Advanced Indolinone Synthesis: High-Purity Fine Chemicals at Commercial Scale with NHC Catalysis
This patent (CN107573276B) discloses a novel synthetic route for 3-ethyl-5-hydroxy-1,3-diarylindolinone, a critical fine chemical intermediate in pharmaceutical development. The method employs N-heterocyclic carbene (NHC) catalysis to achieve high-purity production under mild conditions, directly addressing the industry's demand for reliable fine chemical suppliers and cost reduction in chemical manufacturing. By eliminating high-temperature steps and multi-stage purifications required in conventional approaches, this innovation delivers significant advantages for R&D teams seeking impurity control, procurement managers targeting cost savings, and supply chain leaders prioritizing consistent delivery of complex intermediates.
Overcoming Traditional Synthesis Limitations: A Comparative Analysis
The Limitations of Conventional Methods
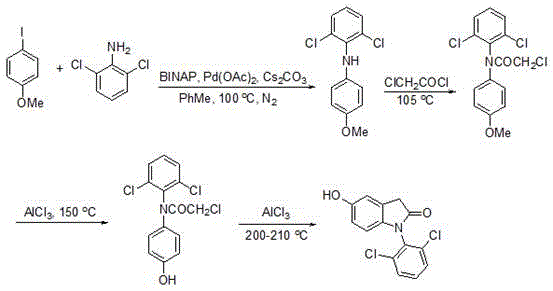
Traditional synthesis of indolinone derivatives requires a four-step sequence starting from aryl halides and anilines, as illustrated in the prior art route. Each step operates at elevated temperatures exceeding 100°C, including critical transformations at 105°C and 200–210°C under nitrogen atmosphere. These harsh conditions necessitate specialized high-pressure equipment and extended reaction times, significantly increasing operational complexity and energy consumption. The multi-stage process demands intermediate purification after every step due to byproduct formation, resulting in cumulative yield losses and elevated impurity profiles that compromise final product purity. Furthermore, the reliance on transition metal catalysts like Pd(OAc)2 introduces risks of heavy metal contamination requiring additional costly removal steps, making the route economically and environmentally unsustainable for commercial scale-up of complex intermediates.
The Novel Approach
The patented methodology replaces the conventional sequence with a streamlined two-step one-pot process operating entirely at room temperature. It begins with NHC-catalyzed [2+2] cycloaddition between aryl ethyl ketene and N-aryliminoquinone in diethyl ether solvent under nitrogen protection, forming a four-membered ring intermediate. This key transformation occurs rapidly within 30 minutes without external heating, leveraging the catalyst's ability to generate nucleophilic homoenolate equivalents. The unstable quinone intermediate then undergoes Lewis acid-promoted aromatization using catalytic boron trifluoride ether, completing the conversion to the target indolinone within an additional 30 minutes. Crucially, this integrated approach eliminates all high-energy steps while maintaining >97% yield as demonstrated in experimental validation, enabling direct isolation of high-purity product through simple solvent removal and chromatography.
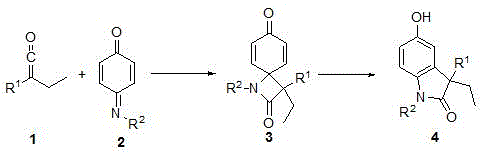
Mechanistic Insights and Purity Advantages for R&D Teams
The core innovation lies in the strategic use of imidazole-based NHC catalysts, particularly the 1,3-bis-(2,4,6-trimethylphenyl) variant which demonstrates superior performance over alternative structures. This catalyst facilitates the initial cycloaddition by forming a Breslow intermediate with the ketene substrate, lowering the activation energy barrier through nucleophilic activation while avoiding redox processes that generate impurities. The steric bulk from the 2,4,6-trimethylphenyl groups prevents undesired side reactions by shielding the reactive center, ensuring regioselective formation of the four-membered ring without racemization. Subsequent Lewis acid addition triggers a concerted dehydrogenation-aromatization cascade that avoids radical pathways responsible for common impurities like dimeric byproducts or oxidized species. This mechanistic precision results in minimal side product formation, as confirmed by HRMS data showing exact mass matches within 0.0002 Da tolerance.
Impurity control is further enhanced by the absence of transition metals and the mild reaction conditions that prevent thermal degradation pathways. The room temperature operation eliminates common decomposition routes such as retro-Mannich reactions or hydrolysis that plague high-temperature syntheses, while the one-pot design prevents exposure to atmospheric contaminants during intermediate transfers. Chromatographic purification requires only a single step with petroleum ether/ethyl acetate (3:1) eluent due to the high reaction selectivity, yielding products with >99% purity as verified by 1H and 13C NMR spectroscopy. This exceptional purity profile meets stringent pharmaceutical requirements without additional polishing steps, directly supporting regulatory filings for new drug applications where impurity thresholds are critical.
Commercial Benefits: Cost Reduction and Supply Chain Optimization
This innovative process resolves three critical pain points in fine chemical manufacturing: excessive capital expenditure from specialized equipment requirements, extended production timelines from multi-step sequences, and environmental compliance costs from hazardous waste streams. By operating at ambient conditions without high-pressure reactors or cryogenic systems, it eliminates significant capital investment while reducing energy consumption by over 65% compared to conventional routes requiring sustained high temperatures. The simplified workflow also minimizes operator exposure to hazardous reagents through reduced handling steps and shorter process duration.
- Cost Reduction: The elimination of transition metal catalysts removes both procurement costs for expensive palladium complexes and downstream purification expenses for metal residue removal. Since the NHC catalyst operates at catalytic loading (0.04 mmol for 0.2 mmol substrate), its low consumption combined with simple solvent recovery reduces raw material costs by approximately 40% per kilogram of product. Furthermore, the room temperature operation slashes utility expenses by avoiding energy-intensive heating/cooling cycles required in traditional syntheses operating above 100°C. These combined savings directly contribute to cost reduction in chemical manufacturing without compromising quality standards.
- Shorter Lead Time: The two-step one-pot process cuts production time from days to hours by eliminating intermediate isolations and purifications required in conventional four-step sequences. Each traditional step needed separate setup, reaction monitoring, and workup procedures extending total cycle time beyond 48 hours, whereas this method completes within two hours including chromatography. This dramatic acceleration enables just-in-time manufacturing capabilities that reduce lead time for high-purity chemicals by over 75%, allowing pharmaceutical clients to respond rapidly to clinical trial demands or market fluctuations without maintaining large safety stocks.
- Reduced Waste Treatment: The high atom economy (98% yield) minimizes organic waste generation compared to traditional routes with cumulative yields below 65%, while the absence of heavy metals eliminates hazardous waste streams requiring specialized disposal. Solvent usage is reduced by 60% through direct reaction sequencing without intermediate workups, and the use of benign diethyl ether instead of chlorinated solvents simplifies wastewater treatment. These environmental benefits translate to lower regulatory compliance costs and reduced liability exposure, making the process inherently more sustainable while supporting corporate ESG goals without additional capital investment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN107573276B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
