Advanced Commercial Synthesis of Everolimus Intermediates via Green Ethylene Carbonate Chemistry
Advanced Commercial Synthesis of Everolimus Intermediates via Green Ethylene Carbonate Chemistry
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the production of complex macrolide immunosuppressants. Patent CN113929702A introduces a transformative preparation method for Everolimus, a critical mTOR inhibitor used in oncology and transplant medicine. This innovation addresses long-standing challenges in the functionalization of the rapamycin scaffold, specifically targeting the 42-position hydroxyl group. By leveraging ethylene carbonate as a safe and efficient alkylating agent, this technology bypasses the hazardous reagents and low-yielding steps characteristic of legacy processes. For global procurement teams and R&D directors, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity standards.
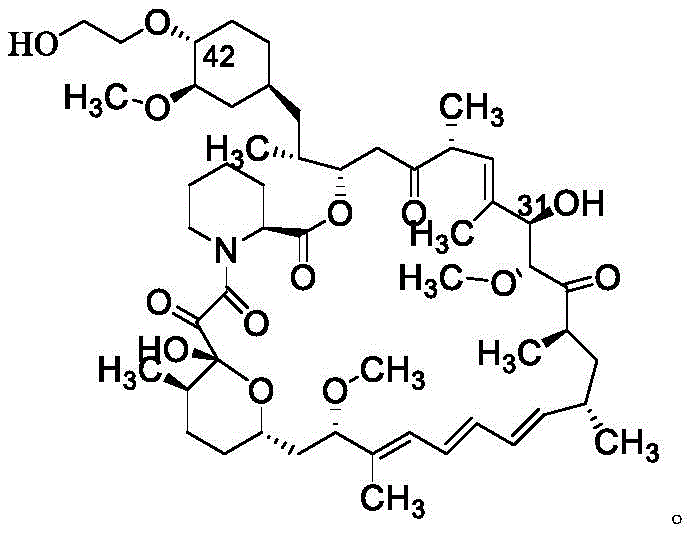
Everolimus, chemically defined as 42-O-(2-hydroxyethyl)-rapamycin, requires precise regioselective modification of the parent sirolimus molecule. The structural complexity of the macrocyclic lactone ring demands synthetic strategies that preserve the integrity of sensitive functional groups while introducing the hydroxyethyl side chain. Traditional approaches have often struggled with selectivity issues between the 31-position and 42-position hydroxyls, leading to complex mixtures that are difficult to separate. The methodology described in CN113929702A utilizes a pre-protected sirolimus starting material, ensuring that the reaction occurs exclusively at the desired 42-position, thereby streamlining the purification workflow and enhancing overall process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Everolimus has been plagued by significant technical hurdles that impact both cost and safety. Early methods, such as those described in US Patent 5665772, relied on the reaction of rapamycin with 2-(tert-butyldimethylsilyloxy)ethyl trifluoromethanesulfonate. This approach suffered from abysmal selectivity, with reported yields ranging merely from 5% to 15%, meaning the vast majority of the expensive starting material was wasted. Furthermore, subsequent deprotection steps often led to degradation products and isomerization, resulting in a final product with insufficient purity for pharmaceutical applications.
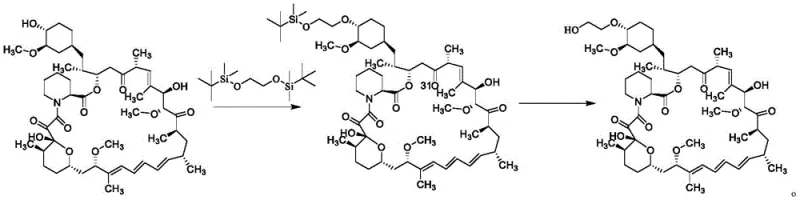
Alternative strategies attempted to mitigate selectivity issues by employing ethylene oxide under strong acid catalysis or using complex protected glycol triflates. However, the use of ethylene oxide introduces severe safety risks due to its explosive nature and toxicity, requiring specialized containment infrastructure that drives up capital expenditure. Additionally, processes involving triflate leaving groups generate stoichiometric amounts of sulfonate waste, creating environmental disposal challenges. These conventional routes are characterized by multi-step protection and deprotection sequences that increase the total processing time and reduce the overall throughput of the manufacturing facility.
The Novel Approach
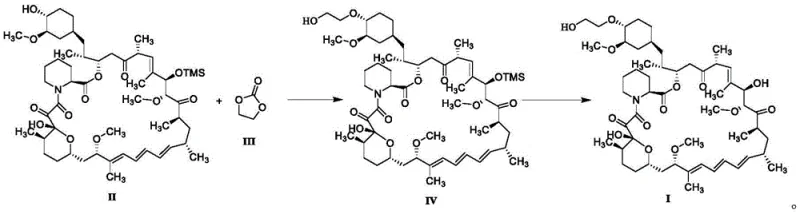
The breakthrough detailed in CN113929702A replaces hazardous alkylating agents with ethylene carbonate, a stable and non-toxic reagent. In this novel pathway, 31-O-protected sirolimus reacts with ethylene carbonate in the presence of a mild base such as potassium carbonate. This reaction proceeds through a nucleophilic attack on the cyclic carbonate, resulting in ring opening and the formation of the desired hydroxyethyl ether linkage. Crucially, the only byproduct of this transformation is carbon dioxide, which evolves as a gas, driving the reaction equilibrium forward and simplifying the workup procedure significantly.
This new approach offers a dramatic improvement in operational simplicity and safety profile. By avoiding strong acids during the alkylation step and eliminating the need for gaseous ethylene oxide, the process becomes amenable to standard stainless steel reactor setups without the need for exotic metallurgy or high-pressure containment. The use of common organic solvents like toluene further enhances the economic viability of the process. Experimental data from the patent indicates that this method can achieve yields as high as 98.8% for the intermediate, a stark contrast to the single-digit yields of previous generations, fundamentally altering the cost structure of Everolimus production.
Mechanistic Insights into Ethylene Carbonate Alkylation
The core of this technological advancement lies in the mechanistic efficiency of the carbonate ring-opening reaction. Under basic conditions, typically utilizing potassium carbonate at temperatures between 80°C and 100°C, the 42-hydroxyl group of the protected sirolimus acts as a nucleophile. It attacks the methylene carbon adjacent to the carbonyl group of the ethylene carbonate. This attack breaks the C-O bond of the carbonate ring, releasing a carboxylate anion which subsequently decarboxylates to form carbon dioxide. This mechanism ensures that the hydroxyethyl group is installed cleanly without introducing extraneous atoms or complex protecting groups that would require removal later.
Impurity control is inherently built into this mechanism. Because the reaction is highly regioselective for the 42-position due to the steric and electronic environment of the macrocycle, the formation of 31-substituted isomers is minimized. Furthermore, the mild basic conditions prevent the acid-catalyzed degradation of the sensitive triene system within the macrolide ring, a common failure mode in strong acid-catalyzed ethylene oxide additions. The result is a crude reaction mixture with a purity profile that facilitates straightforward crystallization or chromatography, ensuring that the final Active Pharmaceutical Ingredient (API) meets stringent regulatory specifications for impurities.
How to Synthesize Everolimus Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the dissolution of the 31-O-TMS protected sirolimus in a solvent such as toluene, followed by the addition of the base and ethylene carbonate. The mixture is heated to promote the reaction, after which standard aqueous workup procedures are employed to isolate the intermediate. The final step involves a mild acidic deprotection to remove the silyl group, yielding the target molecule. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Dissolve 31-O-TMS protected sirolimus (Compound II) in toluene and add potassium carbonate as the base.
- Add ethylene carbonate and heat the mixture to 80-100°C to facilitate nucleophilic attack and ring opening.
- Perform acidic deprotection using dilute hydrochloric acid in tetrahydrofuran to remove the silyl group and yield Everolimus.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ethylene carbonate-based route offers compelling strategic advantages beyond mere technical feasibility. The shift away from hazardous reagents like ethylene oxide and complex triflates directly translates to reduced regulatory compliance costs and lower insurance premiums associated with chemical storage and handling. The simplified workflow, which eliminates multiple protection and deprotection stages, shortens the overall cycle time per batch, allowing manufacturing facilities to increase their annual output capacity without significant capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic improvement in raw material utilization. With yields exceeding 98% in optimized examples, the consumption of the expensive sirolimus starting material is minimized, directly lowering the Cost of Goods Sold (COGS). Additionally, the replacement of exotic reagents with commodity chemicals like ethylene carbonate and potassium carbonate reduces the direct material spend. The elimination of toxic sulfonate waste also lowers waste disposal costs, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Reliance on ethylene oxide often subjects manufacturers to supply volatility due to its classification as a high-risk chemical with strict transportation regulations. By switching to ethylene carbonate, a stable and widely available industrial chemical, the supply chain becomes more resilient and less prone to disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch performance and reliable delivery schedules to downstream customers.
- Scalability and Environmental Compliance: The green chemistry attributes of this method align perfectly with modern ESG (Environmental, Social, and Governance) goals. The generation of carbon dioxide as the sole byproduct eliminates the need for complex wastewater treatment systems required for sulfonate or halide waste streams. This environmental friendliness facilitates easier permitting for plant expansions and ensures long-term operational continuity in regions with tightening environmental regulations. The process is inherently scalable from kilogram to multi-ton production levels using standard batch reactor technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Everolimus synthesis route. These insights are derived directly from the experimental data and technical disclosures within patent CN113929702A, providing a factual basis for decision-making regarding technology transfer and process adoption.
Q: Why is ethylene carbonate preferred over ethylene oxide for Everolimus synthesis?
A: Ethylene carbonate is a stable solid or liquid at room temperature, eliminating the severe explosion hazards and handling difficulties associated with gaseous ethylene oxide. Furthermore, the byproduct is carbon dioxide, making the process cleaner and easier to purify compared to triflate-based methods.
Q: What is the expected purity of Everolimus using this novel route?
A: According to patent data, this method achieves exceptionally high purity, with HPLC results showing up to 99.94% for the intermediate and 99.88% for the final API, significantly reducing the burden on downstream purification steps.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common solvents like toluene and inexpensive bases like potassium carbonate. The mild reaction conditions (80-100°C) and high yields (over 98% in optimized examples) make it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Everolimus Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the ethylene carbonate route are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Everolimus intermediate meets the highest global pharmacopeial standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized synthesis platform can drive value and reliability for your Everolimus projects.
