Advanced Pd-Catalyzed Carbonylation for Scalable Production of Bioactive 2,3-Dihydroquinolone Scaffolds
Advanced Pd-Catalyzed Carbonylation for Scalable Production of Bioactive 2,3-Dihydroquinolone Scaffolds
The 2,3-dihydroquinolone skeleton represents a privileged structure in medicinal chemistry, serving as the core framework for numerous bioactive molecules with potent therapeutic properties. As illustrated in the structural diversity of known pharmacophores, these compounds exhibit significant antitumor activity against human cell lines and possess powerful analgesic capabilities, making them highly sought-after targets for pharmaceutical development. 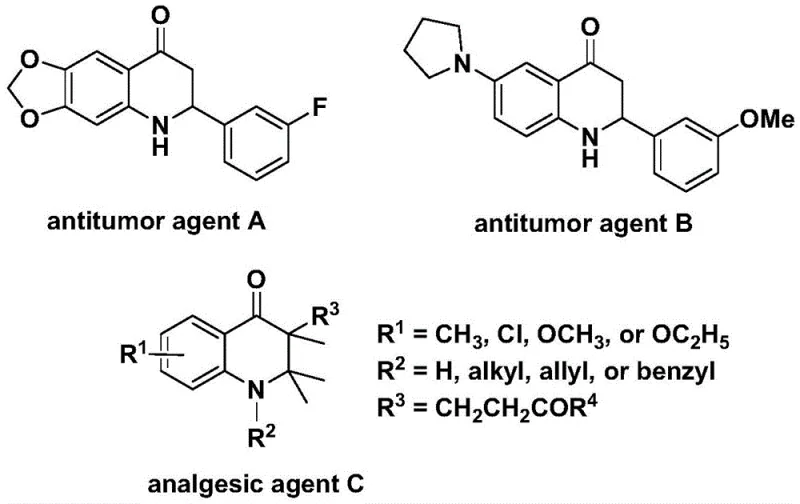 Recent advancements in synthetic methodology have focused on improving the efficiency and safety of constructing this heterocyclic core. Patent CN112239456B discloses a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds that leverages a transition metal palladium-catalyzed carbonylation reaction. This innovative approach utilizes N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials, offering a robust pathway that addresses many limitations of traditional synthetic routes while ensuring high reaction efficiency and broad substrate compatibility.
Recent advancements in synthetic methodology have focused on improving the efficiency and safety of constructing this heterocyclic core. Patent CN112239456B discloses a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds that leverages a transition metal palladium-catalyzed carbonylation reaction. This innovative approach utilizes N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials, offering a robust pathway that addresses many limitations of traditional synthetic routes while ensuring high reaction efficiency and broad substrate compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone ring system has relied on methods that often suffer from significant operational drawbacks and safety concerns. Traditional carbonylation reactions frequently require the direct use of carbon monoxide gas, which poses severe toxicity risks and necessitates specialized high-pressure equipment, thereby complicating the scale-up process for industrial applications. Furthermore, many existing protocols exhibit limited functional group tolerance, leading to side reactions or decomposition when complex substrates are employed. The reliance on harsh reaction conditions, such as extreme temperatures or strong acidic/basic environments, can also result in poor regioselectivity and difficult purification processes. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to deliver high-purity materials consistently.
The Novel Approach
The methodology outlined in the patent introduces a transformative solution by employing a palladium-catalyzed system that utilizes a solid carbon monoxide surrogate, specifically 1,3,5-trimesic acid phenyl ester (TFBen). This strategic substitution eliminates the hazards associated with gaseous CO, allowing the reaction to proceed under much milder and safer conditions. The process involves the reaction of N-pyridine sulfonyl-o-iodoaniline with olefins in the presence of a palladium catalyst and a phosphine ligand within an organic solvent. 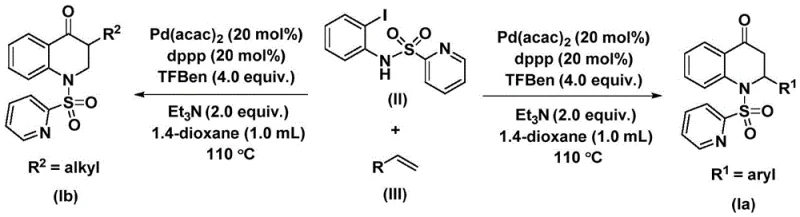 This novel approach not only simplifies the operational procedure but also significantly enhances the reaction applicability, enabling the synthesis of a wide array of 2-aryl and 3-alkyl substituted derivatives. The ability to tune the substrate design allows for the precise installation of diverse functional groups, thereby widening the practicability of the method for generating complex libraries of bioactive compounds.
This novel approach not only simplifies the operational procedure but also significantly enhances the reaction applicability, enabling the synthesis of a wide array of 2-aryl and 3-alkyl substituted derivatives. The ability to tune the substrate design allows for the precise installation of diverse functional groups, thereby widening the practicability of the method for generating complex libraries of bioactive compounds.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The catalytic cycle begins with the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent transformations. Following this activation, the carbon monoxide molecule, which is released in situ from the decomposition of the 1,3,5-trimesic acid phenyl ester additive, inserts into the aryl-palladium bond. This insertion forms an acyl-palladium intermediate, effectively building the carbonyl functionality directly into the growing molecular framework. The use of the pyridine sulfonyl group on the nitrogen atom plays a dual role: it acts as a directing group to facilitate the initial activation and subsequently participates in the cyclization event to close the heterocyclic ring.
Subsequently, the olefin substrate coordinates with the palladium center of the acyl-palladium intermediate and undergoes migratory insertion. This step creates a new carbon-carbon bond and generates a palladium-alkyl species. The final stage of the catalytic cycle involves a reductive elimination step, which releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst to continue the cycle. 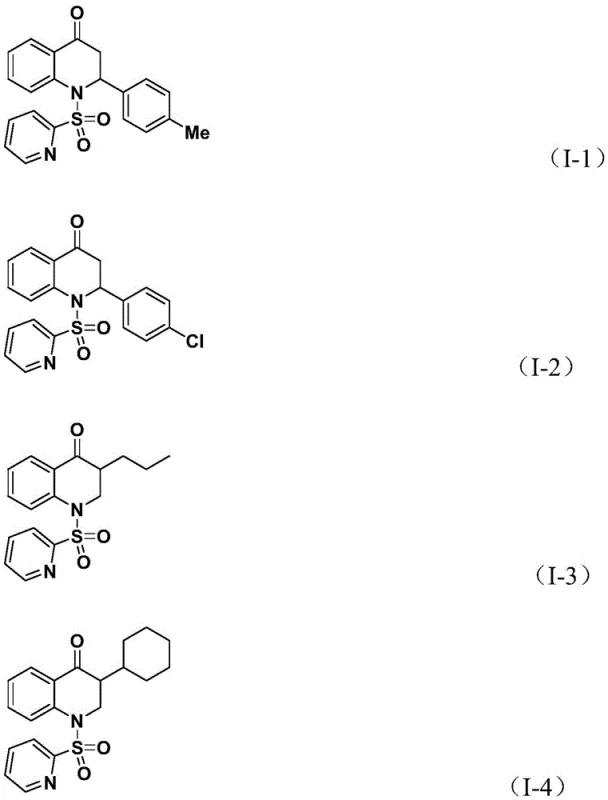 This mechanism ensures high atom economy and minimizes the formation of unwanted byproducts. The rigorous control over the catalytic cycle allows for excellent impurity control, as the specific coordination environment provided by the dppp ligand and the pyridine sulfonyl moiety directs the reaction pathway selectively towards the desired heterocyclic product, reducing the burden on downstream purification processes.
This mechanism ensures high atom economy and minimizes the formation of unwanted byproducts. The rigorous control over the catalytic cycle allows for excellent impurity control, as the specific coordination environment provided by the dppp ligand and the pyridine sulfonyl moiety directs the reaction pathway selectively towards the desired heterocyclic product, reducing the burden on downstream purification processes.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
To implement this synthesis effectively, precise control over reaction parameters and reagent stoichiometry is essential. The protocol recommends using bis(acetylacetone)palladium as the catalyst precursor and 1,3-bis(diphenylphosphino)propane (dppp) as the ligand, typically in a molar ratio that ensures sufficient catalytic turnover without excessive metal loading. The reaction is conducted in an aprotic organic solvent such as 1,4-dioxane, which effectively dissolves the reactants and promotes the catalytic cycle. Triethylamine is added as a base to neutralize acidic byproducts and facilitate the reaction progress. The detailed standardized synthesis steps, including specific mixing orders and workup procedures, are provided in the guide below to ensure reproducibility and high yield.
- Combine bis(acetylacetone)palladium, dppp ligand, triethylamine, 1,3,5-trimesic acid phenyl ester, N-pyridine sulfonyl-o-iodoaniline, and the chosen olefin substrate in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100-120 °C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic benefits that directly impact the bottom line and operational resilience. The shift from hazardous gaseous reagents to stable solid surrogates drastically simplifies the safety infrastructure required for production, reducing regulatory compliance burdens and insurance costs associated with handling toxic gases. Moreover, the use of readily available and inexpensive olefin starting materials, combined with a robust catalyst system, ensures a stable supply of raw materials that is less susceptible to market volatility. This stability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and specialized gas handling systems leads to substantial capital expenditure savings and lower operational overheads. By utilizing a solid CO surrogate, the process avoids the complex logistics and safety measures required for carbon monoxide, resulting in significantly reduced processing costs. Additionally, the high reaction efficiency and selectivity minimize waste generation and reduce the consumption of expensive purification materials, further driving down the cost of goods sold for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The compatibility of this method with a broad range of commercially available olefins and aryl iodides ensures that raw material sourcing remains flexible and resilient. Suppliers can easily switch between different substrate variants without retooling the entire production line, allowing for rapid response to changing market demands. The robustness of the palladium catalytic system also means that batch-to-batch variability is minimized, ensuring consistent quality and reliable delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The reaction conditions, operating at moderate temperatures between 100-120 °C, are highly amenable to scale-up from gram-level laboratory synthesis to multi-ton commercial production. The use of common organic solvents like dioxane facilitates solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. This scalability ensures that the technology can support the growing demand for complex heterocyclic compounds without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. Understanding these details is vital for R&D teams evaluating the feasibility of integrating this technology into their existing workflows. The answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing a reliable foundation for process development decisions.
Q: What is the primary advantage of using 1,3,5-trimesic acid phenyl ester in this synthesis?
A: It serves as a safe and solid carbon monoxide surrogate, eliminating the need for handling toxic CO gas while efficiently releasing CO in situ for the carbonylation step.
Q: Can this method accommodate both aryl and alkyl substituted olefins?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully synthesizing both 2-aryl and 3-alkyl substituted 2,3-dihydroquinolone compounds with high yields.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds in 1,4-dioxane at 110 °C for 48 hours using Pd(acac)2 and dppp as the catalytic system, with triethylamine as a base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,3-dihydroquinolone derivative adheres to the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced Pd-catalyzed technology for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your supply chain efficiency.
