Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale-Up
Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, due to their pervasive presence in bioactive molecular frameworks. A groundbreaking development in this sector is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward by utilizing a metal-free oxidative cyclization strategy that bypasses the need for expensive transition metal catalysts and harsh reaction conditions. The core innovation lies in the direct coupling of 2-methylquinoline derivatives with trifluoroacetimidohydrazides using a tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP) system. For R&D directors and procurement specialists, this patent offers a compelling pathway to access high-value intermediates with improved purity profiles and reduced environmental impact, positioning it as a critical asset for modern supply chains.
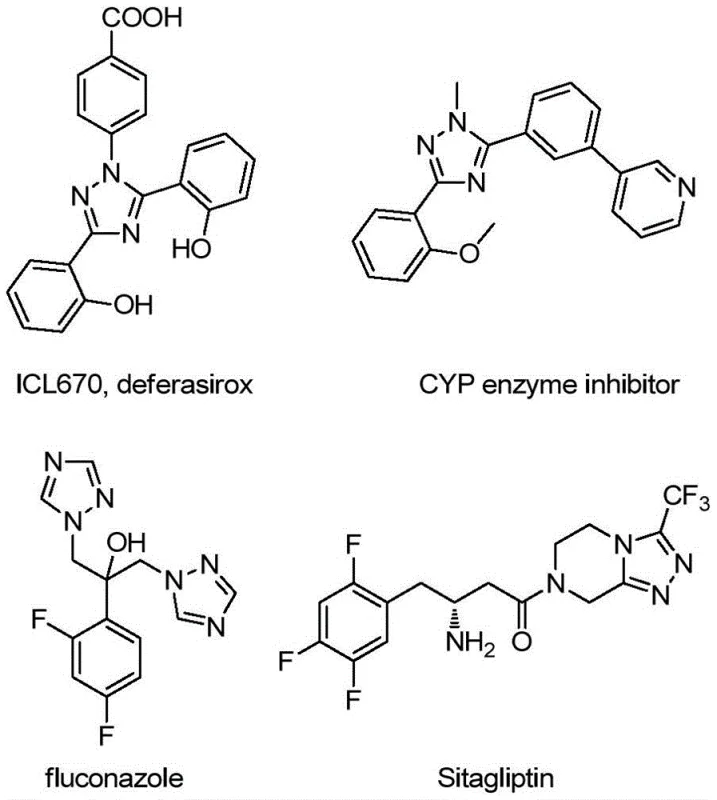
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiencies that hinder large-scale commercial application. Traditional protocols often rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step synthetic sequence to achieve the final heterocyclic structure. This multi-step approach not only results in a dismal total yield of approximately 17%, leading to substantial material waste, but also requires severe reaction conditions that demand rigorous control of temperature and pressure. Furthermore, the reliance on carboxylic acid precursors often involves activation steps that generate stoichiometric amounts of hazardous byproducts, complicating downstream purification and waste management. For a reliable pharmaceutical intermediate supplier, these factors translate into higher production costs, longer lead times, and increased regulatory scrutiny regarding solvent residues and heavy metal contamination, making such legacy routes economically unviable for high-volume manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN113307790B introduces a streamlined, one-pot oxidative cyclization protocol that fundamentally reshapes the economic landscape of triazole synthesis. By employing readily available 2-methylquinoline and trifluoroacetimidohydrazide as starting materials, the process eliminates the need for pre-functionalized carboxylic acids and reduces the synthetic complexity to a single transformation. The reaction is promoted by a catalytic system comprising tetrabutylammonium iodide and tert-butyl peroxide, which facilitates the direct conversion of the methyl group into the requisite aldehyde intermediate in situ, followed by condensation and cyclization. This novel approach operates effectively at moderate temperatures between 80°C and 100°C and does not require stringent anhydrous or oxygen-free environments, drastically simplifying operational requirements. The ability to tolerate diverse functional groups on both the quinoline and hydrazide moieties further enhances the versatility of this method, allowing for the rapid generation of diverse libraries of bioactive candidates.
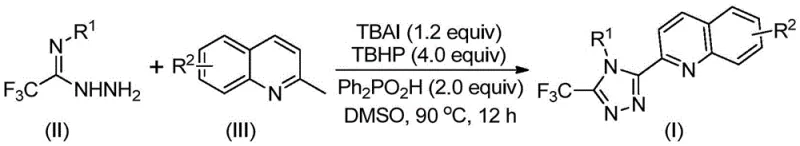
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic elegance of this transformation lies in its radical-mediated pathway, which avoids the use of toxic heavy metals while maintaining high catalytic efficiency. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate by the TBAI/TBHP system, generating a reactive 2-quinolinecarbaldehyde intermediate. This aldehyde subsequently undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone species. The presence of diphenylphosphoric acid is critical at this stage, as it likely acts as a proton shuttle or hydrogen-bond donor to facilitate the subsequent oxidative iodination and intramolecular electrophilic substitution. This sequence culminates in the formation of the 1,2,4-triazole ring through aromatization, driven by the thermodynamic stability of the heterocyclic system. The entire process can proceed via a free radical mechanism, where iodine radicals generated from TBAI abstract hydrogen atoms, propagating the oxidation cycle without the need for external metal oxidants. This metal-free nature is particularly advantageous for pharmaceutical applications, as it inherently minimizes the risk of heavy metal impurities in the final API, thereby reducing the burden on quality control laboratories.
From an impurity control perspective, the specificity of the TBAI/TBHP system ensures that side reactions are minimized, leading to cleaner reaction profiles. The use of diphenylphosphoric acid not only accelerates the reaction rate but also suppresses the formation of over-oxidized byproducts or polymerization of the aldehyde intermediate. The reaction conditions are mild enough to preserve sensitive functional groups such as halogens, methoxy, and nitro groups on the aromatic rings, as evidenced by the successful synthesis of various substituted derivatives in the patent examples. This high level of chemoselectivity is crucial for R&D teams aiming to synthesize complex analogs for structure-activity relationship (SAR) studies. Furthermore, the post-treatment process is straightforward, involving simple filtration and silica gel column chromatography, which are standard unit operations in any chemical manufacturing facility, ensuring that the technology can be seamlessly integrated into existing production lines without significant capital investment.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis route is designed for ease of execution, making it accessible for both laboratory-scale discovery and pilot-plant production. The protocol involves charging a reaction vessel with the specific molar ratios of tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in a polar aprotic solvent such as DMSO. The mixture is then heated to the optimal temperature range and stirred for a defined period until conversion is complete. Detailed standardized synthesis steps for specific derivatives, including precise stoichiometry and work-up procedures, are outlined in the guide below to ensure reproducibility and safety.
- Mix tetrabutylammonium iodide, tert-butyl peroxide, diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours under air atmosphere.
- Filter the mixture, load onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The shift from a five-step legacy process to a one-pot synthesis drastically reduces the number of unit operations, which in turn lowers labor costs, energy consumption, and equipment occupancy time. By eliminating the need for expensive transition metal catalysts, the process removes the costly and time-consuming step of heavy metal scavenging and residual testing, which is a major bottleneck in API manufacturing. The use of commodity chemicals like TBAI and TBHP, which are widely available from multiple global suppliers, mitigates the risk of raw material shortages and price volatility associated with specialized reagents. This supply chain robustness ensures consistent production schedules and reliable delivery timelines for downstream customers.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the significant simplification of the synthetic route and the use of inexpensive, bulk-available reagents. By consolidating multiple reaction steps into a single pot, manufacturers can achieve substantial savings in solvent usage, waste disposal, and personnel hours. The avoidance of precious metal catalysts eliminates a major cost center, while the high yields reported in the patent examples mean that less starting material is required to produce the same amount of product, further driving down the cost of goods sold. Additionally, the mild reaction conditions reduce energy demands for heating and cooling, contributing to a lower overall carbon footprint and operational expenditure.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 2-methylquinoline and aromatic amines ensures a stable and diversified supply base. Unlike proprietary intermediates that may be sourced from a single vendor, these commodities can be procured from multiple chemical distributors globally, reducing the risk of supply disruption. The robustness of the reaction to moisture and oxygen means that storage and handling requirements are less stringent, lowering logistics costs and minimizing the potential for batch failures due to environmental exposure. This reliability is critical for maintaining continuous manufacturing flows and meeting the just-in-time delivery expectations of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively on gram scales with clear pathways to kilogram and tonne-level production. The use of DMSO, a high-boiling polar solvent, facilitates heat transfer and mixing in large reactors, while the absence of toxic heavy metals simplifies wastewater treatment and regulatory compliance. The atom economy of the oxidative cyclization is superior to traditional methods, generating fewer byproducts and reducing the volume of hazardous waste that requires disposal. This alignment with green chemistry principles not only satisfies increasingly strict environmental regulations but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolio.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid and five steps with low yields, this patent describes a one-pot oxidative cyclization using cheap 2-methylquinoline, achieving yields up to 97% without heavy metal catalysts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available reagents like TBAI and TBHP, operates under ambient air without strict anhydrous conditions, and utilizes standard solvents like DMSO, making it highly scalable for commercial manufacturing.
Q: What is the role of diphenylphosphoric acid in this reaction?
A: Diphenylphosphoric acid acts as a crucial additive that promotes the oxidative cyclization efficiency, significantly improving reaction yields and substrate tolerance compared to reactions performed without it.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the one described in CN113307790B for advancing drug discovery and development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative cyclization reactions, including rigorous QC labs that enforce stringent purity specifications to guarantee the quality of every batch. We are committed to delivering high-purity intermediates that meet the exacting standards of the global pharmaceutical industry, leveraging our technical expertise to optimize yields and minimize impurities.
We invite you to collaborate with us to unlock the full potential of this technology for your specific projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can enhance your margins. Please contact our technical procurement team today to request specific COA data for related triazole derivatives and discuss route feasibility assessments for your target molecules. Let us be your partner in turning complex chemical challenges into commercial successes.
