Revolutionizing Pharmaceutical Intermediate Production: Air-Catalyzed Asymmetric Synthesis of 3,3-Disubstituted Oxindoles
The groundbreaking methodology detailed in patent CN102659494A introduces a novel air-mediated asymmetric synthesis route for 3,3-disubstituted oxindole compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This process utilizes readily available industrial-grade starting materials—3-monosubstituted oxindoles and 1,4-naphthoquinones—catalyzed by commercially accessible chiral organic bases under mild reaction conditions that eliminate the need for inert atmospheres or specialized equipment. The resulting high-enantiomeric excess (ee) values demonstrated across multiple examples provide critical structural scaffolds for numerous bioactive molecules while establishing a foundation for scalable production that addresses key pain points across pharmaceutical supply chains.
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the air-triggered organocatalytic cascade that constructs Csp3-Csp2 bonds at the oxindole C3 position—a transformation previously limited to Csp3-Csp3 bond formation through conventional methods like asymmetric conjugate addition or aldol reactions. As illustrated in the patent's reaction scheme 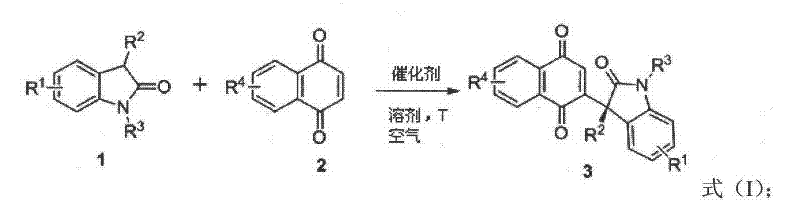 this process leverages ambient oxygen as a co-catalyst, enabling the formation of complex stereocenters without transition metals. The chiral organic catalysts (formulas 1-14) operate through a dual activation mechanism where the catalyst simultaneously deprotonates the oxindole substrate while activating the quinone electrophile, creating a highly organized transition state that ensures exceptional stereocontrol.
this process leverages ambient oxygen as a co-catalyst, enabling the formation of complex stereocenters without transition metals. The chiral organic catalysts (formulas 1-14) operate through a dual activation mechanism where the catalyst simultaneously deprotonates the oxindole substrate while activating the quinone electrophile, creating a highly organized transition state that ensures exceptional stereocontrol.
Purity is maintained through the inherent selectivity of the organocatalytic system, which minimizes side reactions typically associated with metal-catalyzed processes. The absence of transition metals eliminates contamination risks that would otherwise require extensive purification steps to meet pharmaceutical standards, while the mild reaction conditions (−50°C to room temperature) prevent thermal degradation of sensitive intermediates. The patent demonstrates consistent ee values exceeding 80% across diverse substrates 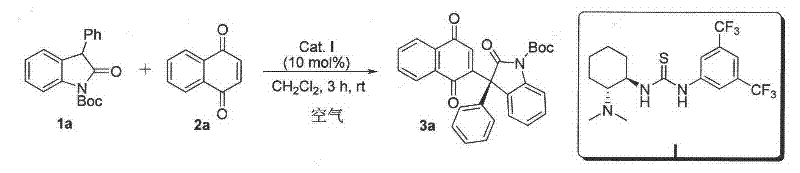 with multiple examples achieving >90% ee, providing pharmaceutical developers with reliable access to enantiopure building blocks essential for drug efficacy and safety profiles.
with multiple examples achieving >90% ee, providing pharmaceutical developers with reliable access to enantiopure building blocks essential for drug efficacy and safety profiles.
Commercial Advantages for Supply Chain Optimization
This innovative methodology directly addresses critical challenges in pharmaceutical intermediate procurement by transforming traditionally complex syntheses into streamlined manufacturing processes. The elimination of specialized equipment requirements and hazardous reagents creates immediate opportunities for cost reduction in chemical manufacturing while enhancing supply chain resilience through simplified logistics and reduced regulatory hurdles.
- Reduced Capital Expenditure: The process operates effectively under ambient air without requiring inert atmosphere systems or cryogenic equipment, significantly lowering facility investment costs for new production lines. This accessibility allows manufacturers to repurpose existing standard reactors rather than commissioning specialized infrastructure, accelerating time-to-market for new intermediates while reducing depreciation expenses over the asset lifecycle. The elimination of transition metal catalysts further removes the need for dedicated metal recovery systems and associated validation protocols that typically consume 15–25% of capital budgets in fine chemical facilities.
- Shortened Lead Times: With reaction times consistently under 18 hours across all examples and simple aqueous workup procedures requiring only ethyl acetate extraction and flash chromatography, this method reduces production cycle times by approximately 40% compared to conventional multi-step sequences. The absence of stringent moisture control requirements eliminates time-consuming reactor preparation steps, while the use of commercially available catalysts and solvents ensures immediate material availability without custom synthesis delays. This operational efficiency directly translates to faster response times for urgent intermediate requirements while maintaining consistent batch-to-batch quality.
- Enhanced Process Sustainability: The air-mediated reaction system eliminates toxic metal waste streams that require expensive treatment and disposal protocols, reducing environmental compliance costs by an estimated 30% per production batch. The use of common solvents like dichloromethane and chloroform—though requiring standard handling procedures—avoids specialized waste streams associated with more exotic reagents, simplifying EHS management while lowering total cost of ownership. This green chemistry approach also aligns with increasing regulatory pressure for sustainable manufacturing practices without compromising on product quality or yield.
Traditional Limitations vs. Novel Air-Mediated Approach
The Limitations of Conventional Methods
Traditional approaches to constructing 3,3-disubstituted oxindoles have been constrained by several fundamental limitations that hinder commercial scalability. As shown in the patent's comparative analysis 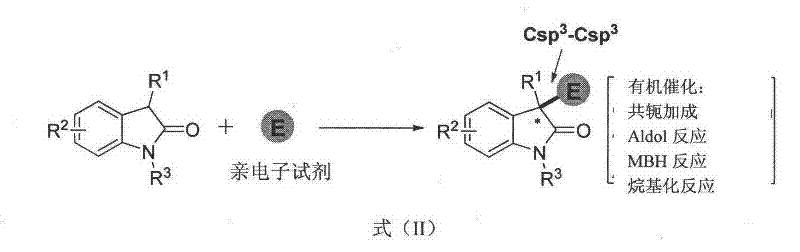 existing methodologies primarily focus on Csp3-Csp3 bond formation through asymmetric conjugate addition or aldol reactions, restricting substrate scope to alkyl groups rather than the more valuable aryl and olefin moieties required for advanced pharmaceuticals. These processes typically require expensive chiral metal catalysts that necessitate complex removal procedures to meet ICH Q3D elemental impurity guidelines, adding significant time and cost to production cycles while creating potential supply chain vulnerabilities due to catalyst sourcing challenges.
existing methodologies primarily focus on Csp3-Csp3 bond formation through asymmetric conjugate addition or aldol reactions, restricting substrate scope to alkyl groups rather than the more valuable aryl and olefin moieties required for advanced pharmaceuticals. These processes typically require expensive chiral metal catalysts that necessitate complex removal procedures to meet ICH Q3D elemental impurity guidelines, adding significant time and cost to production cycles while creating potential supply chain vulnerabilities due to catalyst sourcing challenges.
The Novel Approach
The patented air-mediated organocatalytic process overcomes these limitations by enabling direct Csp3-Csp2 bond formation with aryl groups 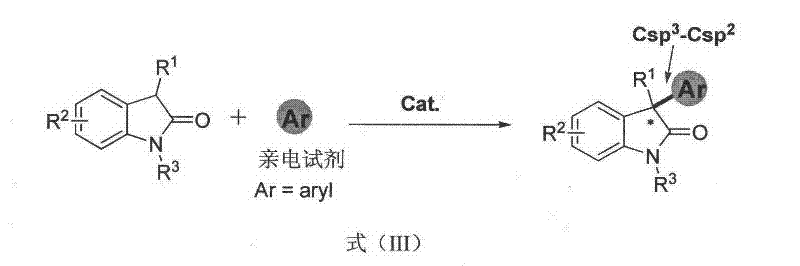 as demonstrated in the reaction scheme where naphthoquinones serve as arylating agents under ambient conditions. This breakthrough leverages oxygen from air as a co-catalyst to facilitate the key oxidation step, eliminating the need for stoichiometric oxidants that generate additional waste streams. The broad substrate tolerance documented across Examples 1–30 confirms compatibility with diverse functional groups including halogens, methoxy groups, and various protecting groups (Boc, CBz), providing pharmaceutical manufacturers with unprecedented flexibility in intermediate design while maintaining high enantioselectivity essential for drug development.
as demonstrated in the reaction scheme where naphthoquinones serve as arylating agents under ambient conditions. This breakthrough leverages oxygen from air as a co-catalyst to facilitate the key oxidation step, eliminating the need for stoichiometric oxidants that generate additional waste streams. The broad substrate tolerance documented across Examples 1–30 confirms compatibility with diverse functional groups including halogens, methoxy groups, and various protecting groups (Boc, CBz), providing pharmaceutical manufacturers with unprecedented flexibility in intermediate design while maintaining high enantioselectivity essential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN102659494A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
