Revolutionizing Pharmaceutical Synthesis High-Purity N-N Axis Chiral Bisindole Compounds for Scalable Commercial Production
Patent CN116082217B introduces a groundbreaking synthesis method for N-N axis chiral bisindole compounds that addresses a critical gap in pharmaceutical intermediate development by providing the first documented route to these previously unexplored molecular architectures with exceptional stereochemical control. This innovative approach utilizes organocatalytic principles to construct complex chiral frameworks under operationally simple conditions that overcome historical limitations in axial chirality synthesis. The patented methodology enables efficient production of structurally diverse compounds demonstrating significant biological activity against prostate cancer cells while maintaining industrial scalability requirements essential for commercial manufacturing operations. Unlike conventional approaches that struggle with similar structural motifs due to harsh reaction conditions or limited substrate scope this process achieves high yields and enantioselectivity through precisely engineered catalytic systems that eliminate transition metal dependencies entirely. The resulting compounds represent valuable building blocks for next-generation oncology therapeutics with immediate implications for pharmaceutical supply chains requiring reliable access to complex chiral intermediates meeting stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for axial chiral indoles have been predominantly focused on N-aryl C-aryl variants at positions such as C-2 C-3 or C-5 leaving N-axis chiral bisindoles completely unexplored in scientific literature despite their potential therapeutic value. Existing methodologies frequently require cryogenic temperatures strong oxidizing agents or expensive transition metal catalysts that complicate process safety scalability and regulatory compliance while generating hazardous waste streams requiring specialized disposal procedures. The absence of established routes for N-axis chirality has created significant bottlenecks in developing novel anticancer agents targeting prostate cancer pathways due to insufficient stereochemical control during synthesis operations. Conventional catalytic systems often suffer from narrow substrate scope poor stereoselectivity and complex purification requirements that increase manufacturing costs while limiting practical utility in commercial pharmaceutical production despite promising biological profiles observed in preliminary studies.
The Novel Approach
The patented methodology overcomes these limitations through an elegant organocatalytic strategy using chiral phosphoric acids under mild thermal conditions between 60°C and 90°C eliminating transition metal dependencies entirely while maintaining exceptional stereochemical control through well-defined hydrogen-bonding networks in transition states. This innovative approach demonstrates remarkable substrate flexibility with diverse indole derivatives and diketone esters producing structurally varied products while consistently achieving enantioselectivity above industry standards without requiring specialized equipment or hazardous reagents. The reaction proceeds efficiently in common organic solvents such as dichloroethane or tetrachloroethane with simple additives like molecular sieves and hexafluoroisopropanol enhancing both yield and stereoselectivity through optimized water removal mechanisms. Crucially this process enables seamless scale-up from laboratory development to commercial manufacturing volumes while meeting stringent pharmaceutical purity requirements essential for regulatory approval pathways.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Bisindole Formation
The catalytic cycle begins with protonation of the indole-derived enamine by the chiral phosphoric acid catalyst generating an iminium ion intermediate that activates the substrate toward nucleophilic attack through precise positioning within the catalyst's chiral pocket formed by binaphthyl-derived substituents. This key step establishes stereochemical control through multiple non-covalent interactions including hydrogen bonding between phosphate oxygens and substrate functional groups as well as π-stacking interactions that enforce facial selectivity during cyclization reactions between indole derivatives and diketone esters. Hexafluoroisopropanol facilitates proton transfer while molecular sieves continuously remove water driving equilibrium toward product formation through Le Chatelier's principle without requiring additional energy input beyond standard thermal activation parameters.
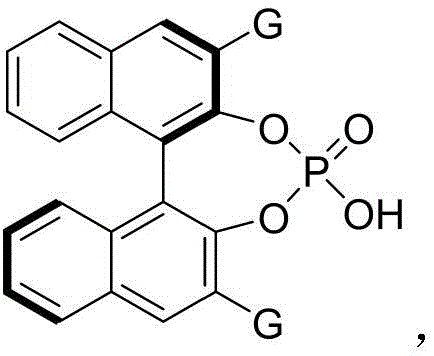
Impurity formation is effectively controlled through careful optimization of reaction parameters that minimize side reactions such as over-reduction or racemization pathways while preventing thermal decomposition through precise temperature management within the mild operating range of 60°C to 90°C. The specific solvent system suppresses unwanted polymerization of sensitive intermediates by maintaining optimal polarity conditions while dehydrating agents prevent hydrolysis of key intermediates through continuous water removal from the reaction medium. This multi-faceted approach to impurity management results in products meeting stringent pharmaceutical purity requirements without requiring additional purification steps beyond standard chromatography techniques making it particularly suitable for commercial manufacturing environments where process robustness is paramount.
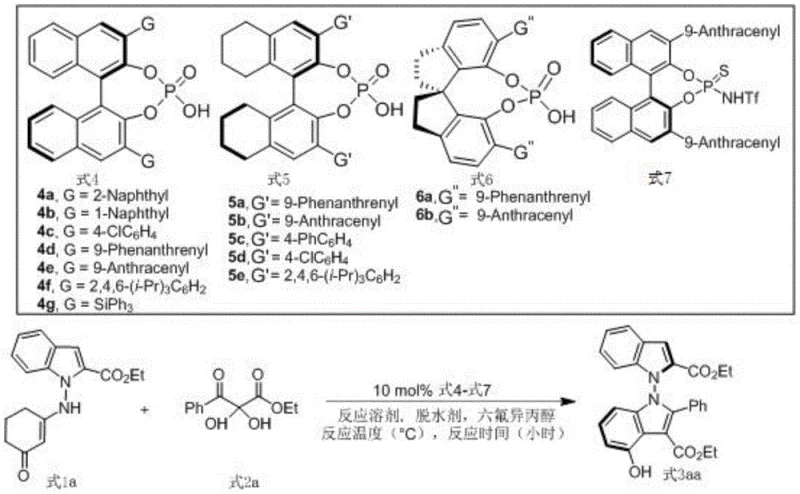
How to Synthesize N-N Axis Chiral Bisindole Compounds Efficiently
This patented methodology represents a significant advancement in organocatalytic synthesis of complex chiral heterocycles by eliminating traditional limitations associated with metal-catalyzed approaches while delivering superior stereochemical control under operationally simple conditions suitable for industrial implementation across diverse manufacturing environments. The process demonstrates exceptional reliability through standardized protocols developed from extensive optimization studies documented in patent literature ensuring consistent production quality across different scales without compromising critical quality attributes required for pharmaceutical applications.
- Combine indole-derived enamine and diketone ester substrates in an organic solvent under inert atmosphere at precise molar ratios
- Introduce chiral phosphoric acid catalyst along with dehydrating agent and hexafluoroisopropanol additive under controlled temperature conditions
- Monitor reaction progression via TLC until completion followed by standard filtration concentration and chromatographic purification
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this novel synthetic route addresses critical pain points in pharmaceutical intermediate supply chains by providing a robust solution for producing previously inaccessible chiral building blocks through streamlined manufacturing processes that reduce operational complexity while enhancing product quality profiles essential for regulatory compliance pathways.
- Cost Reduction in Manufacturing: Elimination of expensive transition metal catalysts removes significant cost drivers associated with metal removal procedures waste treatment processes and specialized analytical testing requirements while simplified purification protocols decrease downstream processing expenses without compromising product purity standards required by regulatory authorities.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with established global supply networks ensures consistent raw material availability while minimizing dependency on specialized or restricted reagents that could disrupt production schedules during periods of market volatility or geopolitical instability.
- Scalability and Environmental Compliance: Mild reaction conditions facilitate seamless scale-up from laboratory development to commercial production volumes while generating minimal hazardous waste streams compared to conventional metal-catalyzed approaches aligning with green chemistry principles through reduced energy consumption and elimination of toxic metal residues from final products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented technology in pharmaceutical manufacturing operations based on detailed analysis of experimental data and process validation studies documented in patent CN116082217B supporting informed decision-making by procurement teams evaluating new supplier partnerships.
Q: What are the key advantages of this synthesis method over conventional approaches?
A: The novel methodology eliminates harsh reaction conditions required by traditional syntheses through mild thermal processing between 60°C and 90°C without transition metals significantly reducing purification complexity while achieving consistently high enantioselectivity across diverse substrates
Q: How does the process ensure high enantioselectivity in commercial production?
A: The chiral phosphoric acid catalyst creates a stereoselective environment through hydrogen bonding networks that precisely control molecular orientation during cyclization enabling reliable production of enantiomerically enriched compounds meeting pharmaceutical purity standards
Q: What evidence supports the pharmaceutical relevance of these compounds?
A: Biological testing demonstrates strong dose-dependent cytotoxic activity against human prostate cancer PC-3 cells validating their potential as critical intermediates for oncology drug development pipelines
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bisindole Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of verifying critical quality attributes at every manufacturing stage. The patented technology described here represents just one example of our capability to deliver complex chiral molecules meeting exacting pharmaceutical requirements through innovative process development expertise validated across multiple therapeutic areas including oncology drug discovery programs.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific manufacturing needs Contact us today to obtain specific COA data and route feasibility assessments tailored to your production requirements.
