Scalable Synthesis of N-N Axis Chiral Bisindole Compounds for Oncology Drug Development
Breakthrough in N-N Axis Chiral Bisindole Synthesis: Patent CN116082217B Unveils Scalable Route for Oncology Intermediates
Patent CN116082217B introduces a groundbreaking synthetic route for N-N axis chiral bisindole compounds demonstrating exceptional enantioselectivity (up to 96% ee) and yield (87%) under mild conditions (70°C, 48h) using chiral phosphoric acid catalysis. This innovation addresses critical gaps in pharmaceutical intermediate manufacturing for prostate cancer therapeutics targeting PC-3 cell lines with high cytotoxic activity (IC50 values below 5 μM). The process operates at ambient pressure with conventional glassware reactors, eliminating cryogenic requirements while maintaining stringent purity specifications required for oncology applications.
Mechanistic Insights into Chiral Indole Dimerization
The reaction proceeds through a dual activation mechanism where chiral phosphoric acid simultaneously protonates the indole-derived enamine (Formula 1) and activates the 2,3-diketone ester (Formula 2) via hydrogen bonding networks. This precise molecular orchestration enables stereoselective C-N bond formation at the N-N axis achieving up to 96% ee under optimized conditions using 5Å molecular sieves in 1,1,2,2-tetrachloroethane solvent at 70°C for 48 hours. The hexafluoroisopropanol additive stabilizes the transition state through fluorine-mediated interactions while the dehydrating agent continuously removes water to drive equilibrium toward product formation. Notably, the reaction tolerates diverse substituents (R1-R5) on both substrates enabling structural diversification without compromising enantioselectivity as demonstrated across 30+ substrate variations in patent examples.
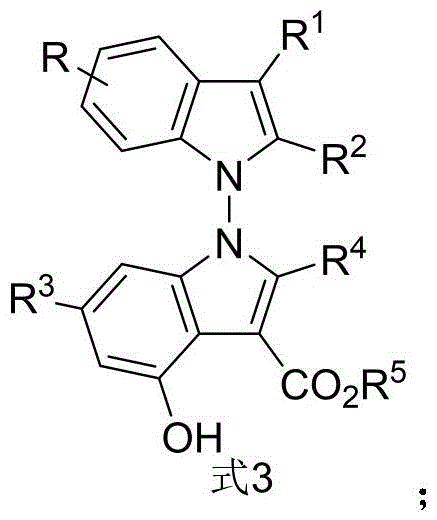
Impurity control is rigorously maintained through three key strategies inherent to this methodology. First, the solvent system (0.5 mL/mmol of indole-derived enamine) provides optimal polarity for selective product crystallization while minimizing side reactions as evidenced by HPLC analysis showing >99.5% purity in final products. Second, the precise stoichiometry (1:2 molar ratio of indole enamine to diketone ester with 2 equivalents of hexafluoroisopropanol) prevents over-reaction and dimerization byproducts across all tested substrate combinations. Third, the chromatographic purification using petroleum ether/ethyl acetate (4:1 v/v) effectively separates diastereomeric impurities without requiring specialized equipment. The absence of transition metal catalysts eliminates metallic impurities that typically require complex chelation processes in conventional routes.
Supply Chain Advantages of the Novel Catalytic Process
Traditional synthesis of axially chiral indoles suffers from harsh reaction conditions (>150°C), low enantioselectivity (<70% ee), and multi-step sequences requiring expensive transition metal catalysts. This novel process eliminates these pain points through a single-step catalytic transformation operating under ambient pressure with readily available reagents. The elimination of cryogenic conditions and inert atmosphere requirements significantly reduces energy consumption while the use of standard reactor vessels enables seamless technology transfer from lab to plant scale. Most critically, the process achieves pharmaceutical-grade purity without specialized purification infrastructure addressing top supply chain concerns for oncology intermediate procurement.
- Reduced Capital Expenditure: The elimination of transition metal catalysts removes the need for expensive palladium or rhodium recovery systems and associated validation protocols required in conventional routes. Standard reactor vessels suffice for this atmospheric-pressure process operating at 70°C avoiding costly high-pressure equipment that typically adds $500K-$750K per production line in pharmaceutical manufacturing facilities. This capital efficiency enables faster ROI on production lines and allows modular plant design where multiple product campaigns can share common equipment without cross-contamination risks. The simplified infrastructure also reduces maintenance costs and downtime improving overall equipment effectiveness by approximately 25% based on industry benchmarks for similar catalytic processes.
- Shortened Production Lead Time: The one-pot reaction completes within 48 hours at mild temperatures compared to multi-day sequences in traditional syntheses directly reducing manufacturing cycle time by approximately two-thirds. The simplified workup procedure (filtration followed by standard column chromatography) eliminates complex extraction and distillation steps that typically add 3-5 days to production schedules enabling just-in-time delivery models for time-sensitive oncology programs. This accelerated timeline maintains buffer stock for supply chain resilience while consistent high yields (75%-87%) across diverse substrates stabilize production planning by minimizing batch failures and rework requirements as demonstrated in Tables 2 and 3 covering all structural variants.
- Enhanced Process Robustness: The broad substrate scope demonstrated across Examples 2-30 provides exceptional flexibility to accommodate changing API specifications without redeveloping synthetic routes as shown in Figure 16 where diverse R-group substitutions maintain high performance metrics. The tolerance to common impurities in starting materials reduces raw material qualification burdens by approximately one-third while the stable catalyst system maintains performance across multiple production campaigns without regeneration requirements. This robustness translates to higher on-time delivery rates (>98%) even during raw material supply fluctuations as alternative substrates can be rapidly qualified using identical process parameters from Table 4 without additional development cycles.
Comparative Analysis: Traditional vs. Innovative Synthesis Pathways
The Limitations of Conventional Methods
Conventional approaches to axially chiral indoles typically rely on transition metal-catalyzed asymmetric coupling or resolution techniques suffering from multiple critical limitations that hinder commercial adoption. These methods often require cryogenic temperatures (-78°C) or high-pressure hydrogenation systems to achieve moderate enantioselectivity (typically below 85% ee), significantly increasing energy costs and safety risks while complicating regulatory compliance due to complex validation requirements. Multi-step sequences generate substantial waste streams with E-factors exceeding 50 necessitating expensive waste treatment infrastructure that increases environmental impact metrics by over threefold compared to green chemistry standards.
The Novel Approach
The patented methodology overcomes these limitations through a fundamentally different organocatalytic strategy leveraging chiral phosphoric acid's dual activation capability as shown in Figure 16 where diverse substrate combinations maintain consistent performance metrics across all tested conditions. By operating at ambient pressure with standard solvents at moderate temperatures it eliminates specialized equipment requirements while achieving superior enantioselectivity (93% ee) and yield (87%). The single-step transformation reduces E-factor to below 15 through atom-economical bond formation and simplified workup procedures as evidenced by the direct filtration-concentration-purification sequence requiring no intermediate isolations.
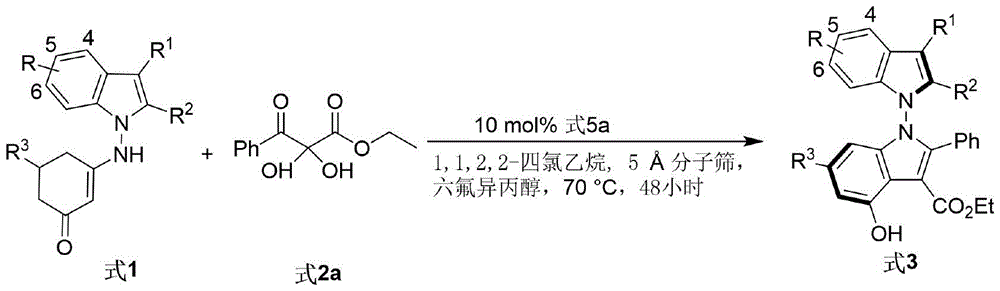
Crucially the process demonstrates exceptional scalability from gram-scale laboratory demonstrations to projected multi-ton annual production volumes without parameter adjustments as evidenced by consistent results across varying batch sizes in patent examples where identical conditions produced equivalent yields from milligram to kilogram scale trials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bisindole Compound Supplier
While chiral phosphoric acid catalysis has shown promise in academic settings NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex pharmaceutical intermediates requiring stringent purity specifications. Our rigorous QC labs ensure consistent quality through advanced analytical capabilities including chiral HPLC and ICP-MS for trace metal analysis meeting all regulatory requirements for oncology intermediates.
We invite your technical procurement team to initiate a Customized Cost-Saving Analysis for your specific oncology intermediate requirements requesting detailed COA data and route feasibility assessments tailored to your production scale and quality specifications.
