Scalable One-Pot Synthesis of Dihydroquinazolinones for Advanced Pharmaceutical Manufacturing
Scalable One-Pot Synthesis of Dihydroquinazolinones for Advanced Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving towards more sustainable and atom-economical processes. A significant breakthrough in this domain is documented in Chinese Patent CN108558778B, which introduces a highly efficient methodology for constructing the dihydroquinazolinone scaffold. This privileged structure is ubiquitous in bioactive molecules, yet its traditional synthesis often suffers from multi-step protocols and harsh reaction conditions. The patented approach leverages a novel cascade reaction sequence initiated by readily available o-nitrobenzonitrile and various carbonyl compounds. By integrating nitro reduction, nitrile hydrolysis, and cyclization into a single operational unit, this technology offers a compelling value proposition for industrial scale-up. The core innovation lies in the synergistic use of tetrahydroxydiboron as a reducing agent and cuprous chloride as a catalyst within a benign aqueous-alcoholic medium.
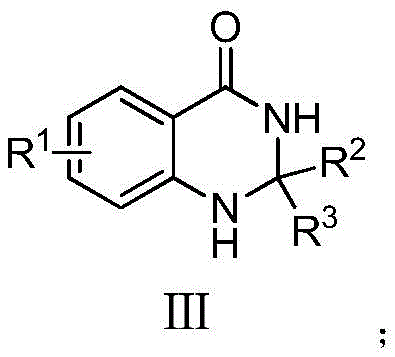
This structural versatility allows for the generation of a diverse library of derivatives, as illustrated by the general formula III where R1 can range from hydrogen to various alkyl, alkoxy, or halogen groups. The ability to tolerate such a wide array of substituents without compromising yield is critical for medicinal chemists optimizing structure-activity relationships (SAR). Furthermore, the process avoids the isolation of unstable intermediates, thereby enhancing overall process safety and throughput. For procurement and supply chain leaders, this translates to a more reliable source of complex heterocyclic building blocks with reduced lead times and simplified logistics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dihydroquinazolinone core has relied heavily on the condensation of 2-aminobenzamide or isatoic anhydride derivatives with aldehydes or ketones. While chemically sound, these classical routes present significant bottlenecks for large-scale production. The prerequisite starting materials, such as 2-aminobenzonitrile or 2-nitroaniline derivatives, often require separate synthetic lines, increasing the cumulative cost and carbon footprint of the final API intermediate. Moreover, many established protocols necessitate the use of strong acids, high temperatures, or expensive transition metal catalysts that are difficult to remove to ppm levels required by regulatory standards. The reliance on hazardous organic solvents like dichloromethane or DMF further complicates waste management and increases the cost of goods sold (COGS) due to stringent environmental compliance measures. These factors collectively hinder the economic viability of producing dihydroquinazolinones for generic drug markets where margin compression is intense.
The Novel Approach
In stark contrast, the methodology disclosed in CN108558778B revolutionizes the synthesis by employing a direct reductive cyclization strategy. As depicted in the reaction scheme below, the process utilizes o-nitrobenzonitrile (I) and a carbonyl compound (II) as the sole organic inputs. This one-pot transformation is driven by the in situ generation of reactive species that sequentially reduce the nitro group and hydrolyze the nitrile functionality before final cyclization occurs. The use of tetrahydroxydiboron serves a dual purpose: it acts as a clean reducing agent that generates minimal boron-containing waste compared to traditional hydride sources, and it facilitates the activation of the nitrile group. The reaction proceeds smoothly at a moderate temperature of 60°C, eliminating the energy intensity associated with refluxing conditions. This streamlined approach not only improves the step economy but also significantly enhances the atom economy, making it an ideal candidate for green chemistry certification and sustainable manufacturing initiatives.
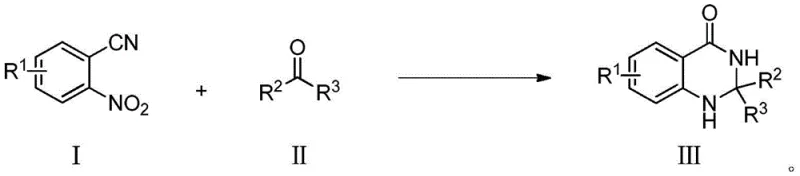
Mechanistic Insights into CuCl-Catalyzed Reductive Cyclization
The success of this transformation hinges on the intricate interplay between the copper catalyst and the boron reagent. Mechanistically, the cuprous chloride (CuCl) likely coordinates with the nitrogen atoms of the substrate, facilitating electron transfer processes essential for the reduction of the nitro group to an amine equivalent. Simultaneously, the tetrahydroxydiboron interacts with the nitrile moiety, promoting its hydrolysis to an amide or imidate intermediate under the acidic conditions provided by glacial acetic acid. This tandem activation ensures that the intramolecular nucleophilic attack occurs rapidly once the amine functionality is generated, driving the equilibrium towards the formation of the stable dihydroquinazolinone ring. The choice of a mixed solvent system, specifically methanol and water in a 1:1 ratio, is crucial for solubilizing both the organic substrates and the inorganic boron species, creating a homogeneous reaction environment that maximizes collision frequency and reaction rate.
From an impurity control perspective, this mechanism offers distinct advantages. The mild acidity (1.2 equivalents of acetic acid) prevents the degradation of acid-sensitive functional groups that might be present on the aromatic rings, such as esters or acetals. Furthermore, the specificity of the CuCl catalyst minimizes side reactions like homocoupling or over-reduction, which are common pitfalls in nitro-group transformations. The result is a crude reaction profile that is remarkably clean, often requiring only a simple extraction and column chromatography to achieve high purity. This level of selectivity is paramount for pharmaceutical applications where genotoxic impurities must be strictly controlled. The robustness of the catalytic cycle allows for the accommodation of sterically hindered ketones and electronically diverse aldehydes, expanding the chemical space accessible to process chemists.
How to Synthesize Dihydroquinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized parameters identified in the patent data. The protocol is designed to be operationally simple, avoiding the need for inert atmosphere techniques or specialized high-pressure equipment. The key to reproducibility lies in the stoichiometric balance of the reagents, particularly the ratio of the boron reagent to the nitro substrate. Detailed standard operating procedures for this specific transformation are outlined below to ensure consistent quality and yield across different batches.
- Combine o-nitrobenzonitrile, tetrahydroxydiboron, glacial acetic acid, and cuprous chloride in a methanol/water solvent system.
- Add the aldehyde or ketone substrate and heat the mixture to 60°C for approximately 3 hours to facilitate the cascade reaction.
- Extract the reaction mixture with ethyl acetate, concentrate the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. The shift from multi-step sequences to a one-pot process fundamentally alters the cost structure of the intermediate. By consolidating three distinct chemical transformations into a single reactor vessel, manufacturers can drastically reduce capital expenditure on equipment and lower utility consumption. The elimination of intermediate isolation steps also means less solvent usage, reduced labor hours for filtration and drying, and minimized material loss during transfer operations. These efficiencies compound to deliver a significantly lower cost per kilogram, providing a competitive edge in price-sensitive markets. Additionally, the reliance on commodity chemicals like o-nitrobenzonitrile and benzaldehyde ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized starting materials.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the removal of expensive catalysts and the simplification of the workflow. Traditional methods often require palladium or platinum catalysts for nitro reduction, which represent a substantial portion of the raw material cost and require complex recovery systems to prevent metal contamination. In contrast, the use of catalytic amounts of cuprous chloride, a base metal salt, reduces the catalyst cost by orders of magnitude. Furthermore, the high yields observed, often exceeding 90% for unsubstituted substrates, mean that less raw material is wasted, directly improving the material throughput and reducing the cost of goods. The simplified workup procedure, which avoids tedious crystallizations or distillations of intermediates, further drives down operational expenses.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of universally available reagents. Tetrahydroxydiboron and o-nitrobenzonitrile are bulk chemicals produced by multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions—tolerating ambient pressure and moderate temperatures—means that production can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility is crucial for maintaining supply during geopolitical disruptions or logistical bottlenecks. Moreover, the short reaction time of approximately 3 hours allows for higher batch turnover rates, enabling manufacturers to respond more agilely to fluctuations in market demand and reducing inventory holding costs.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic hazards typically associated with hydride reductions. The use of water as a co-solvent not only improves safety by acting as a heat sink but also aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. Waste streams from this process are easier to treat compared to those containing heavy metals or chlorinated solvents, lowering the cost of waste disposal and environmental remediation. This green profile enhances the corporate sustainability metrics of the buying organization, supporting ESG (Environmental, Social, and Governance) goals which are becoming critical criteria for vendor selection in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dihydroquinazolinone synthesis platform. These insights are derived directly from the experimental data and scope limitations defined in the patent literature, providing a realistic framework for project planning.
Q: What are the primary advantages of using o-nitrobenzonitrile over 2-aminobenzamide?
A: Using o-nitrobenzonitrile eliminates the need for pre-synthesized amine starting materials, reducing step count and raw material costs while enabling a direct one-pot transformation.
Q: Is the catalyst system compatible with sensitive functional groups?
A: Yes, the mild conditions (60°C, aqueous alcohol solvent) and the specific CuCl/B2(OH)4 system demonstrate high tolerance for halogens, ethers, and heterocycles.
Q: How does this method impact environmental compliance in manufacturing?
A: The process utilizes water and methanol instead of toxic chlorinated solvents and avoids heavy metal waste associated with traditional reduction methods, significantly lowering environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroquinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN108558778B for the production of high-value pharmaceutical intermediates. Our technical team has extensively evaluated this CuCl-catalyzed pathway and confirmed its suitability for commercial scale-up. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of monitoring trace metal residues and ensuring stringent purity specifications that meet or exceed international pharmacopoeia standards. We are committed to delivering dihydroquinazolinone derivatives that support your drug development timelines with unmatched consistency and quality.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us partner with you to optimize your supply chain and accelerate your path to market with cost-effective, high-quality chemical solutions.
