Scalable Synthesis of Indoloazepine Seven-Membered Rings via Monovalent Silver Catalysis
Introduction to Advanced Indoloazepine Synthesis
The synthesis of indoloazepine seven-membered rings represents a critical challenge in modern medicinal chemistry, given their prevalence in bioactive natural products and alkaloid frameworks. Patent CN109384789B introduces a groundbreaking methodology that leverages monovalent silver catalysis to construct these complex heterocyclic scaffolds under exceptionally mild conditions. This innovation addresses long-standing inefficiencies in the field by enabling the direct intramolecular cyclization of indole-containing tryptamine alkynamide substrates at room temperature. By utilizing accessible silver salts such as silver trifluoromethanesulfonate in a toluene medium, this process eliminates the need for hazardous reagents and energy-intensive heating protocols. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology offers a robust pathway to high-value scaffolds with superior atom economy and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indoloazepine core has been plagued by cumbersome multi-step sequences and harsh reaction environments that hinder efficient manufacturing. Early methodologies, such as the ring expansion strategy reported by Stephen P. Waters, necessitated vigorous reflux conditions and employed solvents with significant toxicity profiles, creating substantial barriers to safe scale-up. Similarly, approaches involving intramolecular aminolysis often relied on corrosive strong acids and sulfur-containing compounds, which complicated purification and generated difficult-to-manage waste streams. Furthermore, radical-mediated pathways typically suffered from poor atom economy and required the use of expensive initiators like azobisisobutyronitrile, leading to lower overall yields and higher production costs. These legacy techniques collectively impose severe constraints on cost reduction in API manufacturing due to their high energy consumption and complex downstream processing requirements.
The Novel Approach
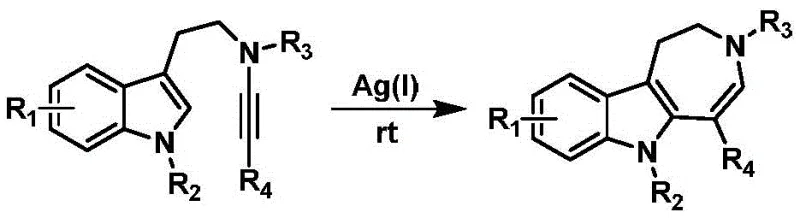
In stark contrast, the monovalent silver-catalyzed protocol described in the patent revolutionizes this synthetic landscape by enabling a direct, one-step cyclization at ambient temperatures. This novel approach utilizes the unique ability of silver(I) ions to activate alkyne triple bonds, facilitating a smooth intramolecular attack by the nucleophilic indole moiety without the need for external heating or aggressive activators. The reaction proceeds efficiently in toluene, a solvent favored for its industrial availability and ease of recovery, thereby streamlining the entire workflow. By bypassing the need for pre-functionalization or protective group manipulations common in older routes, this method drastically reduces the number of unit operations. This simplicity translates directly into enhanced process reliability and makes the technology an ideal candidate for the commercial scale-up of complex heterocycles in a GMP environment.
Mechanistic Insights into Ag(I)-Catalyzed Cyclization
The core of this technological advancement lies in the precise activation of the alkyne functionality by the monovalent silver cation. Upon coordination with the silver catalyst, the electron density of the triple bond is significantly polarized, rendering the beta-carbon highly electrophilic and susceptible to nucleophilic attack. In the context of the tryptamine alkynamide substrate, the proximal indole nitrogen or carbon center acts as the internal nucleophile, initiating a cascade that closes the seven-membered ring. This mechanism avoids the high-energy transition states associated with thermal cyclizations, allowing the reaction to proceed rapidly even at 0-50°C. The choice of counterion in the silver salt, such as triflate or hexafluoroantimonate, plays a crucial role in modulating the Lewis acidity of the metal center, ensuring optimal turnover rates without promoting decomposition of the sensitive indole core.
From an impurity control perspective, the mildness of the silver-catalyzed system is paramount for maintaining product integrity. Traditional acidic or radical conditions often lead to polymerization of the alkyne or degradation of the indole ring, generating complex impurity profiles that are difficult to separate. By operating at room temperature with a selective Lewis acid catalyst, side reactions such as over-alkylation or oxidative degradation are effectively suppressed. The use of neutral alumina for final purification further ensures that acid-sensitive functionalities remain intact. This high level of chemoselectivity is essential for producing high-purity indoloazepine derivatives that meet the stringent specifications required for downstream pharmaceutical applications, minimizing the risk of genotoxic impurities.
How to Synthesize Indoloazepine Efficiently
The practical implementation of this synthesis is designed for straightforward execution in standard laboratory or pilot plant settings. The process begins with the dissolution of the tryptamine alkynamide substrate in toluene, followed by the addition of a catalytic amount of the silver salt. The mixture is simply stirred at room temperature, with reaction progress monitored via thin-layer chromatography using standard hexane and ethyl acetate systems. Upon completion, the solvent is removed under reduced pressure, and the crude product is purified directly by column chromatography. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Charge the indole-containing tryptamine alkynamide substrate into a reaction vessel and dissolve in toluene solvent.
- Add the monovalent silver catalyst (e.g., AgOTf, 5-10 mol%) to the solution and stir at room temperature (0-50°C) for 3-10 hours.
- Remove the solvent via rotary evaporation and purify the crude mixture directly by column chromatography on neutral alumina to isolate the target indoloazepine.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this silver-catalyzed methodology offers transformative benefits regarding cost structure and logistical stability. The elimination of extreme thermal conditions and hazardous reagents significantly lowers the barrier to entry for manufacturing, reducing both capital expenditure on specialized equipment and operational expenditure on energy and safety compliance. The reliance on commercially available silver salts and common solvents like toluene ensures a stable supply chain that is less vulnerable to the fluctuations often seen with exotic ligands or custom reagents. This robustness allows for more accurate forecasting and reducing lead time for high-purity intermediates, ensuring that production schedules are met consistently without unexpected delays caused by reagent shortages or process failures.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the drastic simplification of the synthetic route, which removes multiple steps associated with protection and deprotection strategies found in conventional methods. By utilizing inexpensive monovalent silver catalysts that function effectively at low loadings, the direct material costs are kept minimal compared to precious metal alternatives like palladium or rhodium. Furthermore, the ability to run the reaction at room temperature eliminates the energy costs associated with prolonged heating or cryogenic cooling, contributing to substantial overall cost savings. The streamlined workup procedure, which avoids complex aqueous extractions or neutralization steps, further reduces labor and waste disposal expenses, enhancing the overall profit margin for the final active ingredient.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including the tryptamine derivatives and sulfonyl protecting groups, are commodity chemicals with well-established global supply networks. This accessibility mitigates the risk of supply disruptions that often plague processes dependent on bespoke or single-source reagents. Additionally, the tolerance of the reaction to various substituents on the indole and alkyne moieties means that a single platform technology can be adapted to produce a wide library of analogues without re-optimizing the entire process. This flexibility allows manufacturers to respond rapidly to changing market demands or clinical trial requirements, ensuring a continuous and reliable flow of materials to downstream partners.
- Scalability and Environmental Compliance: From an environmental standpoint, the high atom economy of this cyclization reaction aligns perfectly with green chemistry principles, minimizing the generation of stoichiometric by-products. The use of toluene, while requiring proper handling, is a solvent with established recovery and recycling protocols in the fine chemical industry, reducing the environmental footprint of the manufacturing process. The absence of heavy metal contaminants that are difficult to remove, combined with the mild reaction conditions, simplifies the regulatory approval process for new drug applications. This ease of scale-up from gram to kilogram quantities ensures that the transition from R&D to commercial production is seamless, supporting long-term sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this silver-catalyzed synthesis. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing clarity on substrate compatibility and operational parameters. Understanding these details is crucial for project managers evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the advantages of the silver-catalyzed method over traditional ring expansion?
A: Unlike traditional methods that require harsh reflux conditions and toxic solvents, the silver-catalyzed protocol operates at room temperature in toluene, significantly improving safety and atom economy while simplifying post-processing.
Q: Which silver catalysts are suitable for this transformation?
A: The patent specifies several effective monovalent silver salts, including silver trifluoromethanesulfonate (AgOTf), silver hexafluoroantimonate, silver tetrafluoroborate, and silver bis-trifluoromethanesulfonimide.
Q: Is this method scalable for industrial production?
A: Yes, the method utilizes readily available raw materials, inexpensive catalysts, and mild reaction conditions without the need for extreme temperatures or pressures, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoloazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of indoloazepine intermediate meets the highest quality standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of seven-membered ring formation with confidence and consistency.
We invite you to collaborate with us to leverage this efficient synthesis for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall cost of goods sold.
