Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe methodologies for constructing complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN112239456B, which discloses a highly efficient preparation method for substituted 2,3-dihydroquinolone compounds. This technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. The 2,3-dihydroquinolone skeleton is a privileged structure in medicinal chemistry, widely recognized for its presence in molecules exhibiting potent biological activities, including significant anti-cancer properties against human cells and powerful analgesic effects. As illustrated in the structural examples of known bioactive agents, the versatility of this core structure makes it a critical target for synthetic chemists aiming to develop new therapeutic candidates.
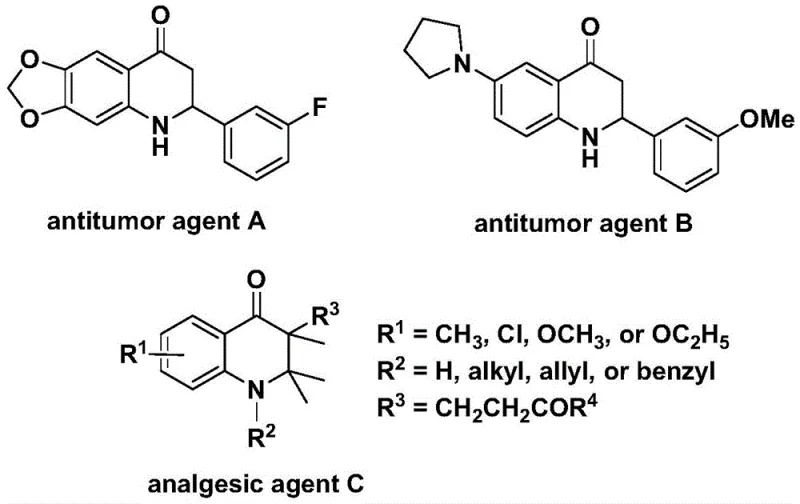
For R&D directors and process chemists, the ability to access these scaffolds through a direct and efficient carbonylation pathway represents a substantial advancement over traditional multi-step syntheses. The patent highlights that while carbonylation reactions are a cornerstone for synthesizing carbonyl-containing compounds, their application specifically to 2,3-dihydroquinolone skeletons has been relatively limited until now. This new method addresses that gap by offering a protocol that is not only operationally simple but also compatible with a wide range of functional groups, thereby widening the practical applicability for generating diverse libraries of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing carbonyl heterocycles like 2,3-dihydroquinolones has often relied on classical cyclization strategies that may suffer from harsh reaction conditions, limited substrate scope, or the requirement for pre-functionalized starting materials that are expensive or difficult to prepare. Many existing literature reports on synthetic methods for these skeletons indicate that while various approaches exist, they often lack the generality required for rapid analog generation. Furthermore, traditional carbonylation methods frequently necessitate the use of gaseous carbon monoxide, which poses significant safety hazards and requires specialized high-pressure equipment, creating barriers for widespread adoption in standard laboratory or pilot plant settings. The complexity of managing toxic gases and the potential for side reactions under high pressure can lead to lower yields and difficult purification processes, ultimately impacting the cost-effectiveness and timeline of drug discovery programs.
The Novel Approach
In contrast, the methodology described in patent CN112239456B introduces a transformative approach by employing a solid carbon monoxide substitute, specifically 1,3,5-trimesic acid phenol ester, which releases CO in situ under the reaction conditions. This innovation eliminates the need for handling dangerous CO gas, drastically simplifying the operational setup and enhancing safety profiles for chemical manufacturing. The reaction proceeds smoothly in common organic solvents like dioxane at moderate temperatures ranging from 100°C to 120°C. The versatility of this system is exemplified by its ability to synthesize both 2-aryl and 3-alkyl substituted 2,3-dihydroquinolone compounds simply by varying the olefin substrate. The structural diversity achievable through this method is impressive, accommodating aryl groups with various substituents such as methyl, tert-butyl, methoxy, fluorine, or chlorine at ortho, meta, or para positions, as well as linear, branched alkyl, and silyl groups. This broad substrate tolerance ensures that chemists can rapidly explore structure-activity relationships without being constrained by synthetic limitations.
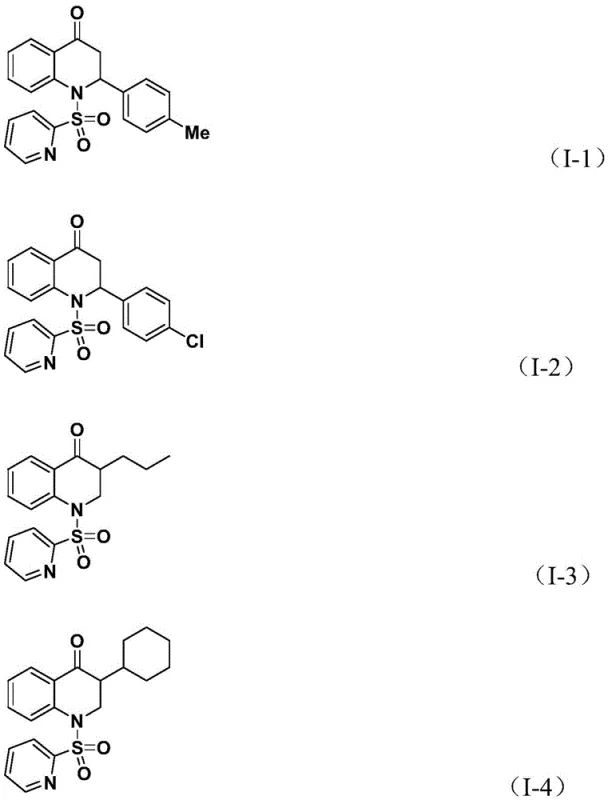
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
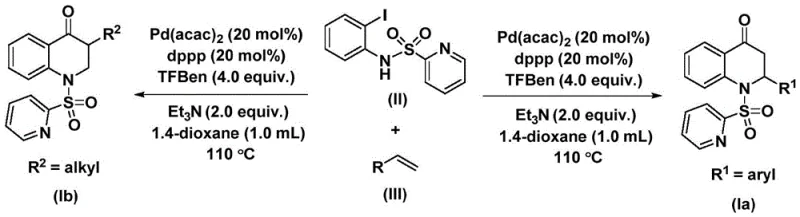
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing the process for commercial scale-up. The reaction is initiated by the oxidative addition of the palladium catalyst, specifically palladium bis(acetylacetonate) coordinated with the 1,3-bis(diphenylphosphino)propane (dppp) ligand, into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is the entry point for the carbonylation sequence. Subsequently, carbon monoxide, which is continuously released from the decomposition of the 1,3,5-trimesic acid phenol ester additive, inserts into the aryl-palladium bond to form an acyl-palladium intermediate. This in situ generation of CO ensures a steady, controlled concentration of the carbonyl source, minimizing side reactions associated with excess gas pressure.
Following the formation of the acyl-palladium species, the olefin substrate coordinates to the metal center and undergoes migratory insertion, resulting in a palladium-alkyl intermediate. This step effectively builds the carbon framework of the dihydroquinolone ring system. The catalytic cycle is completed by a reductive elimination step, which releases the final substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) species to continue the cycle. The choice of triethylamine as an additive plays a critical role in neutralizing acidic byproducts and facilitating the turnover of the catalyst. The entire process is depicted in the general reaction scheme below, which clearly outlines the convergence of the iodoaniline derivative and the olefin to form the target heterocycle with high atom economy and efficiency.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction parameters to maximize yield and purity. The patent provides a robust protocol where the molar ratio of the palladium catalyst to the ligand and the CO source is optimized to ensure high conversion rates. Typically, the reaction is conducted in anhydrous dioxane, which has been identified as the preferred solvent due to its ability to fully dissolve the reactants and promote the catalytic cycle effectively. The post-treatment process is notably straightforward, involving simple filtration followed by silica gel mixing and standard column chromatography purification. This simplicity in workup is a significant advantage for process chemistry, as it reduces the time and resources needed for isolation. For detailed operational parameters and specific stoichiometric ratios validated across multiple examples, please refer to the standardized synthesis guide below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and the chosen olefin in an organic solvent such as dioxane.
- Heat the reaction mixture to a temperature between 100°C and 120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target substituted 2,3-dihydroquinolone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers compelling advantages that directly address common pain points in the sourcing of complex pharmaceutical intermediates. The reliance on commercially available starting materials, such as readily accessible olefins and easily synthesized N-pyridine sulfonyl-o-iodoaniline derivatives, ensures a stable and resilient supply chain. Unlike methods requiring exotic or custom-synthesized precursors with long lead times, the inputs for this process can be sourced from multiple vendors, mitigating the risk of supply disruptions. Furthermore, the elimination of high-pressure gas equipment reduces the capital expenditure required for manufacturing infrastructure, making it easier for contract manufacturing organizations (CMOs) to adopt this technology without significant retrofitting.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of inexpensive and abundant raw materials combined with a highly efficient catalytic system. By replacing hazardous gaseous carbon monoxide with a solid surrogate, the method removes the need for specialized high-pressure reactors and the associated safety protocols, which translates to significantly reduced operational costs. The high reaction efficiency and broad substrate compatibility mean that fewer batches are rejected due to poor yields, leading to substantial cost savings in raw material consumption. Additionally, the simplified post-treatment procedure minimizes solvent usage and labor hours required for purification, further driving down the overall cost of goods sold (COGS) for these valuable intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the availability of reagents. The tolerance for various functional groups allows manufacturers to utilize a wide range of olefin feedstocks, providing flexibility in sourcing strategies. If a specific olefin becomes unavailable or price-volatile, alternative substrates with similar electronic or steric properties can often be substituted without compromising the integrity of the synthetic route. This adaptability ensures that production schedules remain uninterrupted, securing reliable delivery timelines for downstream API manufacturers who depend on these critical building blocks for their drug development pipelines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having already been demonstrated at the gram level with clear pathways for expansion to industrial tonnage. The use of a solid CO source not only enhances safety but also aligns with green chemistry principles by reducing the risk of toxic gas emissions. The reaction operates in standard organic solvents that can be recovered and recycled, minimizing waste generation. The straightforward workup involving filtration and chromatography is easily adaptable to larger scales, ensuring that environmental compliance is maintained without sacrificing throughput. This combination of safety, efficiency, and scalability makes the method an ideal candidate for sustainable commercial production of high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope, safety profile, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process development decisions. Understanding these nuances is essential for integrating this synthetic route into existing manufacturing workflows and maximizing its potential for cost-effective production.
Q: What are the key advantages of this carbonylation method over traditional synthesis routes?
A: This method utilizes a solid carbon monoxide substitute (1,3,5-trimesic acid phenol ester) instead of hazardous high-pressure CO gas, significantly enhancing operational safety. Additionally, it demonstrates excellent substrate compatibility, allowing for the synthesis of both 2-aryl and 3-alkyl substituted derivatives with high efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is simple to operate, uses cheap and easily obtainable raw materials, and has been expanded to the gram level, providing a strong foundation for industrial large-scale production and application.
Q: What types of substituents are tolerated on the olefin substrate?
A: The reaction shows broad tolerance for various functional groups. The olefin substrate (R group) can be a substituted or unsubstituted aryl group (with methyl, tert-butyl, methoxy, fluorine, or chlorine substituents), a linear or branched alkyl group, or even a silyl group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate seamlessly into reliable industrial supply. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging technologies such as the palladium-catalyzed carbonylation described in CN112239456B, we can offer our partners a competitive edge through superior quality and consistent availability of complex heterocyclic building blocks.
We invite you to collaborate with us to explore how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data for our available inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your trusted partner in bringing next-generation therapeutics to market faster and more efficiently.
