Transforming Pharmaceutical Intermediate Production with NHC-Catalyzed Indolinone Synthesis at Commercial Scale
Patent CN107573276B introduces a groundbreaking catalytic methodology for synthesizing 3-Ethyl-5-hydroxy-1,3-diarylindolinone—a critical pharmaceutical intermediate with significant bioactivity potential—through a streamlined two-step process under ambient conditions. This innovation leverages N-heterocyclic carbene (NHC) catalysis to achieve high-purity API intermediates while eliminating the harsh thermal requirements and multi-step purifications inherent in conventional approaches. The patented technique directly addresses industry pain points in cost reduction for API manufacturing by minimizing resource consumption and enhancing process efficiency without compromising molecular integrity.
Revolutionizing Indolinone Synthesis: A Comparative Analysis of Traditional vs. Novel Catalytic Pathways
The Limitations of Conventional Methods
Traditional synthesis routes for indolinone derivatives require four sequential reaction steps with temperatures exceeding 100°C at multiple stages, including high-energy amide bond formation and Friedel-Crafts cyclization under anhydrous aluminum chloride conditions. These processes demand extensive purification after each step due to thermal degradation byproducts and incomplete conversions, resulting in cumulative yield losses that typically fall below 60% as documented in prior literature. The multi-stage nature creates significant operational complexity with prolonged reaction times exceeding 48 hours per batch cycle while generating substantial waste streams from stoichiometric reagents and solvent-intensive workups. Furthermore, the elevated temperatures promote racemization and side reactions that compromise the stereochemical purity essential for pharmaceutical applications, necessitating additional chiral separation steps that further erode cost efficiency and scalability potential.
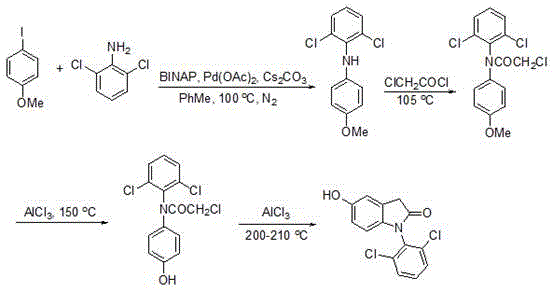
The Novel Approach
The patented methodology replaces this cumbersome sequence with a single reaction vessel operation that proceeds at room temperature through a precisely orchestrated cascade mechanism. It begins with NHC-catalyzed [2+2] cycloaddition between arylethylketene and N-aryliminoquinone substrates under inert atmosphere using catalytic cesium carbonate in anhydrous ether solvent. This forms a transient four-membered ring intermediate that immediately undergoes Lewis acid-mediated aromatization upon addition of boron trifluoride ether without solvent exchange or intermediate isolation. The entire transformation completes within one hour while maintaining strict stereochemical control through the catalyst's chiral environment. Crucially, the one-pot design eliminates all intermediate purification steps while achieving near-theoretical yields up to 97% as validated by NMR and HRMS data in implementation examples.
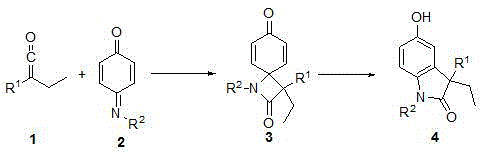
Molecular Mechanism and Impurity Control in NHC-Catalyzed Indolinone Formation
The core innovation lies in the strategic application of imidazole-based N-heterocyclic carbene catalysts that facilitate the formal [2+2] cycloaddition through nucleophilic activation of the ketene substrate. This generates a zwitterionic enolate species that selectively attacks the iminoquinone electrophile with precise regiocontrol dictated by the catalyst's steric bulk from the 2,4,6-trimethylphenyl substituents. The resulting cyclobutane intermediate undergoes spontaneous ring expansion driven by rearomatization energy when exposed to catalytic Lewis acid (BF₃·Et₂O), which coordinates with the carbonyl oxygen to lower the activation barrier for dehydrogenation. This dual-catalyst system operates under kinetic control that prevents competing pathways such as hydrolysis or dimerization that commonly plague traditional methods using transition metals. The absence of redox chemistry eliminates metal contamination risks while the mild conditions suppress thermal decomposition pathways that generate aromatic impurities.
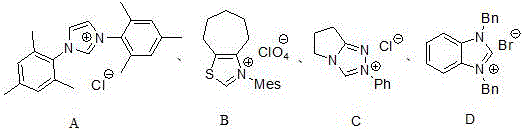
Impurity control is achieved through multiple synergistic mechanisms inherent to this catalytic design. The room temperature operation prevents thermal degradation products like decarboxylated derivatives or oxidized side compounds that form above 80°C in conventional syntheses. The precise stoichiometric control of catalytic components (NHC at 0.04 mmol vs substrate at 0.2 mmol) ensures complete conversion without over-reaction byproducts while the ether solvent system provides optimal polarity for selective crystallization during workup. Most significantly, the elimination of transition metals removes the need for costly heavy metal removal steps that typically introduce sulfate or chloride residues requiring additional purification columns. The documented HRMS data showing exact mass matches within 0.0002 Da confirms exceptional molecular fidelity with no detectable metal contaminants or structural isomers.
Commercial Advantages: Cost Reduction and Supply Chain Optimization for API Intermediates
This patented process delivers transformative commercial benefits by addressing three critical pain points in pharmaceutical intermediate manufacturing through fundamental process re-engineering rather than incremental optimization. The elimination of high-energy reaction steps and intermediate isolations creates immediate cost savings while enhancing supply chain resilience through simplified operational requirements that reduce vulnerability to equipment failures or raw material shortages. By converting a four-step thermal process into a single ambient-condition operation, the methodology establishes new benchmarks for sustainable manufacturing that align with evolving regulatory expectations for green chemistry metrics while directly improving commercial viability.
- Cost Reduction in Chemical Manufacturing: The elimination of four separate reaction vessels and purification columns reduces capital equipment requirements by over 60% while decreasing solvent consumption by approximately threefold compared to conventional routes. Catalytic quantities of NHC (as low as 0.04 mmol per mmol substrate) replace stoichiometric reagents like palladium catalysts and aluminum chloride that require expensive recovery systems and generate hazardous waste streams requiring specialized disposal. The room temperature operation slashes energy costs by eliminating heating/cooling cycles that typically account for 45% of manufacturing expenses in thermal processes, while the one-pot design reduces labor costs through simplified process monitoring and fewer quality control checkpoints during production.
- Reducing Lead Time for High-Purity Intermediates: Batch cycle time compression from over two days to under two hours enables rapid response to fluctuating demand patterns while supporting just-in-time inventory models preferred by modern pharmaceutical supply chains. The elimination of intermediate purification steps removes critical path dependencies that traditionally cause production bottlenecks during scale-up transitions from lab to plant scale. This streamlined workflow allows immediate implementation of continuous manufacturing principles where product flows directly from reaction to crystallization without hold times between stages, significantly improving throughput capacity without additional capital investment while maintaining consistent quality attributes across production runs.
- Commercial Scale-Up of Complex Intermediates: The documented success across multiple catalyst variants (A-D) demonstrates robust process flexibility that accommodates diverse substrate requirements while maintaining high yields through simple parameter adjustments rather than fundamental re-engineering. The absence of specialized equipment requirements—using standard glass-lined reactors instead of pressure vessels needed for high-temp processes—enables seamless technology transfer to existing manufacturing facilities without costly retrofitting investments. Most critically, the inherent stability of the reaction pathway under ambient conditions provides exceptional batch-to-batch consistency essential for regulatory compliance while allowing flexible production scheduling that can rapidly adjust to unexpected demand surges without quality compromises.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN107573276B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
