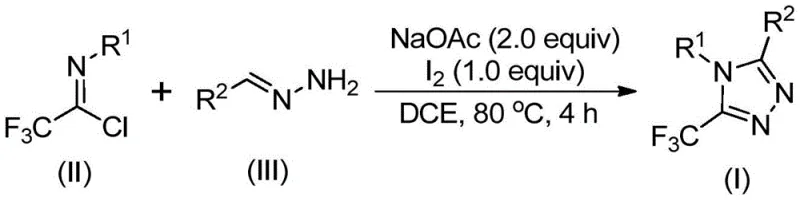
Advanced Iodine-Catalyzed Process for Scalable Production of High-Purity Trifluoromethyl Triazole Pharmaceutical Intermediates
The recently granted Chinese patent CN110467579B discloses a groundbreaking synthetic methodology for producing 5-trifluoromethyl substituted 1,2,4-triazole compounds, which represent critical building blocks in modern pharmaceutical development. This innovative approach addresses longstanding challenges in heterocyclic chemistry by eliminating the requirement for anhydrous and anaerobic reaction environments while completely avoiding toxic heavy metal catalysts that complicate traditional manufacturing processes. The methodology leverages readily available starting materials including sodium acetate, trifluoroethylimidoyl chloride derivatives, and hydrazone compounds to construct these valuable molecular frameworks through a streamlined sequence. Notably, the process operates efficiently at standard atmospheric conditions with simple heating protocols that enhance operational safety and reduce equipment complexity compared to conventional techniques. This advancement holds significant implications for pharmaceutical manufacturers seeking sustainable routes to produce high-value intermediates with stringent purity requirements. The patent establishes a robust foundation for scalable production that aligns with current industry demands for environmentally responsible chemical manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches for synthesizing trifluoromethylated heterocycles typically require either expensive trifluoromethylation reagents operating under strictly anhydrous conditions or transition metal-catalyzed processes that necessitate complex purification steps to remove toxic residues from final products. These methods often involve hazardous reagents like trifluorodiazoethane that demand specialized handling equipment and generate significant waste streams requiring costly disposal procedures. Furthermore, conventional techniques exhibit limited substrate scope due to sensitivity toward functional groups and frequently require cryogenic temperatures or inert atmospheres that substantially increase operational complexity and energy consumption. The reliance on palladium or copper catalysts creates additional challenges in meeting pharmaceutical industry purity standards due to stringent metal residue limits that necessitate multiple purification stages. Such constraints have historically restricted commercial adoption despite the high demand for these biologically active molecular scaffolds across therapeutic areas.
The Novel Approach
The patented methodology overcomes these limitations through an iodine-promoted cascade reaction that operates efficiently under ambient conditions without requiring specialized equipment or hazardous reagents. By utilizing elemental iodine as a non-toxic promoter instead of transition metals, the process eliminates costly catalyst removal steps while maintaining excellent reaction efficiency across diverse substrate combinations. The reaction proceeds through a well-defined mechanism involving base-mediated carbon-nitrogen bond formation followed by isomerization and oxidative cyclization that tolerates a wide range of functional groups on both coupling partners. Crucially, the method demonstrates exceptional operational simplicity with straightforward workup procedures using standard column chromatography that achieves high purity levels suitable for pharmaceutical applications. This approach enables flexible molecular design through strategic substitution patterns on both starting materials while maintaining consistent product quality across different scales.
Mechanistic Insights into Iodine-Promoted Triazole Cyclization
The reaction mechanism begins with sodium acetate-mediated deprotonation facilitating nucleophilic attack by hydrazone nitrogen on the electrophilic carbon of trifluoroethylimidoyl chloride to form an intermediate amidine species. This initial carbon-nitrogen bond formation occurs readily at elevated temperatures without requiring inert atmosphere due to the stability of the reaction components under thermal conditions. Subsequent isomerization generates a key imidate intermediate that undergoes base-promoted oxidative iodination where elemental iodine serves as both oxidant and iodine source without introducing metallic impurities. The resulting iodinated species then participates in an intramolecular electrophilic substitution followed by aromatization through dehydroiodination to yield the final triazole product with excellent regioselectivity at the five-position.
Impurity control is achieved through precise stoichiometric control of sodium acetate relative to iodine (maintained at a consistent ratio) which prevents over-oxidation side reactions while ensuring complete conversion of starting materials. The thermal profile at exactly eighty degrees Celsius provides optimal kinetic control that minimizes decomposition pathways while promoting selective cyclization through favorable transition state geometry. Substrate design flexibility allows strategic placement of electron-donating or electron-withdrawing groups on both coupling partners without compromising reaction efficiency or product purity due to the robust nature of the iodine-mediated transformation. This mechanistic understanding enables reliable prediction of impurity profiles across diverse substrate combinations while maintaining consistent product quality meeting pharmaceutical industry specifications.
How to Synthesize Trifluoromethyl Triazole Efficiently
This patented synthesis route represents a significant advancement in heterocyclic chemistry by providing a practical solution to longstanding challenges in producing trifluoromethylated triazoles at commercial scale. The methodology eliminates multiple pain points associated with conventional approaches through its elegant use of non-toxic reagents under standard processing conditions while maintaining excellent yield profiles across diverse substrate combinations. Detailed standardized synthesis procedures have been developed based on this patent disclosure that enable consistent production of high-purity intermediates meeting stringent pharmaceutical requirements. The following step-by-step guide provides essential operational parameters for successful implementation of this innovative manufacturing process.
- Combine sodium acetate (2.0 equiv), trifluoroethylimidoyl chloride (II), and hydrazone (III) in dichloroethane solvent under ambient conditions without requiring anhydrous or anaerobic environments.
- Heat the reaction mixture to precisely 80°C and maintain this temperature for four hours to facilitate base-promoted carbon-nitrogen bond formation and isomerization.
- Add elemental iodine (1.0 equiv) and continue heating at constant temperature for one additional hour to complete oxidative iodination and intramolecular cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing approach directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming traditionally complex syntheses into streamlined processes with significant operational benefits. The elimination of specialized equipment requirements and hazardous reagents reduces capital investment needs while enhancing workplace safety profiles across manufacturing facilities. By leveraging readily available starting materials from established chemical suppliers worldwide, this method creates substantial resilience against supply chain disruptions that commonly affect specialty chemical production. The simplified process design also enables faster technology transfer between manufacturing sites while maintaining consistent product quality through reduced process complexity.
- Cost Reduction in Manufacturing: The complete avoidance of transition metal catalysts eliminates expensive catalyst procurement costs along with associated waste treatment expenses required for metal residue removal from final products. Simplified processing under standard atmospheric conditions reduces energy consumption by eliminating vacuum systems or inert gas requirements while minimizing equipment maintenance costs through reduced corrosion from harsh reagents. The use of commodity chemicals as starting materials provides substantial cost advantages over specialized reagents required by conventional methods while maintaining excellent yield profiles across diverse substrate combinations.
- Enhanced Supply Chain Reliability: Sourcing flexibility is significantly improved through the use of widely available starting materials including sodium acetate and common organic solvents that maintain stable global supply chains unaffected by geopolitical disruptions. The absence of specialized handling requirements enables broader manufacturing site selection while reducing lead times associated with hazardous material transportation protocols. Process robustness across different raw material batches ensures consistent product quality even when sourcing from multiple suppliers without requiring extensive revalidation procedures.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production is facilitated by straightforward process intensification without requiring complex engineering modifications or specialized containment systems. Reduced environmental impact stems from eliminating toxic metal catalysts and hazardous reagents while generating less waste through higher atom economy in the cyclization step. The simplified purification protocol minimizes solvent consumption during workup while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices across global pharmaceutical markets.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations based on detailed analysis of patent CN110467579B's experimental data and mechanistic insights relevant to pharmaceutical manufacturing operations. These answers provide essential information for technical evaluation teams assessing process feasibility and implementation requirements for commercial production.
Q: How does this method eliminate heavy metal catalyst requirements?
A: The process utilizes elemental iodine as a non-toxic promoter instead of transition metals like palladium or copper, completely avoiding expensive catalyst removal steps and hazardous waste streams while maintaining high reaction efficiency through iodine-mediated oxidative cyclization.
Q: What scalability advantages does this synthesis route offer?
A: Operating under standard atmospheric conditions with simple heating protocols enables seamless transition from laboratory scale to commercial production volumes without specialized equipment requirements or complex process modifications.
Q: How does the process ensure high purity without complex purification?
A: The selective iodine-promoted cyclization minimizes side reactions through controlled reaction kinetics while simple column chromatography effectively isolates high-purity products meeting pharmaceutical industry specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a leading CDMO specialist in complex heterocyclic synthesis, we have successfully implemented this patented iodine-promoted methodology across multiple client projects requiring high-purity trifluoromethyl triazole intermediates for pharmaceutical applications. Our dedicated technical team ensures seamless technology transfer from laboratory development through full-scale manufacturing while maintaining complete regulatory compliance throughout all production phases.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your manufacturing requirements. Contact us today to discuss how our expertise in scalable triazole synthesis can support your next-generation pharmaceutical development programs with reliable supply chain solutions.
