Revolutionizing Amide Synthesis: Scalable NHC-Catalyzed Process for High-Purity Pharmaceutical Intermediates
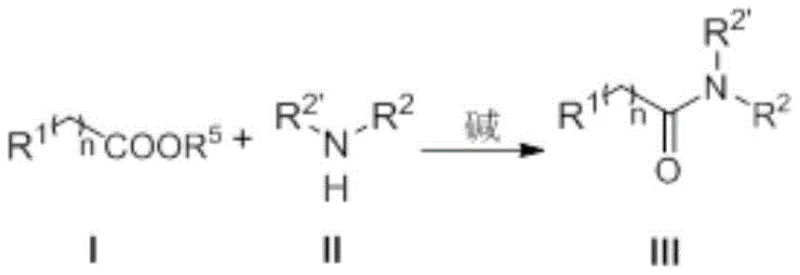
Patent CN108558692B introduces a groundbreaking methodology for synthesizing amide compounds through nitrogen heterocyclic carbene (NHC) catalysis, addressing critical limitations in traditional amide bond formation essential for approximately 25% of pharmaceutical molecules. This innovation operates under exceptionally mild conditions at room temperature with reaction times ranging from 0.1 to 1.5 hours, achieving consistently high yields between 93% and 96% across diverse substrates without requiring hazardous reagents or transition metals. The process eliminates the need for activating agents like HATU or Ph3P/I2 that characterize conventional methods, thereby removing significant purification challenges associated with byproduct separation while maintaining stringent purity standards required in drug manufacturing. By leveraging organic acid esters and amines as direct precursors under inert atmosphere, this approach establishes a sustainable pathway that significantly enhances process efficiency while reducing environmental impact through the absence of irritants or toxic residues. The method's robustness across various functional groups including nitro-substituted and alkoxy-containing substrates demonstrates exceptional versatility for producing complex pharmaceutical intermediates at commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional amide synthesis predominantly relies on two approaches that present substantial operational challenges for pharmaceutical manufacturing. The first method activates carboxylic acids using reagents like Ph3P/I2 or HATU to form reactive esters before amine coupling, but suffers from prolonged reaction times exceeding several hours, incomplete conversions requiring excess reagents, and complex byproduct mixtures that necessitate extensive purification steps including multiple chromatographic separations or crystallizations. The second approach utilizes transition metal-catalyzed oxidative amination of aldehydes, which introduces significant complications through metal contamination requiring rigorous removal processes to meet pharmaceutical purity standards, narrow substrate scope limiting applicability to specific functional groups, and extended reaction durations that impede production throughput. Both conventional methodologies generate substantial waste streams containing hazardous materials that increase disposal costs and environmental compliance burdens while creating supply chain vulnerabilities through dependency on specialized reagents with variable availability. These inherent limitations result in higher manufacturing costs, extended lead times, and inconsistent product quality that compromise the reliability required for commercial pharmaceutical production.
The Novel Approach
The patented NHC-catalyzed methodology fundamentally transforms amide synthesis by establishing a direct coupling pathway between organic acid esters and amines under exceptionally mild conditions without transition metals or hazardous activators. By employing nitrogen heterocyclic carbenes as organocatalysts in combination with bases like DBU or K2CO3, the process generates highly reactive acyl azolium intermediates that facilitate rapid nucleophilic substitution with amines at room temperature within minutes rather than hours. This innovative mechanism eliminates the need for pre-activation steps while maintaining excellent functional group tolerance across diverse substrates including nitro-, methoxy-, and alkyl-substituted compounds as demonstrated in multiple implementation examples yielding products at 93-96% efficiency. The simplified workup procedure involving aqueous quenching followed by standard extraction and silica gel chromatography significantly reduces processing complexity compared to conventional methods that require specialized purification techniques to remove metal residues or activation byproducts. This streamlined approach not only enhances operational safety through the absence of irritants but also establishes a more resilient supply chain by utilizing readily available starting materials and common laboratory solvents like tetrahydrofuran.
Mechanistic Insights into NHC-Catalyzed Amide Formation
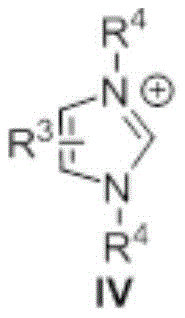
The catalytic cycle begins with the deprotonation of the azolium salt precursor by base to generate the active N-heterocyclic carbene species that subsequently reacts with organic acid esters to form key acyl azolium intermediates through nucleophilic addition followed by elimination of alkoxide. This highly electrophilic intermediate undergoes rapid nucleophilic attack by amines to form a tetrahedral adduct that collapses through proton transfer to release the final amide product while regenerating the carbene catalyst for subsequent cycles. The exceptional reactivity stems from the strong electron-donating properties of the NHC ligand that stabilize the acyl azolium intermediate while enhancing its electrophilicity toward amine nucleophiles, enabling complete conversion within minutes at ambient temperature without requiring elevated pressures or specialized equipment. This mechanism operates effectively across a broad substrate scope including sterically hindered amines and electron-deficient esters due to the catalyst's ability to modulate intermediate stability through strategic substitution patterns on the azolium core structure.
Impurity control is inherently achieved through the selective nature of the NHC-mediated mechanism which avoids common side reactions associated with traditional methods such as racemization or over-activation that generate difficult-to-separate byproducts. The absence of transition metals eliminates metal leaching concerns that typically require additional purification steps to meet pharmaceutical quality standards below parts-per-million thresholds. The mild reaction conditions prevent thermal degradation pathways that could produce colored impurities or decomposition products, while the straightforward workup procedure using standard extraction techniques effectively separates any minor byproducts without requiring specialized chromatography media or multiple recrystallization steps. This inherent selectivity combined with simplified purification protocols ensures consistent production of high-purity amide intermediates meeting stringent regulatory requirements for pharmaceutical applications without compromising yield or process efficiency.
How to Synthesize Amide Intermediates Efficiently
This patented methodology represents a significant advancement in amide synthesis technology specifically designed for pharmaceutical intermediate production where purity and process reliability are paramount concerns for R&D directors overseeing drug development pipelines. The procedure eliminates multiple pain points associated with conventional approaches including lengthy reaction times, complex purification requirements, and metal contamination risks that can derail scale-up efforts during late-stage development phases. By operating under ambient conditions with standard laboratory equipment, this method provides exceptional flexibility for process chemists seeking to optimize synthetic routes while maintaining compatibility with existing manufacturing infrastructure across global facilities.
- Combine nitrogen heterocyclic carbene catalyst (0.1-0.2 equiv) and base (0.1-0.2 equiv) in anhydrous tetrahydrofuran under nitrogen atmosphere with stirring at room temperature.
- Sequentially add organic acid ester (1.0 equiv) followed by organic amine (1.0-3.0 equiv) at 0°C, then allow reaction mixture to warm to room temperature for 0.1-1.5 hours.
- Quench reaction with water, extract product using dichloromethane or ethyl acetate, and purify crude material via silica gel column chromatography with ethyl acetate/petroleum ether (1: 5 v/v).
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this NHC-catalyzed methodology delivers substantial strategic value for procurement and supply chain decision-makers by addressing critical pain points in pharmaceutical intermediate sourcing through fundamental process improvements rather than incremental optimizations. The elimination of transition metal catalysts removes significant supply chain vulnerabilities associated with rare metal dependencies while avoiding costly purification steps required to meet regulatory metal limits in final drug substances.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and hazardous activation reagents like HATU directly reduces raw material expenses while eliminating complex metal removal processes that require specialized equipment and additional quality control testing. The simplified workup procedure using standard extraction solvents instead of multi-step purification protocols significantly lowers processing costs through reduced solvent consumption, shorter cycle times, and decreased labor requirements per batch without compromising product quality.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials including common organic solvents like tetrahydrofuran and standard bases such as DBU or K2CO3 creates a more resilient supply chain with multiple sourcing options globally. The absence of specialized reagents or hazardous materials reduces import/export restrictions and customs delays while enabling consistent production across different geographic locations without process revalidation.
- Scalability and Environmental Compliance: The ambient temperature operation and straightforward scale-up characteristics allow seamless transition from laboratory development to commercial production volumes without reoptimization, significantly reducing time-to-market for new drug candidates. The environmentally friendly profile with minimal waste generation aligns with increasing regulatory pressure for sustainable manufacturing practices while reducing disposal costs associated with hazardous byproducts from conventional methods.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by procurement teams evaluating this patented technology for integration into their pharmaceutical intermediate supply chains, focusing specifically on implementation challenges and quality assurance protocols relevant to commercial manufacturing environments.
Q: How does NHC catalysis overcome limitations of conventional amide synthesis methods?
A: Unlike traditional methods requiring harsh reagents like Ph3P/I2 or transition metals that cause lengthy reaction times and difficult byproduct separation, this NHC-catalyzed process operates under mild conditions (room temperature, 0.1-1.5 hours) with no metal residues. The catalyst forms highly reactive intermediates that enable rapid nucleophilic substitution between esters and amines, eliminating the need for activating agents while maintaining high substrate adaptability across diverse functional groups.
Q: What supply chain advantages does this method offer for pharmaceutical manufacturing?
A: The process eliminates transition metal catalysts that require complex purification steps, significantly reducing raw material dependencies and potential supply bottlenecks. Using readily available organic solvents like tetrahydrofuran and common bases such as DBU or K2CO3 ensures consistent material sourcing. The short reaction time and simplified workup enable faster batch turnover while maintaining stringent purity specifications required for pharmaceutical intermediates.
Q: How does this technology support sustainable scale-up for commercial production?
A: The ambient temperature operation and absence of hazardous reagents reduce energy consumption and waste generation compared to conventional methods. The consistent high yields (93-96% across multiple substrates) and straightforward purification via silica gel chromatography facilitate seamless scale-up from laboratory to industrial scale without reoptimization. This environmentally friendly approach aligns with green chemistry principles while maintaining economic viability through reduced processing complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Intermediate Supplier
Our patented NHC-catalyzed amide synthesis technology represents a paradigm shift in producing high-value pharmaceutical intermediates where purity consistency and process reliability are non-negotiable requirements for global drug manufacturers. As an established CDMO leader with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we combine deep expertise in organocatalysis with state-of-the-art manufacturing capabilities to deliver stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation for comprehensive impurity profiling.
Leverage our technical procurement team's expertise to develop a Customized Cost-Saving Analysis tailored to your specific amide intermediate requirements - contact us today to request detailed COA data and route feasibility assessments demonstrating how this innovative methodology can enhance your supply chain resilience while maintaining uncompromising quality standards.
