Advanced Metal-Free Synthesis of 4-Trifluoromethylnicotinic Acid for Commercial Agrochemical Production
Advanced Metal-Free Synthesis of 4-Trifluoromethylnicotinic Acid for Commercial Agrochemical Production
The global demand for high-efficiency, low-toxicity agrochemicals has placed significant pressure on the supply chains of key heterocyclic intermediates. Specifically, 4-trifluoromethylnicotinic acid serves as a critical building block for Flonicamid, a next-generation pyridylamide insecticide widely used to control sucking pests in cotton and vegetable crops. Recent intellectual property developments, specifically patent CN114349694A, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in the manufacturing of this vital compound. This new approach leverages a concise two-step sequence starting from inexpensive formate and acetate esters, bypassing the need for costly transition metal catalysts that have historically plagued this sector. For R&D directors and procurement strategists, understanding the nuances of this metal-free pathway is essential for securing a competitive edge in the agrochemical intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4-trifluoromethylnicotinic acid has been hindered by complex multi-step sequences that rely heavily on precious metal catalysis. Traditional routes often involve the cyclization of ethyl trifluoroacetoacetate followed by chlorination and subsequent palladium-catalyzed hydrogenolysis to remove chlorine atoms. These processes are not only capital intensive due to the requirement for palladium on carbon catalysts but also introduce significant risks regarding heavy metal residues in the final active pharmaceutical or agrochemical ingredient. Furthermore, alternative pathways utilizing vinyl ether and trifluoroacetyl chloride often depend on unstable intermediates like methyl 3,3-dimethoxypropionate, which are difficult to source commercially and prone to degradation during storage. The reliance on imported specialty reagents and the moderate overall yields, often hovering around 60%, create substantial volatility in supply continuity and cost structures for downstream manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the innovative strategy disclosed in the referenced patent utilizes a direct condensation of ethyl formate and methyl acetate to generate a stable 3-amino acrylate intermediate. This foundational step eliminates the need for exotic starting materials, relying instead on commodity chemicals that are readily available in bulk quantities globally. The subsequent reaction with 4-ethoxy-1,1,1-trifluoro-3-buten-2-one proceeds under mild alkaline conditions to form the pyridine ring directly, achieving exceptional purity levels without the need for extensive purification columns typically required to remove metal catalysts. By streamlining the synthesis into fewer operational units and removing the dependency on noble metals, this route offers a robust platform for cost reduction in pesticide manufacturing. The simplicity of the workup, involving straightforward acidification and precipitation, further enhances the operational efficiency, making it an ideal candidate for commercial scale-up of complex heterocyclic intermediates.
Mechanistic Insights into Ester Condensation and Cyclization
The core of this synthetic breakthrough lies in the precise control of the Claisen-type condensation and the subsequent intramolecular cyclization. In the first stage, the reaction between ethyl formate and methyl acetate is mediated by a strong base such as sodium ethoxide in a solvent like methyl tert-butyl ether (MTBE). This environment facilitates the formation of an enolate species which attacks the formate carbonyl, eventually leading to the formation of methyl 3-aminoacrylate upon treatment with ammonium chloride. The choice of MTBE as a solvent is particularly strategic, as it provides excellent solubility for the organic reactants while allowing for easy separation of inorganic salts, thereby minimizing side reactions that could lead to polymeric impurities. This careful orchestration of reaction conditions ensures that the amino-acrylate intermediate is generated with high fidelity, setting the stage for the crucial ring-closing step.
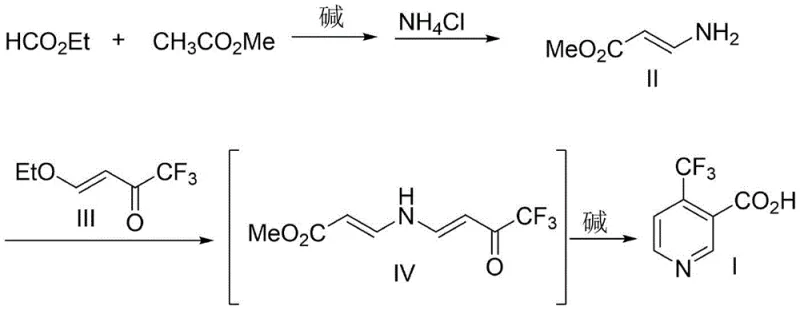
Following the formation of the amino-acrylate, the mechanism shifts to a nucleophilic attack on the electron-deficient double bond of the trifluorinated ketone. The nitrogen atom of the amino group attacks the beta-carbon of the enone system, initiating a cascade that ultimately closes the pyridine ring. The presence of the trifluoromethyl group exerts a strong electron-withdrawing effect, which activates the ketone towards nucleophilic attack but also requires careful pH control during the hydrolysis phase to prevent decomposition. The final step involves base-mediated hydrolysis of the ester moiety followed by acidification to pH 2, which precipitates the target 4-trifluoromethylnicotinic acid as a high-purity white solid. This mechanistic pathway effectively suppresses the formation of regio-isomers, a common issue in pyridine synthesis, ensuring that the high-purity 4-trifluoromethylnicotinic acid produced meets the stringent specifications required for biological activity testing and final formulation.
How to Synthesize 4-Trifluoromethylnicotinic Acid Efficiently
Implementing this synthesis at a pilot or production scale requires strict adherence to the molar ratios and temperature profiles outlined in the patent data to maximize yield and minimize waste. The process begins with the preparation of the amino-acrylate precursor, where maintaining the reaction temperature between -10°C and 30°C is critical to controlling the exotherm and preventing the degradation of the sensitive enamine intermediate. Once the intermediate is isolated or used in situ, it is reacted with the trifluorinated ketone in an alcoholic solvent such as ethanol or methanol, followed by the addition of an alkoxide base to drive the cyclization. The detailed standardized synthesis steps, including specific stirring rates, addition times, and quenching procedures necessary for reproducible results, are provided in the technical guide below.
- Prepare methyl 3-aminoacrylate by reacting ethyl formate and methyl acetate with a base in MTBE, followed by ammonium salt treatment.
- React the resulting 3-aminoacrylate with 4-ethoxy-1,1,1-trifluoro-3-buten-2-one in an alcohol solvent.
- Add base to induce cyclization and hydrolysis, then acidify to precipitate the final 4-trifluoromethylnicotinic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift towards this metal-free synthesis represents a significant opportunity to de-risk the sourcing of critical agrochemical intermediates. By eliminating the dependence on palladium and copper catalysts, manufacturers can avoid the price volatility associated with precious metals and the regulatory burdens of demonstrating residual metal clearance in final products. The use of commodity esters like ethyl formate and methyl acetate ensures that raw material availability remains stable even during global supply chain disruptions, providing a level of security that imported specialty reagents cannot match. Furthermore, the simplified downstream processing reduces the consumption of solvents and energy, contributing to a more sustainable manufacturing footprint that aligns with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost center from the bill of materials, while also negating the need for specialized filtration equipment required for catalyst recovery. The high overall yield reported in the patent examples implies that less raw material is wasted per kilogram of finished product, driving down the effective cost of goods sold significantly. Additionally, the ability to use standard stainless steel reactors rather than glass-lined vessels required for highly corrosive alternatives further reduces capital expenditure requirements for production facilities.
- Enhanced Supply Chain Reliability: Sourcing strategies can be simplified by relying on bulk chemicals that are produced by multiple vendors globally, reducing the risk of single-source supplier failure. The robustness of the reaction conditions means that production schedules are less likely to be impacted by minor variations in raw material quality or environmental fluctuations, ensuring consistent on-time delivery to downstream formulators. This reliability is crucial for maintaining the production timelines of finished insecticides like Flonicamid, where seasonal demand peaks require uninterrupted supply flows.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of toxic heavy metals simplify the waste treatment process, lowering the costs associated with effluent disposal and environmental permitting. The process is inherently safer to operate at scale due to the lack of high-pressure hydrogenation steps or pyrophoric reagents, facilitating easier technology transfer from laboratory to multi-ton production scales. This scalability ensures that the supply chain can rapidly respond to increased market demand without requiring extensive re-engineering of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived from the specific advantages highlighted in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The answers provided reflect the operational realities of scaling this chemistry from gram to ton quantities.
Q: Why is this new synthesis route superior to traditional palladium-catalyzed methods?
A: Traditional methods often rely on expensive and toxic precious metal catalysts like palladium or copper, which require complex removal steps and increase production costs. This novel route utilizes common ester condensation and base-catalyzed cyclization, eliminating the need for heavy metal catalysts entirely while achieving yields exceeding 96%.
Q: What are the key stability advantages of the intermediates in this process?
A: Previous routes utilized unstable intermediates like methyl 3,3-dimethoxypropionate, which complicated storage and handling. This method generates stable 3-amino acrylate intermediates in situ using robust starting materials like ethyl formate and methyl acetate, significantly improving process reliability.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions are mild, operating between -10°C and 85°C, and utilize common solvents like MTBE and ethanol. The absence of harsh reagents and the high purity of the final product (over 98%) make it highly scalable for commercial agrochemical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethylnicotinic Acid Supplier
As the agrochemical industry continues to evolve towards more sustainable and cost-effective manufacturing practices, the adoption of advanced synthetic routes like the one described in CN114349694A becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovations to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 4-trifluoromethylnicotinic acid meets the highest international standards for agrochemical intermediates. We understand that consistency is paramount in crop protection, and our dedicated process engineering team works tirelessly to optimize every parameter of the synthesis for maximum efficiency.
We invite procurement leaders and R&D teams to engage with us to explore how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this metal-free route for your operations. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of this high-value intermediate into your production pipeline.
