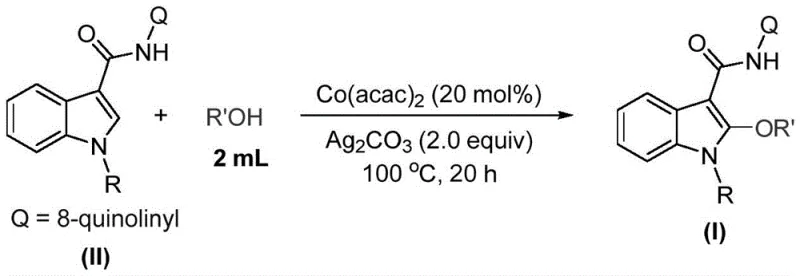
In stark contrast, the methodology disclosed in patent CN115772157A offers a streamlined, direct solution. By employing a cobalt-catalyzed C-H activation strategy, this process bypasses the need for pre-halogenated substrates or noble metals. The reaction utilizes cobalt acetylacetonate, an inexpensive and readily available catalyst, in conjunction with silver carbonate as an oxidant. This combination allows for the direct alkoxylation of the indole C2 position using simple alcohols as both the solvent and the alkoxylating agent. This dual role of the alcohol simplifies the reaction matrix significantly, reducing the number of components and potential side reactions.
The robustness of this novel approach is evidenced by its broad substrate scope and mild reaction conditions. Operating at temperatures between 90°C and 110°C, the system is compatible with a wide array of functional groups on the indole nitrogen, including alkyl, aryl, and benzyl substituents. This versatility ensures that the method can be applied to the synthesis of diverse libraries of compounds without the need for extensive process re-optimization. For R&D teams, this means a reliable platform technology that can be rapidly deployed to generate new analogues, thereby accelerating the drug discovery pipeline while ensuring cost reduction in API manufacturing through simplified processing.
Mechanistic Insights into Cobalt-Catalyzed C-H Alkoxylation
Understanding the mechanistic underpinnings of this transformation is crucial for appreciating its efficiency and selectivity. The reaction proceeds through a sophisticated catalytic cycle initiated by the oxidation of the cobalt(II) catalyst by silver carbonate. This oxidation step generates a high-valent cobalt(III) species, which is the active catalytic entity. This cobalt(III) intermediate then coordinates with the indole substrate, specifically directing the metal center to the C2 position of the indole ring. This coordination is facilitated by the directing group inherent in the substrate structure, ensuring high regioselectivity for the C2-alkoxylation over other potential sites.
Following coordination, the mechanism involves a single electron transfer (SET) process that generates a radical cobalt(II) complex. This radical species is subsequently re-oxidized by silver carbonate, concurrently activating the C-H bond at the 2-position of the indole to form a stable cobalt(III) organometallic intermediate. The final stage of the cycle involves the coordination of the alcohol solvent to the metal center, followed by migratory insertion and reductive elimination. This sequence releases the desired 2-alkoxy indole product and regenerates the cobalt catalyst, allowing the cycle to continue. This elegant mechanism minimizes waste and maximizes atom economy, key factors for sustainable chemical manufacturing.
From an impurity control perspective, this mechanism offers distinct advantages. The use of silver carbonate as a terminal oxidant produces benign byproducts that are easily removed during the workup phase. Furthermore, the high selectivity of the cobalt catalyst reduces the formation of regioisomers or over-alkoxylated byproducts, which are common issues in less selective electrophilic substitution reactions. This inherent selectivity simplifies the downstream purification process, typically requiring only filtration and standard column chromatography to achieve high purity levels. For quality control laboratories, this translates to cleaner crude profiles and more consistent batch-to-batch reproducibility.
How to Synthesize 2-Alkoxy Indole Efficiently
The practical implementation of this synthesis is designed to be accessible and robust, making it ideal for both laboratory research and pilot plant operations. The general procedure involves charging a reaction vessel with the cobalt catalyst, the indole substrate, and the silver carbonate oxidant. The alcohol, which serves as the reactant, is added in excess to act as the solvent, ensuring high concentration of the nucleophile. The mixture is then heated to approximately 100°C and stirred for a period ranging from 16 to 24 hours to ensure complete conversion. Detailed standardized synthesis steps are provided in the guide below.
- Combine cobalt acetylacetonate catalyst, the specific indole substrate, and silver carbonate oxidant in a reaction vessel.
- Add the corresponding alcohol solvent (acting as both solvent and reactant) and stir the mixture thoroughly.
- Heat the reaction mixture to 100°C for approximately 20 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the shift to this cobalt-catalyzed methodology offers profound strategic benefits. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing scarce noble metals with abundant cobalt salts, manufacturers can insulate themselves from the price volatility associated with precious metal markets. This stability is crucial for long-term contract manufacturing agreements where fixed pricing models are often required. Furthermore, the use of simple alcohols as reagents eliminates the need for specialized, hazardous alkylating agents, enhancing workplace safety and reducing regulatory compliance costs.
- Cost Reduction in Manufacturing: The economic impact of substituting palladium or rhodium with cobalt acetylacetonate cannot be overstated. Cobalt catalysts are orders of magnitude cheaper than their noble metal counterparts, leading to a significant decrease in the bill of materials. Additionally, the simplified workup procedure, which avoids complex extraction or scavenging steps for heavy metals, reduces labor hours and solvent consumption. These cumulative efficiencies result in substantial cost savings per kilogram of produced intermediate, improving overall margin potential for the final API.
- Enhanced Supply Chain Reliability: The reliance on commercially available, commodity-grade reagents enhances supply security. Cobalt acetylacetonate, silver carbonate, and common alcohols are widely sourced from multiple global suppliers, mitigating the risk of single-source bottlenecks. This diversification of the supply base ensures consistent availability of critical starting materials, thereby reducing lead times and preventing production delays. For supply chain heads, this reliability is a key metric in vendor selection and risk management strategies.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The reaction conditions are mild enough to be managed in standard glass-lined or stainless steel reactors without requiring exotic high-pressure equipment. Moreover, the reduced use of toxic heavy metals aligns with increasingly stringent environmental regulations regarding waste disposal and residual metal limits in pharmaceutical products. This 'green' profile facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of using cobalt over palladium for this synthesis?
A: Cobalt catalysts are significantly more abundant and cost-effective than noble metals like palladium or rhodium. This substitution drastically lowers the raw material costs and simplifies the removal of heavy metal residues, which is critical for pharmaceutical compliance.
Q: Does this method support diverse functional groups on the indole ring?
A: Yes, the patented process demonstrates excellent substrate compatibility. It tolerates various substituents at the nitrogen position, including alkyl, aryl, and benzyl groups, allowing for the synthesis of a wide library of derivatives without protecting group strategies.
Q: Is the purification process complex for large-scale production?
A: The workup is designed for simplicity. Following the reaction, the mixture undergoes filtration to remove solid byproducts, followed by standard silica gel column chromatography. This straightforward isolation method facilitates easier scale-up compared to multi-step traditional syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkoxy Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN115772157A. As a premier CDMO partner, we possess the technical expertise to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cost-effective synthesis for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can optimize your supply chain for 2-alkoxy indole intermediates.
