Achieving Commercial Scale-Up of Complex Axial Chiral Indole-Naphthalene Catalysts with Unmatched Purity and Efficiency
Patent CN110452150B represents a significant advancement in asymmetric catalysis through its innovative one-step synthesis methodology for axial chiral indole-naphthalene compounds using organocatalytic principles rather than traditional transition metal systems. This breakthrough approach leverages dynamic kinetic resolution under exceptionally mild reaction conditions to convert racemic starting materials into high-optical-purity products with er values consistently exceeding 95:5 across diverse substrate combinations. The process eliminates multi-step sequences that have historically plagued conventional coupling methodologies while maintaining operational simplicity through standard laboratory equipment requirements. By utilizing commercially available raw materials including compound 7 derivatives and phenolic substrates as reactants, this invention addresses critical industry needs for efficient production of complex chiral scaffolds essential in modern catalytic applications across pharmaceutical development and fine chemical manufacturing sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for axial chiral indole-naphthalene compounds have been severely constrained by their reliance on transition metal-catalyzed coupling reactions requiring stringent anhydrous conditions that generate significant metal waste streams necessitating complex removal protocols before product isolation can occur. These multi-step methodologies typically involve intermediate isolations leading to cumulative yield losses exceeding thirty percent while introducing additional impurity profiles that complicate final purification processes beyond acceptable pharmaceutical standards. The harsh reaction environments often demand specialized equipment including gloveboxes and cryogenic systems that substantially elevate operational costs while limiting scalability potential due to safety concerns associated with pyrophoric reagents or high-pressure operations. Furthermore, inconsistent enantioselectivity resulting from subtle variations in metal catalyst performance has created significant barriers to industrial adoption despite the structural importance of these compounds as catalytic precursors.
The Novel Approach
The patented methodology introduces a paradigm shift by implementing an organocatalytic asymmetric addition reaction that constructs the axial chiral framework in a single synthetic operation from readily available racemic precursors without requiring any transition metals or specialized infrastructure beyond standard laboratory glassware. This innovative process operates under ambient temperature conditions using a mixed solvent system of 1,1,2-tetrachloroethane and p-xylene with molecular sieves to maintain optimal reaction environment while achieving superior enantioselectivity through precise stereochemical control mechanisms inherent to the chiral phosphoric acid catalyst system. The streamlined procedure features simple workup protocols involving filtration through celite followed by standard chromatographic purification using petroleum ether/ethyl acetate mixtures that eliminate complex separation challenges associated with metal-containing systems while maintaining high product quality standards across diverse substrate combinations as demonstrated by extensive experimental validation.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Asymmetric Addition
The catalytic cycle begins with dual activation where the chiral phosphoric acid simultaneously protonates compound 7's indole nitrogen while coordinating compound 8's phenolic hydroxyl group through hydrogen bonding interactions within a well-defined chiral pocket created by the binaphthyl backbone's steric architecture. This stereoselective addition proceeds via a six-membered transition state where precise spatial orientation enforced by substituent groups on the catalyst framework directs nucleophilic attack from only one prochiral face while molecular sieves maintain anhydrous conditions critical for maintaining hydrogen-bonding integrity throughout the reaction sequence. The mild temperature profile (20–30°C) prevents undesired racemization pathways while allowing sufficient molecular mobility for efficient stereochemical induction across diverse substrate combinations as evidenced by consistent er values exceeding 95:5 without requiring cryogenic equipment or inert atmosphere beyond standard nitrogen purging protocols.
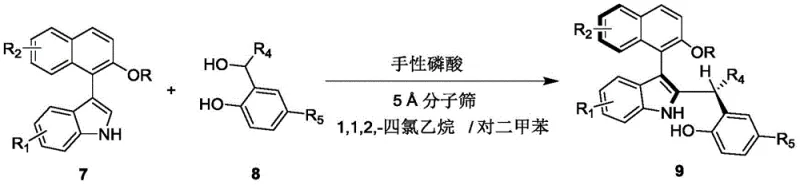
The structural integrity of the binaphthyl-derived chiral phosphoric acid catalyst proves paramount to achieving exceptional enantioselectivity through its precisely engineered steric environment where substituents like anthracenyl groups create optimal spatial constraints that differentiate between prochiral faces via π-stacking interactions while maintaining sufficient flexibility for broad substrate tolerance across various R-group combinations on both reactants. This sophisticated molecular architecture enables effective substrate discrimination through non-covalent interactions including van der Waals forces and hydrogen bonding networks that collectively enforce facial selectivity without requiring covalent modification or additional activating agents beyond the primary catalytic species itself.
Impurity formation is systematically minimized through careful optimization of multiple parameters including solvent composition ratios between tetrachloroethane and p-xylene that balance substrate solubility with transition state stabilization requirements while molecular sieves prevent hydrolysis side reactions that could compromise stereochemical integrity during extended reaction periods at ambient temperatures. The chromatographic purification protocol employing petroleum ether/ethyl acetate mixtures efficiently separates minor diastereomeric impurities without requiring specialized techniques or equipment modifications beyond standard flash chromatography systems already present in most industrial facilities thus ensuring consistent production quality meeting stringent pharmaceutical-grade specifications across multiple manufacturing scales from laboratory validation through commercial production runs.
How to Synthesize Axial Chiral Indole-Naphthalene Compounds Efficiently
This patented synthesis represents a significant advancement in organocatalytic methodology by enabling direct construction of complex axial chiral architectures from simple starting materials through a streamlined one-pot procedure that demonstrates remarkable operational simplicity while delivering exceptional stereochemical control surpassing conventional approaches in both efficiency and environmental profile metrics. The process has been validated across fourteen distinct substrate combinations demonstrating consistent performance characteristics essential for reliable industrial implementation while maintaining compatibility with existing manufacturing infrastructure requirements without necessitating capital investment in new equipment or specialized handling procedures beyond standard chemical processing capabilities found throughout global fine chemical production facilities.
- Combine compound 7 (0.1 mmol) and compound 8 (0.12 mmol) in mixed solvent (1 mL, v/v=1: 4) of 1,1,2-tetrachloroethane and p-xylene with molecular sieves
- Add chiral phosphoric acid catalyst (0.01 mmol) and stir at ambient temperature (25°C) until reaction completion monitored by TLC
- Filter through celite, concentrate under reduced pressure, and purify by silica gel column chromatography using petroleum ether/ethyl acetate (10: 1)
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this novel synthetic methodology delivers substantial strategic benefits across procurement and supply chain operations by addressing critical pain points associated with traditional production routes for complex chiral intermediates through fundamental process innovations that eliminate major cost drivers while enhancing operational resilience across multiple dimensions essential for modern chemical manufacturing enterprises serving global pharmaceutical markets.
- Cost Reduction in Manufacturing: The complete removal of expensive transition metal catalysts from the synthetic sequence eliminates associated procurement costs along with complex metal removal protocols requiring specialized equipment such as scavenging columns or ion-exchange systems that typically add multiple processing steps to conventional manufacturing workflows while simultaneously reducing waste treatment expenses through improved atom economy inherent to this streamlined approach.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials with established global supply chains ensures consistent access to critical starting components while reducing vulnerability to single-source dependencies through broad substrate tolerance that accommodates minor quality variations without compromising final product specifications thus providing greater flexibility in supplier selection strategies during periods of market volatility or regional supply disruptions.
- Scalability and Environmental Compliance: The straightforward scale-up profile from laboratory validation through commercial production is facilitated by ambient temperature operation eliminating energy-intensive heating or cooling requirements while standard filtration and chromatography techniques enable seamless adaptation to existing production facilities without requiring specialized equipment modifications thus supporting sustainable manufacturing practices aligned with evolving regulatory frameworks governing chemical production processes worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial considerations regarding implementation of this patented synthetic methodology for axial chiral indole-naphthalene compounds based on comprehensive analysis of patent documentation combined with practical implementation experience gained through industrial process development activities supporting global clients across pharmaceutical and fine chemical sectors.
Q: How does this method achieve high enantioselectivity without transition metals?
A: The chiral phosphoric acid catalyst creates a well-defined asymmetric environment through hydrogen bonding interactions that enable dynamic kinetic resolution of racemic starting materials under mild conditions.
Q: What are the key advantages over conventional coupling approaches?
A: This one-step process eliminates multiple synthetic operations while avoiding transition metal contamination concerns through organocatalytic activation modes.
Q: How does this process support scalable manufacturing?
A: The ambient temperature operation, simple workup procedures, and compatibility with standard equipment enable seamless scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at parts-per-million levels required for sensitive catalytic applications across multiple industry sectors including pharmaceutical development where optical purity directly impacts final drug efficacy profiles.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific manufacturing requirements including detailed COA data review and comprehensive route feasibility assessments tailored to your production scale needs ensuring seamless integration into existing supply chain infrastructure while maximizing operational efficiency gains derived from this patented technology platform.
