Advanced Manufacturing of Imrecoxib Intermediates via Optimized Cyclization Routes
Introduction to Next-Generation Imrecoxib Manufacturing
The pharmaceutical industry is constantly seeking more efficient, safer, and cost-effective pathways for the synthesis of active pharmaceutical ingredients (APIs) and their critical intermediates. A significant breakthrough in this domain is detailed in patent CN102206178B, which outlines a novel method for preparing Imrecoxib (also known as Erecoxib), a selective COX-2 inhibitor used in the treatment of osteoarthritis. This technology represents a paradigm shift from traditional multi-step syntheses that rely on hazardous oxidants to a streamlined, robust process centered around a dihydrofuranone intermediate. For R&D directors and procurement strategists, understanding this technological leap is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials while mitigating regulatory risks associated with heavy metal residues.
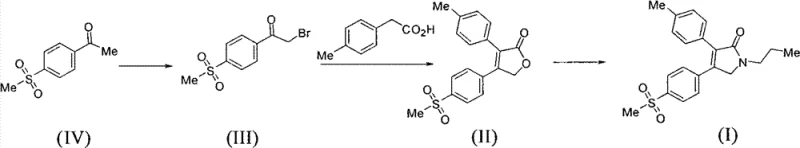
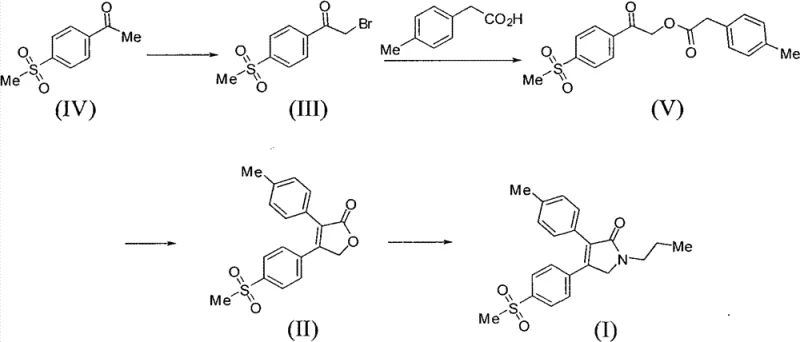
The core innovation lies in the strategic construction of the pyrrolidone ring, the pharmacophore of Imrecoxib, through a concise sequence starting from readily available methylsulfonyl acetophenone. By bypassing the cumbersome oxidation steps found in prior art, this method not only enhances the overall atom economy but also drastically simplifies the downstream purification processes. This report analyzes the technical merits of this route, its mechanistic underpinnings, and the substantial commercial advantages it offers for cost reduction in pharmaceutical intermediates manufacturing. As we delve into the specifics, it becomes evident that this approach addresses the critical pain points of scalability, environmental compliance, and supply chain stability that plague legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard industrial synthesis of Imrecoxib, as described in earlier literature such as US20040029951, was fraught with significant technical and economic inefficiencies. The conventional route typically involves a lengthy seven-step sequence that includes reduction, oxidation, and amide coupling reactions. A major bottleneck in this legacy process is the oxidation step, which necessitates the use of Jones' reagent or pyridine chromium trioxide. These reagents introduce hexavalent chromium, a toxic heavy metal, into the reaction mixture. Controlling chromium residues to meet the stringent limits required for bulk drug substances is notoriously difficult and expensive, often requiring specialized scavenging resins or complex extraction protocols. Furthermore, the oxidation step itself suffers from low yields and generates substantial amounts of hazardous waste, complicating environmental compliance and driving up disposal costs. The cumulative effect of these seven steps results in a low overall yield, high solvent consumption, and a prolonged production cycle, making the conventional method economically unviable for large-scale commercialization.
The Novel Approach
In stark contrast, the method disclosed in CN102206178B offers a radically simplified trajectory that reduces the synthetic burden while enhancing product quality. The new route condenses the synthesis into essentially three key transformations: bromination, cyclization to a dihydrofuranone, and subsequent conversion to the target pyrrolidone. By utilizing methylsulfonyl acetophenone as the starting material, the process avoids the need for external oxidants entirely. Instead, it leverages a base-mediated condensation between an alpha-bromo ketone and p-tolueneacetic acid to construct the core heterocyclic ring. This strategic redesign eliminates the chromium contamination risk at the source, ensuring a cleaner impurity profile from the outset. The operational simplicity is further enhanced by the use of common organic solvents like acetonitrile and ethanol, which are easy to recover and recycle. This streamlined approach not only accelerates the time-to-market for high-purity pharmaceutical intermediates but also aligns perfectly with modern green chemistry principles, offering a sustainable alternative for forward-thinking chemical manufacturers.
Mechanistic Insights into Base-Catalyzed Cyclization
The heart of this novel synthesis lies in the formation of the dihydrofuranone intermediate (Compound II), which serves as the pivotal scaffold for the final API. Mechanistically, this transformation involves the nucleophilic attack of the carboxylate anion of p-tolueneacetic acid on the electrophilic alpha-carbon of the alpha-bromo ketone (Compound III). Under the influence of a basic medium—such as triethylamine, DBU, or inorganic carbonates—the carboxylic acid is deprotonated to form a reactive nucleophile. This species displaces the bromide leaving group, forming an ester linkage initially. Subsequently, an intramolecular Claisen-type condensation or similar cyclization event occurs, closing the five-membered furanone ring. The choice of base and solvent plays a critical role in driving this equilibrium towards the desired cyclic product while minimizing side reactions such as hydrolysis or polymerization. Experimental data suggests that organic bases like triethylamine in polar aprotic solvents like acetonitrile provide the optimal balance of reactivity and solubility, facilitating high conversion rates at moderate temperatures around 75°C.
Following the formation of the dihydrofuranone, the final step involves a ring-opening and reclosure mechanism to install the N-propyl group, yielding the pyrrolidone structure of Imrecoxib. This reaction proceeds via the nucleophilic attack of n-propylamine on the lactone carbonyl, followed by dehydration and cyclization. The patent highlights the utility of using n-propylamine salts, such as n-propylamine acetate, to modulate the reactivity and improve the selectivity of this transformation. This mechanistic nuance is vital for impurity control, as it prevents the formation of open-chain amide byproducts that are difficult to separate. By understanding these mechanistic details, process chemists can fine-tune reaction parameters to maximize yield and purity, ensuring that the final commercial scale-up of complex pharmaceutical intermediates meets the rigorous quality standards demanded by global regulatory agencies. The absence of transition metal catalysts further simplifies the purification train, as there is no need for metal scavenging steps.
How to Synthesize Imrecoxib Efficiently
Implementing this novel synthetic route requires precise control over reaction conditions to ensure reproducibility and high quality. The process begins with the bromination of 4-methylsulfonylacetophenone, followed by the critical cyclization step to form the dihydrofuranone core, and concludes with the aminolysis to generate the final pyrrolidone. Each stage has been optimized in the patent examples to demonstrate robustness across different scales. For technical teams looking to adopt this methodology, the following guide outlines the standardized operational framework derived from the patent data. Detailed parameters regarding stoichiometry, temperature profiles, and work-up procedures are essential for achieving the reported yields and purity levels. Adhering to these proven protocols allows manufacturers to bypass the trial-and-error phase and move directly to pilot-scale validation.
- Bromination of 4-methylsulfonylacetophenone using liquid bromine in acetic acid at 40°C to obtain alpha-bromo ketone intermediate.
- Condensation and cyclization with p-tolueneacetic acid using triethylamine in acetonitrile at 75°C to form the dihydrofuranone core.
- Ring-opening and closure reaction with n-propylamine and n-propylamine acetate at 160°C to yield the final pyrrolidone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized and hazardous oxidizing agents like chromium trioxide, the process reduces dependency on suppliers of controlled substances and minimizes the logistical complexities associated with handling dangerous goods. Furthermore, the use of commodity chemicals such as acetic acid, bromine, and triethylamine ensures a stable and cost-effective supply of reagents. This stability is crucial for maintaining continuous production schedules and avoiding disruptions caused by the scarcity of exotic reagents. The simplified workflow also means fewer unit operations, which directly correlates to reduced capital expenditure on equipment and lower operational overheads.
- Cost Reduction in Manufacturing: The elimination of the oxidation step and the reduction in the total number of synthetic steps lead to significant cost savings. Without the need for expensive chromium reagents and the associated waste treatment costs, the variable cost per kilogram of the intermediate is substantially lowered. Additionally, the higher overall yield resulting from the shorter route means less raw material is wasted, improving the material efficiency of the process. The ability to use simpler solvents that are easier to recover and recycle further contributes to a leaner cost structure, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by reducing the number of potential failure points. Fewer steps mean fewer opportunities for batch failures or quality deviations. The process operates under relatively mild conditions without the need for extreme temperatures or pressures, which reduces equipment wear and tear and maintenance downtime. Moreover, the ease of purification through simple crystallization and filtration steps ensures that production throughput can be maintained at high levels, reducing lead time for high-purity pharmaceutical intermediates and enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a cleaner profile that aligns with increasingly strict global regulations. The absence of heavy metal waste simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. This compliance advantage mitigates the risk of regulatory fines and shutdowns, ensuring long-term operational continuity. The process is inherently scalable, as demonstrated by the patent examples which range from gram to multi-hundred-gram scales, indicating a clear path to ton-scale production without fundamental changes to the chemistry. This scalability ensures that the supply can grow in tandem with the commercial success of the final drug product.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this Imrecoxib synthesis method, we have compiled answers based on the specific data provided in the patent documentation. These questions reflect the concerns typically raised by technical due diligence teams during vendor qualification. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios. The answers below are grounded in the experimental results and comparative analysis presented in the source intellectual property.
Q: How does this new method improve safety compared to prior art?
A: The conventional method utilizes Jones reagent or pyridine chromium trioxide for oxidation, introducing toxic heavy metal residues that are difficult to remove and strictly regulated in APIs. This novel route eliminates the oxidation step entirely, relying on base-catalyzed cyclization, thereby removing the risk of chromium contamination and simplifying purification.
Q: What are the yield expectations for the key cyclization step?
A: Experimental data indicates that the cyclization of the alpha-bromo ketone with p-tolueneacetic acid can achieve yields ranging from approximately 53% to over 70% depending on the base and solvent system used, with optimized conditions favoring triethylamine in acetonitrile.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. It uses common solvents like acetonitrile and ethanol, avoids hazardous cryogenic conditions, and features simple work-up procedures such as filtration and crystallization, making it highly adaptable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imrecoxib Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this optimized synthetic route for Imrecoxib and are fully equipped to leverage it for our global partners. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to industrial reactor is seamless. Our state-of-the-art facilities are designed to handle the specific solvent systems and reaction conditions required for this chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems are in place to deliver reliable performance batch after batch.
We invite you to collaborate with us to unlock the full value of this advanced manufacturing technology. Whether you are looking to secure a long-term supply of high-quality intermediates or need assistance in process optimization, our technical team is ready to support your goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can enhance your supply chain resilience and drive down your overall cost of goods sold.
