Revolutionizing Amide Bond Formation: A Scalable NHC-Catalyzed Route for Commercial Production
Revolutionizing Amide Bond Formation: A Scalable NHC-Catalyzed Route for Commercial Production
The formation of the amide bond remains one of the most critical transformations in modern organic synthesis, particularly within the pharmaceutical and fine chemical sectors where approximately 25% of drug molecules rely on this structural motif. Patent CN108558692B introduces a groundbreaking preparation method that leverages N-heterocyclic carbene (NHC) organocatalysis to drive the direct coupling of organic acid esters and organic amines. This technological advancement represents a paradigm shift away from traditional, resource-intensive activation strategies, offering a pathway that is not only chemically elegant but also commercially viable for large-scale operations. By utilizing a specific NHC catalyst structure in conjunction with mild bases and common organic solvents, this invention achieves high conversion rates under exceptionally gentle conditions, typically ranging from 0°C to 40°C. For R&D directors and process chemists, this implies a robust methodology that minimizes thermal stress on sensitive functional groups, thereby preserving the integrity of complex molecular architectures often found in late-stage pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of amides has been dominated by two primary strategies, both of which carry significant logistical and economic burdens for supply chain managers. The first conventional approach involves the activation of carboxylic acids using reagents such as triphenylphosphine/iodine (Ph3P/I2) or uronium salts like HATU to form highly reactive esters prior to aminolysis. As illustrated in the prior art reaction schemes, this multi-step activation often results in prolonged reaction times, incomplete conversions, and the generation of stoichiometric amounts of difficult-to-remove by-products.
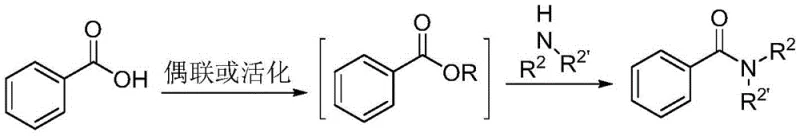
These impurities necessitate rigorous and costly purification steps, such as extensive chromatography or recrystallization, which drastically reduce overall throughput. Furthermore, the second traditional method, involving the oxidative aminolysis of aldehydes via transition metal catalysis, suffers from narrow substrate scope and the potential contamination of the final product with toxic heavy metal residues. These limitations create bottlenecks in manufacturing, leading to increased lead times and higher operational expenditures due to the need for specialized waste treatment and metal scavenging processes.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes an N-heterocyclic carbene catalyst to facilitate the direct reaction between stable organic acid esters and amines, effectively bypassing the need for pre-activation or harsh oxidants. This approach streamlines the synthetic sequence into a single pot operation where the catalyst activates the ester carbonyl in situ, rendering it susceptible to nucleophilic attack by the amine. The reaction profile is remarkably efficient, often reaching completion within 0.1 to 1.5 hours at room temperature, which stands in sharp opposition to the extended timelines of legacy protocols. By eliminating the requirement for stoichiometric activating agents and transition metals, this process inherently reduces the chemical load on the environment and simplifies the downstream workup procedure. For procurement teams, this translates to a simplified bill of materials where expensive coupling reagents are replaced by catalytic amounts of an organic molecule, fundamentally altering the cost structure of amide manufacturing.
Mechanistic Insights into NHC-Catalyzed Ester Aminolysis
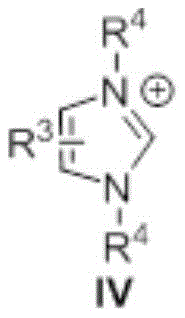
The efficacy of this transformation hinges on the unique electronic properties of the N-heterocyclic carbene catalyst, specifically the imidazolium-derived structure defined in the patent. Mechanistically, the free carbene species acts as a potent nucleophile that attacks the carbonyl carbon of the organic acid ester, forming a highly reactive acyl azolium intermediate. This intermediate possesses significantly enhanced electrophilicity compared to the parent ester, thereby lowering the activation energy barrier for the subsequent nucleophilic substitution by the organic amine. The general reaction scheme clearly depicts this catalytic cycle where the ester and amine converge in the presence of the catalyst and a base to yield the desired amide product with the regeneration of the catalytic species.
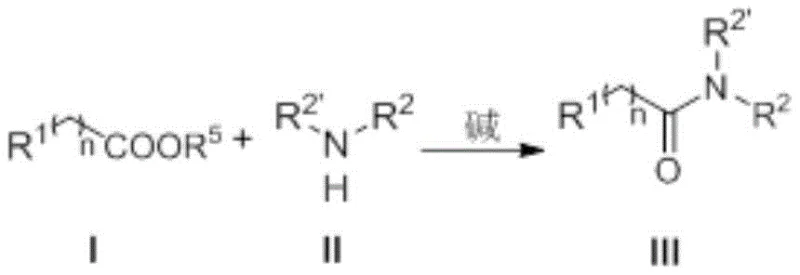
From a purity perspective, this mechanism is advantageous because it avoids the formation of stable side-products often associated with phosphine oxides or urea derivatives found in coupling reagent methods. The specific structure of the catalyst, featuring substituents at the nitrogen atoms that can be tuned for steric and electronic effects, ensures high turnover numbers and stability under the reaction conditions. The patent details that substituents such as methyl, ethyl, or aryl groups on the catalyst backbone contribute to its robustness, allowing it to function effectively even in the presence of diverse functional groups on the substrate. This level of mechanistic control provides R&D teams with the confidence to apply this chemistry to complex scaffolds without fear of catalyst decomposition or erratic reaction kinetics.
How to Synthesize Amide Compounds Efficiently
Implementing this NHC-catalyzed protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The process begins with the establishment of an inert atmosphere, typically using nitrogen or argon, to protect the sensitive carbene species from moisture and oxygen degradation. Following the dissolution of the catalyst and base in a solvent like tetrahydrofuran, the sequential addition of substrates allows for precise control over the exotherm and reaction progression. While the patent provides specific molar ratios and workup procedures involving aqueous quenching and silica gel chromatography, the core innovation lies in the simplicity of mixing stable starting materials under mild thermal conditions. Detailed standardized synthesis steps following this optimized protocol are provided below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction system under an inert gas atmosphere by dissolving the N-heterocyclic carbene catalyst and a suitable base in an organic solvent such as tetrahydrofuran.
- Sequentially add the organic acid ester substrate and the organic amine reactant to the stirred mixture, maintaining the temperature between 0°C and 40°C.
- Upon completion, quench the reaction with water, extract the product with an organic solvent, and purify the crude residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this NHC-catalyzed amidation technology offers profound strategic benefits that extend beyond mere chemical efficiency. The elimination of stoichiometric coupling reagents represents a direct reduction in raw material costs, as these activators are often among the most expensive components in a synthetic route. Furthermore, the avoidance of transition metals removes the necessity for costly metal scavenging resins and the associated analytical testing required to certify low residual metal levels in pharmaceutical ingredients. This simplification of the supply chain reduces dependency on volatile markets for precious metals and specialized reagents, thereby enhancing the resilience of the manufacturing process against global supply shocks.
- Cost Reduction in Manufacturing: The shift from stoichiometric activation to organocatalysis fundamentally lowers the cost of goods sold by removing high-priced reagents like HATU or phosphine complexes from the bill of materials. Additionally, the mild reaction conditions operate near room temperature, which significantly decreases energy consumption related to heating or cryogenic cooling compared to traditional methods that require extreme thermal inputs. The simplified workup procedure, which avoids complex extractions to remove metal salts or phosphine oxides, further reduces solvent usage and labor hours, contributing to substantial overall operational savings without compromising product quality.
- Enhanced Supply Chain Reliability: By relying on stable organic acid esters and amines rather than sensitive acid chlorides or unstable activated esters, the process utilizes starting materials that are easier to store and transport with longer shelf lives. The robustness of the NHC catalyst system means that the reaction is less prone to failure due to minor fluctuations in environmental conditions, ensuring consistent batch-to-batch performance. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the risk of delays caused by failed reactions or the need for reprocessing, ultimately securing a steady flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The absence of heavy metals and the use of benign organic solvents align this process with increasingly stringent environmental regulations and green chemistry principles. Scaling this reaction from gram to tonnage does not introduce the complications of handling toxic metal waste streams, making it easier to obtain regulatory approval for commercial production facilities. The high atom economy and reduced waste generation simplify effluent treatment protocols, lowering the environmental footprint of the manufacturing site and mitigating the risk of compliance-related shutdowns, thus ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed amide synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and licensees. Understanding these nuances is critical for evaluating the feasibility of integrating this method into existing production lines or new drug development pipelines.
Q: What are the primary advantages of using NHC catalysis over traditional coupling reagents?
A: NHC catalysis eliminates the need for expensive and hazardous coupling reagents like HATU or phosphine-based activators, significantly reducing raw material costs and simplifying waste disposal protocols while maintaining high yields under mild conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process operates at near-room temperature with short reaction times and uses common solvents like THF, making it highly scalable and energy-efficient for commercial manufacturing without requiring specialized high-pressure or cryogenic equipment.
Q: What types of substrates are compatible with this NHC-catalyzed amidation?
A: The method demonstrates broad substrate adaptability, successfully converting various organic acid esters, including those with electron-withdrawing or donating groups, and diverse organic amines into high-purity amide products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methods like the one described in CN108558692B for producing high-value amide intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of amide compound meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence ensures that the benefits of this mild, high-yield synthesis route are fully realized in the final product delivered to your facility.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in NHC-catalyzed synthesis can optimize your supply chain and accelerate your time to market.
