Revolutionizing Nitrobenzene Production: A Mild, Aqueous Copper-Catalyzed Strategy for Global Supply Chains
The global demand for high-purity nitrobenzene derivatives continues to surge, driven by their critical role as precursors in the synthesis of pharmaceuticals, agrochemicals, and advanced polymeric materials. However, the traditional manufacturing landscape has long been dominated by hazardous and environmentally taxing processes. Patent CN102180794B introduces a transformative approach to this challenge, detailing a novel synthetic methodology that utilizes arylboronic acids and metal nitrites in the presence of a cuprous catalyst and aqueous ammonia. This technology represents a paradigm shift from the classical electrophilic aromatic substitution, offering a pathway that is not only chemically elegant but also commercially viable for modern green chemistry standards. By leveraging inexpensive and readily available starting materials, this invention addresses the pressing need for sustainable pharmaceutical intermediates production while maintaining rigorous quality control.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the severe limitations of legacy nitration techniques. The disclosed method operates under remarkably mild conditions, typically between 10°C and 30°C, and utilizes water as the primary solvent, thereby eliminating the need for volatile organic compounds (VOCs) and corrosive mineral acids. This fundamental change in reaction engineering allows for the preservation of sensitive functional groups that are otherwise incompatible with traditional harsh nitration environments. As a result, manufacturers can access a broader chemical space, synthesizing complex nitro-aromatic scaffolds that were previously difficult or impossible to produce with high selectivity. This capability is essential for the development of next-generation active pharmaceutical ingredients (APIs) where molecular complexity is increasing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nitrobenzene compounds has relied heavily on direct nitration using a mixture of concentrated nitric acid and sulfuric acid, a process known as mixed-acid nitration. While effective for simple substrates like benzene, this classical approach presents profound drawbacks when applied to more complex aromatic systems. The reaction is highly exothermic and requires stringent temperature control to prevent runaway reactions, posing significant safety risks in large-scale operations. Furthermore, the strongly acidic and oxidative environment is indiscriminate, often leading to the degradation of sensitive functional groups such as amines, aldehydes, and hydroxyls, which limits the scope of accessible chemical structures. The generation of large volumes of acidic waste streams also creates a substantial environmental burden, necessitating costly neutralization and treatment protocols before disposal.
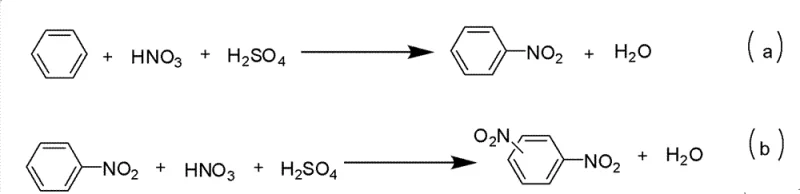
As illustrated in the reaction schemes above, the conventional electrophilic aromatic substitution often suffers from issues of regioselectivity and over-nitration. For instance, the nitration of nitrobenzene to form dinitrobenzene requires even more vigorous conditions, further exacerbating safety and purity concerns. The presence of multiple side reactions complicates the downstream purification process, often requiring energy-intensive distillation or chromatography to isolate the desired mono-nitrated product. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks in the supply chain for critical fine chemical intermediates. Consequently, there is an urgent industry-wide demand for alternative methodologies that can deliver high purity and yield without compromising on safety or environmental compliance.
The Novel Approach
In stark contrast to the brute force of mixed-acid nitration, the method disclosed in CN102180794B employs a sophisticated transition-metal catalyzed substitution strategy. By utilizing arylboronic acids as the starting material and metal nitrites (such as sodium nitrite) as the nitrogen source, the reaction proceeds through a fundamentally different mechanistic pathway. The use of a cuprous catalyst, specifically cuprous oxide (Cu2O), in conjunction with aqueous ammonia facilitates the transfer of the nitro group to the aromatic ring under exceptionally mild conditions. This approach effectively decouples the nitration process from the harsh acidic environment, allowing for the retention of delicate functional groups that define the biological activity of many drug candidates. The reaction typically proceeds at ambient pressure and temperatures ranging from 10°C to 30°C, significantly reducing the energy footprint of the manufacturing process.
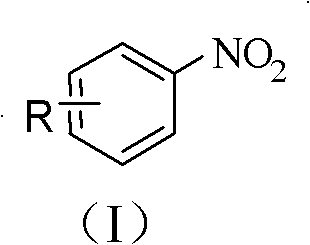
The versatility of this novel approach is further evidenced by its compatibility with a wide array of substituents on the aromatic ring. As shown in the general formula, the 'R' group can encompass alkyl, alkenyl, alkynyl, aryl, halogen, hydroxyl, amino, and carbonyl functionalities. This broad tolerance is a game-changer for medicinal chemists who require diverse building blocks for structure-activity relationship (SAR) studies. Moreover, the use of water as the solvent not only aligns with green chemistry principles but also simplifies the workup procedure, as the organic product can often be easily extracted from the aqueous phase. The combination of high selectivity, mild conditions, and operational simplicity positions this technology as a superior alternative for the commercial scale-up of complex aromatic compounds, offering a clear route to cost reduction in fine chemical manufacturing.
Mechanistic Insights into Cu-Catalyzed Nitro-Substitution
The core of this innovation lies in the unique catalytic cycle mediated by the cuprous species in an ammoniacal medium. While the exact mechanistic details may vary depending on the specific ligands and substrates, the process generally involves the activation of the arylboronic acid by the copper center, followed by transmetallation and subsequent reaction with the nitrite ion. The presence of ammonia plays a dual role: it acts as a base to facilitate the formation of the reactive boronate species and serves as a ligand to stabilize the copper catalyst, preventing its oxidation to inactive cupric species. This stabilization is crucial for maintaining catalytic turnover and ensuring high yields over extended reaction times. The mild nature of the reagents ensures that the reaction proceeds with high chemoselectivity, targeting the boron-carbon bond for substitution while leaving other sensitive moieties intact.
From an impurity control perspective, this mechanism offers distinct advantages over traditional nitration. In mixed-acid processes, impurities often arise from oxidation of the substrate, sulfonation side reactions, or polynitration. In the copper-catalyzed system, the absence of strong oxidants (other than the nitrite itself in a controlled manner) and strong acids minimizes these degradation pathways. The primary byproducts are typically inorganic salts and boric acid derivatives, which are water-soluble and easily removed during the aqueous workup. This results in a cleaner crude product profile, reducing the burden on purification units and improving the overall mass balance of the process. For quality assurance teams, this means a more consistent impurity profile and easier validation of the manufacturing process, which is critical for regulatory compliance in the pharmaceutical sector.
How to Synthesize Nitrobenzene Compounds Efficiently
The practical implementation of this synthesis route is straightforward and adaptable to both laboratory and industrial scales. The standard protocol involves charging a reactor with the arylboronic acid substrate, a catalytic amount of cuprous oxide, aqueous ammonia, and an excess of sodium nitrite in water. The mixture is then stirred at room temperature or slightly elevated temperatures (up to 30°C) for a period ranging from 3 to 48 hours, depending on the electronic nature of the substrate. Electron-rich substrates may react faster, while electron-deficient ones might require longer durations. Upon completion, the reaction mixture is extracted with an organic solvent such as ethyl acetate. The organic layer is washed to remove residual inorganic salts and catalyst, followed by concentration to yield the crude nitrobenzene derivative, which can be further purified if necessary.
- Prepare the reaction mixture by combining arylboronic acid, cuprous oxide catalyst, aqueous ammonia, and sodium nitrite in water.
- Maintain the reaction temperature between 10-30°C under atmospheric pressure for 3 to 48 hours depending on the substrate.
- Upon completion, extract the product with ethyl acetate, wash with alkali or acid as needed, and purify to obtain the high-purity nitrobenzene compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The most immediate impact is seen in the raw material costs and operational safety profile. By replacing expensive and hazardous organic solvents with water, and substituting corrosive mixed acids with benign metal nitrites, the process significantly lowers the cost of goods sold (COGS). Furthermore, the use of cuprous oxide, an abundant and inexpensive catalyst, eliminates the reliance on precious metals like palladium or platinum, which are subject to volatile market pricing and supply constraints. This stability in raw material sourcing enhances supply chain resilience, ensuring consistent production schedules even during periods of global resource scarcity.
- Cost Reduction in Manufacturing: The economic argument for this technology is robust, primarily driven by the elimination of costly waste treatment and safety infrastructure associated with traditional nitration. Since the reaction generates minimal hazardous waste and operates at ambient pressure, the capital expenditure (CAPEX) for specialized corrosion-resistant reactors and high-pressure containment systems is drastically reduced. Additionally, the high functional group tolerance means that protecting group strategies, which add steps and cost to a synthesis, can often be avoided. This streamlining of the synthetic route leads to substantial cost savings and improved overall process efficiency, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as arylboronic acids, sodium nitrite, and ammonia ensures a stable and diversified supply base. Unlike specialized reagents that may have single-source suppliers, these materials are produced globally in large volumes, mitigating the risk of supply disruptions. The mild reaction conditions also allow for manufacturing in a wider range of facilities, including those that may not be equipped for handling aggressive acids, thereby expanding the potential network of contract manufacturing organizations (CMOs). This flexibility is crucial for reducing lead time for high-purity intermediates and responding quickly to market demands.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by its inherent safety and simplicity. The absence of exothermic runaway risks and toxic gas evolution simplifies the engineering controls required for large-scale production. Moreover, the aqueous nature of the reaction aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste disposal. Companies adopting this technology can demonstrate a commitment to sustainability, potentially qualifying for green manufacturing incentives and improving their corporate social responsibility (CSR) profiles. The ability to recycle the catalytic system, as demonstrated in the patent examples, further enhances the environmental credentials and economic viability of the process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology. These insights are derived directly from the experimental data and specifications outlined in the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of this copper-catalyzed method over traditional mixed-acid nitration?
A: Unlike traditional mixed-acid nitration which requires harsh conditions (high temperature, strong acids) and suffers from poor functional group tolerance, this method operates under mild conditions (10-30°C, atmospheric pressure) using water as a solvent. It exhibits exceptional tolerance for sensitive groups like aldehydes, amines, and carboxylic acids, which would typically be degraded or oxidized in conventional processes.
Q: Is the catalyst system reusable for industrial scale-up?
A: Yes, the patent data indicates that the catalytic system demonstrates recyclability. After extracting the product, the remaining aqueous mother liquor can be replenished with fresh substrate and nitrite salt to continue the catalytic cycle, maintaining significant activity over multiple runs. This feature drastically reduces catalyst consumption and waste generation.
Q: What types of substituents are compatible with this synthesis route?
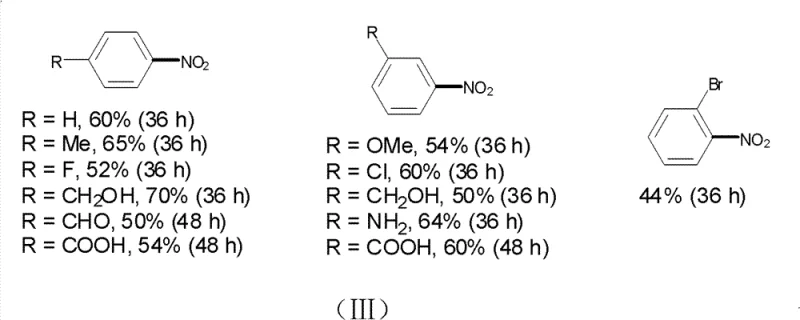
A: The method shows broad substrate scope, tolerating electron-donating groups (alkyl, alkoxy, amino) and electron-withdrawing groups (halogens, nitro, aldehyde, carboxyl). Specific examples in the patent include successful synthesis of p-nitrobenzoic acid, p-nitrobenzaldehyde, and various halogenated nitrobenzenes with yields ranging from 50% to 70%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrobenzene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed nitration technology described in CN102180794B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory method into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this mild, aqueous chemistry are fully realized in your supply chain. We are committed to delivering high-purity nitrobenzene derivatives that meet the most stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. Whether you are looking to diversify your supplier base or seeking a more sustainable alternative to traditional nitration, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis, specific COA data, and detailed route feasibility assessments tailored to your target molecules. Together, we can drive efficiency and innovation in the production of critical chemical intermediates.
