Revolutionizing Sulfone and Sulfonamide Production: A Scalable Radical Coupling Strategy for Global Supply Chains
Revolutionizing Sulfone and Sulfonamide Production: A Scalable Radical Coupling Strategy for Global Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable, efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN109180572B, which discloses a novel preparation method for sulfone and sulfonamide compounds. This technology represents a paradigm shift from traditional nucleophilic substitution methods to a streamlined radical coupling approach. By utilizing iodo aromatic ring compounds and sodium dithionite under inert gas conditions, this process generates aromatic ring sodium sulfinate intermediates in situ, which are subsequently reacted with various electrophiles in a one-pot fashion. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology offers a compelling value proposition characterized by high product purity, minimal side reactions, and exceptional operational simplicity.
The significance of this innovation extends beyond mere academic interest; it addresses critical bottlenecks in the supply chain of active pharmaceutical ingredients (APIs) and agrochemical intermediates. Sulfone and sulfonamide motifs are ubiquitous in medicinal chemistry, found in blockbuster drugs treating conditions ranging from diabetes to cardiovascular diseases. However, their traditional synthesis often involves hazardous reagents and multi-step protocols that inflate costs and extend lead times. The method described in CN109180572B circumvents these issues by employing a catalyst-free radical mechanism that is both green and robust. This report delves deep into the mechanistic intricacies, commercial advantages, and practical implementation of this technology, providing a comprehensive roadmap for industry stakeholders looking to optimize their cost reduction in API manufacturing strategies.
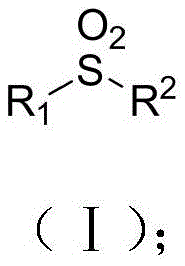
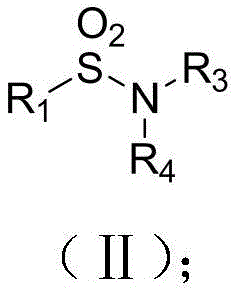
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of sulfones and sulfonamides has relied heavily on the nucleophilic substitution reaction of sulfonyl chlorides with amines or other nucleophiles. While effective in certain contexts, this conventional approach is fraught with significant drawbacks that hinder efficiency and safety. The primary concern is the reliance on sulfonyl chlorination reagents, which are notoriously corrosive and pose severe handling risks in large-scale operations. Furthermore, the preparation of these sulfonyl chlorides often requires strong oxidants and unpleasant sulfuration reagents, creating a hazardous working environment and generating substantial toxic waste streams. From a quality control perspective, the harsh oxidative conditions necessary for these traditional methods can lead to the unintended oxidation of other sensitive functional groups present on the substrate, resulting in complex impurity profiles that are difficult and expensive to remove. These factors collectively contribute to higher production costs and longer cycle times, making the conventional route less attractive for modern, high-throughput manufacturing environments.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data introduces a transformative strategy that leverages the reactivity of sodium dithionite to generate sulfur dioxide radical anions. This method initiates a free radical coupling reaction between iodo aromatic compounds and the sulfur species under heating conditions, typically around 90°C. The beauty of this system lies in its one-pot capability; once the sodium sulfinate intermediate is formed, it can be directly reacted with benzyl bromides, alkyl halides, diaryl iodides, or amines without the need for isolation. This telescoping of steps drastically reduces solvent consumption and processing time. Moreover, the reaction conditions are remarkably mild compared to traditional oxidation methods, preserving the integrity of oxidation-sensitive functional groups such as esters, ethers, and heterocycles. The result is a process that not only delivers high yields—often exceeding 75% to 85% in specific examples—but also ensures high product purity, frequently reaching 98%, thereby simplifying downstream purification and enhancing overall process economics.
Mechanistic Insights into Sodium Dithionite-Mediated Radical Coupling
To fully appreciate the technical superiority of this method, one must understand the underlying chemical mechanism. The reaction begins with the homolytic cleavage of sodium dithionite (Na2S2O4) under thermal conditions, generating sulfur dioxide radical anions (SO2•−). These highly reactive species engage in a single electron transfer (SET) process with the iodo aromatic ring compound. This electron transfer event triggers the formation of a heterocyclic or aromatic carbon radical while simultaneously releasing a molecule of sulfur dioxide. The nascent carbon radical then undergoes a rapid radical coupling reaction with another sulfur dioxide radical anion to form the key aromatic ring sodium sulfinate intermediate. This intermediate is stable enough to persist in the reaction mixture yet reactive enough to undergo subsequent nucleophilic substitution. When an electrophile such as benzyl bromide is introduced, the sulfinate anion attacks the electrophilic carbon, displacing the halide and forming the final sulfone bond. This mechanism avoids the high-energy transition states associated with ionic pathways, allowing for greater tolerance of diverse substrate structures.
Impurity control is a critical aspect of this mechanism that appeals directly to R&D directors focused on quality. Because the reaction proceeds via a radical pathway rather than a strong oxidative one, there is no risk of over-oxidizing sensitive moieties on the aromatic ring. For instance, electron-donating groups like methoxy or methyl, and electron-withdrawing groups like fluorine or cyano, remain intact throughout the process. The patent data highlights successful syntheses of complex molecules, including nucleoside derivatives, where the delicate sugar backbone and multiple hydroxyl groups are preserved without protection. This high level of chemoselectivity minimizes the formation of by-products, leading to cleaner reaction profiles. Consequently, the burden on purification teams is significantly reduced, as the crude product often requires only standard extraction and column chromatography to achieve pharmaceutical-grade purity. This mechanistic elegance translates directly into operational reliability and consistent batch-to-batch quality.
How to Synthesize High-Purity Sulfone Intermediates Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and safety. The process is designed to be straightforward, utilizing common organic solvents like N,N-dimethylformamide (DMF) and standard inert gas techniques. The key to success lies in the precise control of stoichiometry and temperature during the radical generation phase. As detailed in the patent examples, maintaining a molar ratio of approximately 1.0:1.1 for the iodo compound to sodium dithionite ensures complete conversion while minimizing excess reagent waste. The addition of phase transfer catalysts such as tetrabutylammonium iodide and potassium iodide in the second stage further enhances the reaction kinetics, facilitating the coupling with the electrophile at room temperature. For those seeking to replicate these results or scale them up, the following guide outlines the standardized operational steps derived from the patent's exemplary embodiments.
- Charge iodo aromatic ring compound and sodium dithionite into a reaction vessel under inert gas, add DMF solvent, and heat to 90°C to generate the sodium sulfinate intermediate.
- Upon cooling, add phase transfer catalysts (TBAB/KI) and the electrophile (benzyl bromide or alkyl halide) to the reaction mixture.
- Stir at room temperature, quench with brine, extract with ethyl acetate, and purify via column chromatography to obtain the high-purity sulfone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits that extend far beyond the laboratory bench. The shift away from corrosive sulfonyl chlorides and strong oxidants fundamentally alters the cost structure of sulfone production. By utilizing inexpensive and readily available starting materials like sodium dithionite and iodo-aromatics, manufacturers can achieve substantial cost savings on raw material procurement. Furthermore, the elimination of hazardous reagents reduces the regulatory burden and safety compliance costs associated with storage and handling. The one-pot nature of the reaction means fewer unit operations, less solvent usage, and reduced energy consumption, all of which contribute to a lower cost of goods sold (COGS). This economic efficiency makes the method particularly attractive for the commercial scale-up of complex pharmaceutical intermediates, where margin pressure is often intense.
- Cost Reduction in Manufacturing: The economic argument for this process is robust. Traditional methods often require expensive protecting group strategies to shield sensitive functionalities from harsh oxidants, adding steps and cost. This radical coupling method obviates the need for such protections, streamlining the synthetic sequence. Additionally, the high selectivity reduces the formation of difficult-to-remove impurities, lowering the cost of purification materials like silica gel and solvents. The use of catalytic amounts of phase transfer agents further optimizes reagent costs. Overall, the simplified workflow translates into a more lean manufacturing process that maximizes resource utilization and minimizes waste disposal expenses.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the pharmaceutical industry. This method relies on commodity chemicals—sodium dithionite and simple alkyl halides—that are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions (90°C, inert atmosphere) allows for flexible scheduling and easier troubleshooting in production facilities. Moreover, the high yields reported in the patent examples (ranging from 56% to 87% across diverse substrates) ensure consistent output volumes, enabling manufacturers to meet tight delivery deadlines. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis is never delayed by intermediate shortages.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this method stands out for its environmental profile. The absence of heavy metal catalysts and strong oxidants means the waste stream is significantly less toxic and easier to treat. The reaction generates minimal by-products, aligning with the principles of atom economy. From a scalability perspective, the exothermic nature of the radical generation is manageable under controlled heating, and the subsequent room temperature coupling step is easily adaptable to large reactors. The simple workup procedure involving aqueous quenching and organic extraction is standard in almost every chemical plant, requiring no specialized equipment. This ease of scale-up facilitates the transition from kilogram-scale development to multi-ton commercial production without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the nuances of new chemical technologies is essential for making informed sourcing decisions. The following questions address common inquiries regarding the scope, safety, and applicability of this radical coupling method. These answers are derived directly from the technical disclosures in patent CN109180572B and provide clarity on how this process fits into existing manufacturing frameworks. Whether you are evaluating this route for a new drug candidate or looking to optimize an existing supply chain, these insights offer a solid foundation for discussion with your technical partners.
Q: What are the primary advantages of this radical coupling method over traditional sulfonyl chloride routes?
A: This method eliminates the need for corrosive sulfonyl chlorides and unpleasant sulfuration reagents. It operates under mild conditions (90°C) with high selectivity, avoiding the oxidation of sensitive functional groups often seen in traditional strong oxidant methods.
Q: Is this synthesis method compatible with complex heterocyclic substrates?
A: Yes, the patent demonstrates excellent compatibility with diverse heterocycles including pyridine, pyrimidine, thiophene, and even complex nucleosides like adenosine derivatives, yielding products with purity up to 98%.
Q: How does this process impact commercial scalability and waste management?
A: The one-pot nature of the reaction simplifies the workflow significantly, reducing solvent usage and processing time. The use of inexpensive sodium dithionite and standard workup procedures (extraction/column) makes it highly suitable for large-scale commercial production with minimized environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfone Compound Supplier
The technological breakthroughs described in patent CN109180572B represent just one facet of the advanced capabilities available through NINGBO INNO PHARMCHEM. As a leading CDMO and fine chemical manufacturer, we possess the expertise to translate such innovative academic and patent literature into robust, commercial-grade processes. Our team of experienced chemists specializes in scaling diverse pathways, from initial gram-scale optimization to extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the transition from lab to plant requires meticulous attention to detail, particularly regarding stringent purity specifications and regulatory compliance. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive impurity profiling and structural characterization, ensuring that every batch of sulfone or sulfonamide intermediate meets the highest international standards.
We invite pharmaceutical and agrochemical companies to leverage our technical prowess to accelerate their development timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to streamline synthesis and reduce overall expenditure. We encourage potential clients to contact our technical procurement team to request specific COA data for similar compounds and to discuss route feasibility assessments for your proprietary targets. Together, we can navigate the complexities of modern chemical manufacturing, ensuring a secure, efficient, and high-quality supply of critical intermediates for your global operations.
