Advanced Metal-Free Synthesis of 4-Trifluoromethylnicotinic Acid for Scalable Agrochemical Production
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for compounds like flonicamid, a next-generation pyridinamide insecticide. At the heart of this value chain lies 4-trifluoromethylnicotinic acid, a critical building block whose efficient production determines the cost and availability of the final pesticide. A significant technological breakthrough in this domain is detailed in Chinese patent CN114349694B, which discloses a novel, metal-free synthetic methodology that fundamentally reshapes the manufacturing landscape for this key heterocyclic compound. Unlike legacy processes that rely heavily on scarce transition metals and harsh reaction environments, this invention leverages a streamlined condensation and cyclization strategy using readily available formate and acetate esters. For R&D directors and procurement strategists alike, this patent represents a pivotal shift towards more sustainable and economically viable chemical manufacturing, offering a pathway to high-purity intermediates without the baggage of complex catalytic systems.
The significance of this development extends beyond mere academic interest; it addresses critical bottlenecks in the supply chain for fluorinated pyridine derivatives. By establishing a route that avoids the use of palladium and copper catalysts, the technology mitigates the volatility associated with precious metal pricing and supply constraints. Furthermore, the process demonstrates exceptional control over impurity profiles, achieving product purities exceeding 98% in optimized examples, which is a stringent requirement for downstream pharmaceutical and agrochemical applications. As the industry moves towards greener chemistry standards, the adoption of such base-catalyzed, atom-economical processes becomes not just an option but a strategic necessity for maintaining competitiveness in the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-trifluoromethylnicotinic acid has been plagued by significant technical and economic hurdles that hinder efficient industrial production. Traditional routes often commence with ethyl trifluoroacetoacetate and cyanoacetamide, necessitating a multi-step sequence that includes chlorination with phosphorus oxychloride and subsequent hydrogenolysis using palladium on carbon. This reliance on noble metal catalysts introduces severe cost penalties and operational complexities, as the removal of trace metal residues requires additional purification steps that lower overall yield. Moreover, alternative pathways involving vinyl ethyl ether and trifluoroacetyl chloride suffer from the use of expensive, imported raw materials like methyl 3-methoxyenoate, which are not only costly but also chemically unstable, posing storage and handling risks. Other methods utilizing strong bases like LDA for carbonyl insertion reactions operate under extremely harsh conditions with poor regioselectivity, making them unsuitable for the rigorous safety and consistency standards of modern chemical plants.
The Novel Approach
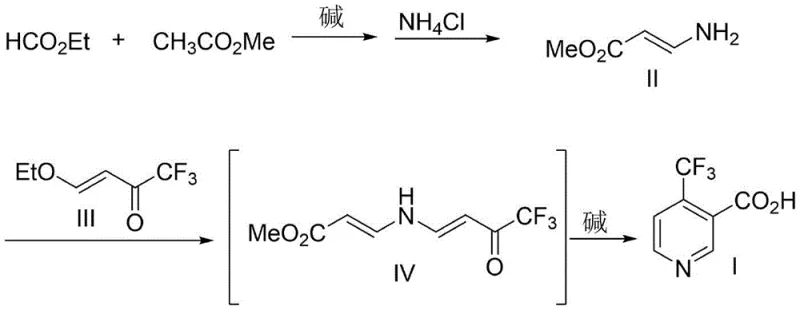
In stark contrast, the methodology outlined in patent CN114349694B offers a transformative solution by utilizing a concise two-step sequence that bypasses these historical limitations entirely. The core innovation lies in the generation of 3-aminoacrylate from simple, commodity chemicals—ethyl formate and methyl acetate—followed by a direct condensation with 4-ethoxy-1,1,1-trifluoro-3-buten-2-one. This approach eliminates the need for any transition metal catalysts, thereby removing the associated costs of catalyst procurement, recovery, and heavy metal waste treatment. The reaction conditions are notably mild, proceeding effectively at temperatures between 10°C and 80°C, which significantly reduces energy consumption compared to high-temperature or high-pressure alternatives. By simplifying the molecular construction of the pyridine ring through a base-mediated cyclization, this novel route ensures a shorter production timeline, higher overall throughput, and a dramatically reduced environmental footprint, aligning perfectly with the principles of green chemistry.
Mechanistic Insights into Base-Catalyzed Cyclization and Condensation
The chemical elegance of this synthesis is rooted in a sophisticated yet robust mechanism that orchestrates the formation of the trifluoromethylated pyridine core through precise nucleophilic attacks and eliminations. The process initiates with a Claisen-type condensation where a base, such as sodium ethoxide, deprotonates the methyl acetate to form an enolate, which subsequently attacks the carbonyl carbon of the ethyl formate. This intermediate is then treated with an ammonium salt, likely facilitating an aminolysis or substitution reaction to yield the crucial 3-aminoacrylate species. This amino-ester acts as a versatile 1,3-dinucleophile equivalent, poised to react with the electrophilic trifluorinated ketone. In the second stage, the 3-aminoacrylate undergoes a Michael-type addition or condensation with 4-ethoxy-1,1,1-trifluoro-3-buten-2-one, forming a linear intermediate that contains all the carbon atoms necessary for the final ring structure. The presence of the electron-withdrawing trifluoromethyl group enhances the electrophilicity of the ketone, driving the reaction forward efficiently even under mild thermal conditions.
Following the initial coupling, the addition of a base triggers the critical ring-closing step, which involves an intramolecular cyclization followed by hydrolysis and aromatization to establish the pyridine system. The base facilitates the deprotonation of active methylene groups and promotes the elimination of ethanol or water molecules, driving the equilibrium towards the aromatic product. This mechanistic pathway is highly advantageous because it avoids the formation of stable by-products that are common in metal-catalyzed cross-couplings. The result is a clean reaction profile where the primary impurities are easily separable salts or unreacted starting materials, rather than complex metal-organic complexes. This clarity in the reaction mechanism translates directly to process reliability, allowing for tight control over the impurity spectrum and ensuring that the final 4-trifluoromethylnicotinic acid meets the stringent purity specifications required for regulatory approval in agrochemical formulations.
How to Synthesize 4-Trifluoromethylnicotinic Acid Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the yield of the key aminoacrylate intermediate and ensure complete cyclization in the second step. The process begins by reacting ethyl formate and methyl acetate with a base like sodium ethoxide in methyl tert-butyl ether at controlled low temperatures, followed by treatment with saturated ammonium chloride solution to isolate the 3-aminoacrylate. This intermediate is then coupled with the trifluorinated ketone in an alcohol solvent, where the subsequent addition of base induces the ring closure. The detailed standardized operating procedures, including specific molar ratios, reaction times, and workup protocols necessary to replicate the high yields reported in the patent, are provided in the technical guide below.
- Prepare 3-aminoacrylate by reacting ethyl formate and methyl acetate with a base in MTBE, followed by ammonium salt treatment.
- React the resulting 3-aminoacrylate with 4-ethoxy-1,1,1-trifluoro-3-buten-2-one in an alcohol solvent.
- Add base to induce ring-closing hydrolysis, followed by acidification to isolate the final 4-trifluoromethylnicotinic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers profound strategic benefits that extend well beyond the laboratory bench. The most immediate impact is seen in the drastic simplification of the raw material portfolio; by shifting away from specialized, imported reagents like methyl 3-methoxyenoate to bulk commodities like ethyl formate and methyl acetate, companies can insulate themselves from supply disruptions and price volatility. The elimination of palladium and copper catalysts removes a significant cost center, as precious metals represent a substantial portion of the variable cost in traditional fine chemical manufacturing. Furthermore, the absence of heavy metals simplifies the waste management workflow, reducing the fees associated with hazardous waste disposal and lowering the regulatory compliance burden. This streamlined material flow enhances the overall agility of the supply chain, allowing for faster response times to market demands for flonicamid and related agrochemical products.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the complete removal of expensive noble metal catalysts from the process flow. In conventional routes, the cost of palladium on carbon and the associated ligands or copper salts can be prohibitive, especially when factoring in the loss of metal during recovery and the cost of refining residues. By replacing these with inexpensive inorganic bases like sodium ethoxide or potassium hydroxide, the variable cost per kilogram of the intermediate is significantly lowered. Additionally, the use of common solvents such as ethanol and methyl tert-butyl ether, which are easily recovered and recycled, further contributes to operational expenditure savings. The higher yields reported in this method also mean less raw material is wasted per unit of product, compounding the cost efficiency gains throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on widely available, commodity-grade starting materials. Ethyl formate and methyl acetate are produced on a massive industrial scale for various applications, ensuring a stable and continuous supply that is not subject to the geopolitical or logistical constraints often faced with specialized fine chemical imports. This localization of raw material sourcing reduces lead times and minimizes the risk of production stoppages due to material shortages. Moreover, the stability of the intermediates generated in this route reduces the need for cold chain logistics or specialized storage facilities, simplifying inventory management and reducing the risk of spoilage during transit. This robustness makes the supply chain more predictable and easier to manage for long-term production planning.
- Scalability and Environmental Compliance: From an operational perspective, the mild reaction conditions and simple equipment requirements make this process exceptionally easy to scale from pilot plant to full commercial production. The absence of high-pressure hydrogenation steps or sensitive organometallic reactions reduces the safety risks and capital investment needed for reactor infrastructure. Environmentally, the process aligns with increasingly strict global regulations regarding heavy metal discharge and solvent emissions. By generating less hazardous waste and avoiding persistent metal contaminants, manufacturers can more easily meet environmental, social, and governance (ESG) targets. This compliance advantage not only avoids potential fines but also enhances the brand reputation of the manufacturer as a sustainable partner in the global agrochemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing frameworks.
Q: What are the primary advantages of this new synthesis route over traditional palladium-catalyzed methods?
A: The primary advantage is the complete elimination of expensive noble metal catalysts like palladium and copper. Traditional routes often require Pd/C for hydrogenolysis or Pd/Cu for oxidation, which significantly increases raw material costs and introduces complex heavy metal removal steps. This new method utilizes common organic bases and inexpensive esters, drastically simplifying the purification process and reducing the environmental burden associated with heavy metal waste disposal.
Q: How does this method address the stability issues of intermediates found in previous patents?
A: Previous methods relied on unstable intermediates like methyl 3,3-dimethoxypropionate, which are difficult to prepare and store. This patented route generates the key 3-aminoacrylate intermediate in situ or via a stable isolation process using robust starting materials like ethyl formate and methyl acetate. This ensures consistent feedstock quality and eliminates the supply chain risks associated with importing unstable or specialized reagents.
Q: Is this process suitable for large-scale industrial manufacturing of flonicamid intermediates?
A: Yes, the process is explicitly designed for industrial scalability. It operates under mild reaction conditions (temperatures ranging from -10°C to 85°C) and uses standard solvents like methyl tert-butyl ether and ethanol. The absence of high-pressure hydrogenation or sensitive organometallic reagents makes the reaction easier to control in large reactors, ensuring high reproducibility and safety profiles required for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethylnicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume outputs. Our state-of-the-art facilities are equipped to handle the specific solvent systems and base-catalyzed reactions required for this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 4-trifluoromethylnicotinic acid meets the highest industry standards. We are committed to delivering consistent quality and supply continuity for our global partners in the agrochemical sector.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the potential economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and enhance your competitive position in the market for high-value fluorinated intermediates.
