Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multicomponent strategy for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This innovation addresses critical bottlenecks in traditional organic synthesis by leveraging a palladium-catalyzed carbonylation cascade that operates under remarkably mild conditions. For R&D directors and process chemists, this represents a significant leap forward in accessing diversified chemical space without the logistical nightmares associated with high-pressure carbon monoxide handling. The methodology not only streamlines the synthetic route but also enhances the safety profile of the manufacturing process, making it an attractive candidate for reliable pharmaceutical intermediate supplier networks looking to optimize their portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has relied heavily on stepwise approaches that often suffer from poor atom economy and harsh reaction conditions. Traditional methods typically involve the direct coupling of two pre-formed heterocyclic substrates, which requires extensive protecting group manipulation and generates significant waste. Alternatively, oxidative cyclization strategies using activated methyl-substituted heterocycles often necessitate stoichiometric amounts of toxic oxidants, leading to difficult purification processes and environmental compliance issues. Furthermore, conventional carbonylation reactions to install the crucial bridging carbonyl group frequently demand the use of gaseous carbon monoxide, a highly toxic and flammable reagent that requires specialized high-pressure equipment and rigorous safety protocols. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity intermediates, creating substantial friction in the supply chain for complex polymer additives or drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN115353511A utilizes a transition metal palladium-catalyzed cascade reaction that elegantly assembles the target scaffold from three simple building blocks: trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. This multicomponent reaction (MCR) strategy allows for the simultaneous formation of multiple chemical bonds in a single operational step, drastically reducing the number of isolation and purification stages required. By employing a formic acid and acetic anhydride mixture as a safe and convenient carbon monoxide surrogate, the process eliminates the need for handling hazardous CO gas, thereby simplifying the reactor setup and enhancing workplace safety. The reaction proceeds efficiently at a mild temperature of 30°C in common organic solvents like tetrahydrofuran (THF), demonstrating exceptional functional group tolerance that is rarely seen in traditional thermal cyclizations.
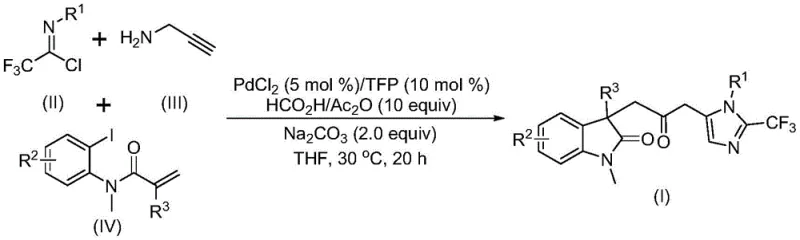
The versatility of this novel approach is further evidenced by its broad substrate scope, accommodating various electronic and steric environments on the aromatic rings. As illustrated in the reaction scheme, the system tolerates electron-donating groups such as methyl and methoxy, as well as electron-withdrawing substituents like halogens, nitro, and trifluoromethyl groups. This flexibility is paramount for medicinal chemists aiming to perform structure-activity relationship (SAR) studies, as it allows for the rapid generation of diverse analog libraries from a common core structure. The ability to synthesize these complex architectures in a one-pot fashion significantly accelerates the drug discovery timeline, providing a competitive edge in the development of new therapeutic agents.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and scale-up. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type reaction, which constructs the indolinone core and forms a divalent alkyl-palladium species. Subsequently, the carbon monoxide surrogate (generated in situ from formic acid and acetic anhydride) inserts into the palladium-carbon bond, yielding an acyl-palladium intermediate. This acyl species is the key precursor for the final ring closure. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate, which undergoes isomerization. The final step involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that releases the final carbonyl-bridged biheterocyclic product and regenerates the palladium catalyst.
From an impurity control perspective, the use of specific ligands such as tri-2-furylphosphine (TFP) plays a vital role in stabilizing the palladium center and preventing the formation of inactive palladium black precipitates. The precise stoichiometry of the reagents, particularly the excess of propargylamine and acrylamide relative to the imidoyl chloride, ensures that side reactions such as homocoupling or incomplete cyclization are minimized. The mild reaction temperature of 30°C further suppresses thermal degradation pathways that often plague high-temperature cyclizations, resulting in cleaner reaction profiles and higher crude purity. This mechanistic clarity allows process engineers to fine-tune parameters such as catalyst loading (typically 5 mol% PdCl2) and reaction time (12-20 hours) to maximize yield while maintaining cost efficiency.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-plant production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, and CO surrogate in an aprotic solvent, followed by the sequential addition of the three coupling partners. The mixture is then stirred at ambient or slightly elevated temperatures until conversion is complete, as monitored by TLC or HPLC. Detailed standardized synthesis steps see the guide below.
- Combine palladium chloride (5 mol%), TFP ligand (10 mol%), sodium carbonate (2.0 equiv), and a formic acid/acetic anhydride mixture in THF solvent.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, then filter and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are profound. The shift from multi-step, hazardous processes to a streamlined one-pot reaction translates directly into significant cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for specialized high-pressure carbon monoxide infrastructure, capital expenditure (CAPEX) for new production lines is drastically reduced, while operational expenditure (OPEX) is lowered through decreased energy consumption and simplified safety monitoring. Furthermore, the use of cheap and readily available starting materials like propargylamine and acrylamide ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or expensive reagents.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling and the reduction of synthetic steps inherently lower the processing costs. Since the reaction utilizes inexpensive palladium chloride and common ligands rather than proprietary high-cost catalysts, the overall catalyst cost per kilogram of product is minimized. Additionally, the high atom economy of the multicomponent reaction means less raw material is wasted as byproducts, leading to substantial cost savings in raw material procurement and waste disposal fees.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as formic acid, acetic anhydride, and sodium carbonate ensures that production is not bottlenecked by the availability of niche reagents. This robustness allows for consistent batch-to-batch quality and reliable delivery schedules, which is critical for maintaining continuous manufacturing operations. The simplicity of the workup procedure, involving basic filtration and standard chromatography, further reduces the turnaround time between batches, enhancing overall throughput.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be effective on the gram scale, indicating strong potential for kilogram and tonne-scale expansion. The avoidance of stoichiometric heavy metal oxidants and toxic gases aligns with increasingly stringent environmental regulations, reducing the burden on wastewater treatment facilities. This green chemistry profile not only lowers compliance costs but also enhances the corporate sustainability image, a key factor for modern supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this Pd-catalyzed method over traditional coupling?
A: This method avoids toxic carbon monoxide gas by using a formic acid/acetic anhydride surrogate, operates at mild temperatures (30°C), and utilizes cheap, commercially available starting materials like propargylamine.
Q: What is the substrate scope for the R1 and R3 groups?
A: The reaction tolerates a wide range of substituents including alkyl, substituted aryl (methyl, methoxy, halogen, nitro, trifluoromethyl), and benzyl groups, allowing for diverse library synthesis.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly demonstrates gram-scale feasibility with high reaction efficiency and simple post-treatment procedures involving filtration and column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-catalyzed multicomponent technology in accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee the highest quality standards for every batch delivered.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your strategic partner in delivering high-value chemical solutions.
