Advanced Photocatalytic Synthesis of Heterocyclic Cyclopropanes for Scalable Pharmaceutical Manufacturing
Advanced Photocatalytic Synthesis of Heterocyclic Cyclopropanes for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust methodologies to construct complex molecular architectures, particularly strained ring systems like cyclopropanes, which are prevalent in bioactive molecules. Patent CN112898202B introduces a groundbreaking preparation method for heterocyclic cyclopropane compounds that addresses significant limitations in current synthetic strategies. This technology leverages visible light redox catalysis to fuse cyclopropane rings onto heterocyclic scaffolds, such as indoles, in a single operational step. Unlike traditional approaches that rely on hazardous carbene intermediates and extreme reaction conditions, this novel process operates under mild, ambient conditions using commercially available raw materials. For R&D directors and process chemists, this represents a paradigm shift towards greener, more efficient synthesis of high-purity pharmaceutical intermediates. The method demonstrates exceptional functional group tolerance and operational simplicity, making it an ideal candidate for both laboratory discovery and commercial scale-up.
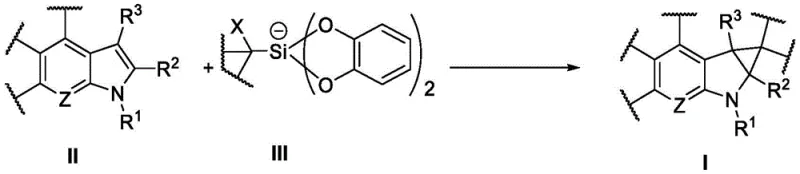
The core innovation lies in the use of a specific silicon-based reagent containing a cation and an anionic structural fragment, which acts as a radical precursor under visible light irradiation. When combined with a heterocyclic substrate containing specific structural fragments (Formula II) in the presence of a photocatalyst, the system efficiently generates the target heterocyclic cyclopropane (Formula I). This transformation is not merely a laboratory curiosity but a viable industrial process characterized by high yields, easy purification of intermediates, and environmental friendliness. By eliminating the need for cryogenic temperatures or pyrophoric reagents, this patent provides a reliable pathway for the cost reduction in pharmaceutical intermediate manufacturing, directly addressing the pain points of safety and scalability in modern drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropane-fused heterocycles has been fraught with challenges that hinder efficient production. Conventional methodologies often depend on the generation of highly reactive carbene intermediates, as noted in prior art such as J. Am. Chem. Soc. 2017, 139, 7697-7700. These carbene-based routes typically necessitate harsh reaction conditions, including the use of strong bases, toxic metal catalysts, or unstable diazo compounds that pose significant safety risks on a large scale. Furthermore, the sensitivity of these intermediates often leads to poor selectivity, resulting in complex impurity profiles that are difficult and costly to remove during downstream processing. The requirement for stringent anhydrous conditions and low temperatures further exacerbates energy consumption and equipment costs. For supply chain managers, these factors translate into longer lead times and higher vulnerability to production disruptions, as the handling of hazardous reagents requires specialized infrastructure and rigorous safety protocols that are not universally available in standard manufacturing facilities.
The Novel Approach
In stark contrast, the method disclosed in CN112898202B utilizes a visible light-driven photocatalytic strategy that fundamentally simplifies the synthetic workflow. By employing a silicon-based radical precursor and a photocatalyst, the reaction proceeds smoothly at room temperature under inert atmosphere, driven solely by blue or white LED light sources. This approach bypasses the need for unstable carbenes entirely, replacing them with stable, commercially available starting materials that are easy to handle and store. The reaction conditions are remarkably mild, typically occurring in common organic solvents like DMSO or acetonitrile mixtures, which facilitates straightforward workup procedures involving simple aqueous washes and column chromatography. This shift from harsh chemical activation to gentle photochemical activation not only enhances operator safety but also significantly reduces the environmental footprint of the synthesis. For procurement teams, this means accessing a supply of critical intermediates that are produced via a safer, more sustainable, and economically viable process, ensuring greater supply chain continuity.
Mechanistic Insights into Visible Light Photocatalytic Cyclization
The mechanistic elegance of this transformation lies in the photoredox cycle initiated by the excitation of the photocatalyst. Upon absorption of visible light, the photocatalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the silicon-based reagent (Formula III). This interaction triggers the homolytic cleavage of the carbon-silicon bond, generating a reactive carbon-centered radical species. This radical then adds selectively to the electron-rich double bond of the heterocyclic substrate (Formula II), forming a new carbon-carbon bond and a transient radical intermediate on the heterocyclic ring. Subsequent intramolecular radical cyclization closes the three-membered cyclopropane ring, followed by oxidation and deprotonation steps to restore aromaticity and yield the final fused product. The use of specific iridium complexes, such as Ir(dFCF3ppy)2(dtbbpy)PF6, or organic photocatalysts like 4CzIPN, ensures high quantum efficiency and robust turnover numbers, allowing the reaction to proceed with low catalyst loading (e.g., 3.0 mol%).
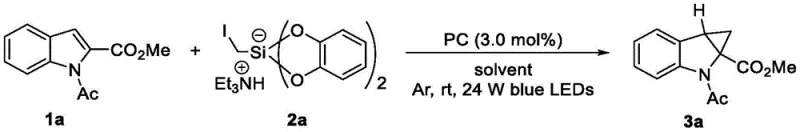
From an impurity control perspective, the mild nature of the photocatalytic conditions is a distinct advantage. High-energy thermal pathways that often lead to decomposition or polymerization of sensitive functional groups are avoided. The radical intermediates generated are short-lived and highly selective, minimizing side reactions such as over-alkylation or ring-opening. The patent data indicates that a wide range of substituents, including halogens (F, Cl, Br), esters, amides, and ethers, are well-tolerated on the heterocyclic core without significant degradation. This chemoselectivity is crucial for R&D directors aiming to synthesize diverse libraries of analogs for structure-activity relationship (SAR) studies. Furthermore, the ability to fine-tune the reaction outcome by adjusting solvent ratios (e.g., DMSO/CH3CN) and light intensity provides an additional layer of process control, ensuring consistent quality and high purity specifications essential for regulatory compliance in pharmaceutical applications.
How to Synthesize Heterocyclic Cyclopropane Efficiently
The practical implementation of this synthesis is designed for ease of execution in both research and production settings. The protocol involves dissolving the heterocyclic substrate and the silicon reagent in a degassed solvent system, adding the photocatalyst, and irradiating the mixture with standard LED arrays. The reaction progress can be conveniently monitored using standard analytical techniques such as TLC or HPLC. A key feature of the optimized procedure is the分批 addition (split addition) of reagents; specifically, a portion of the silicon reagent and photocatalyst is added after an initial period of irradiation (e.g., 24 hours) to drive the reaction to completion and maximize yield. This simple modification prevents the accumulation of side products and ensures high conversion rates. Following the reaction, the workup involves dilution with ethyl acetate, washing with aqueous base and water, drying over sodium sulfate, and purification via column chromatography. This streamlined process eliminates the need for complex distillation or crystallization steps often required in traditional methods.
- Prepare the reaction mixture by combining the heterocyclic alkyl derivative (Formula II), the silicon reagent containing the cation and anionic structural fragment (Formula III), and a photocatalyst such as Ir(dFCF3ppy)2(dtbbpy)PF6 in an organic solvent like DMSO/CH3CN.
- Degas the solution under inert gas (Argon) and irradiate with blue or white LEDs (20W-40W) at room temperature for approximately 24 hours to initiate the radical cyclization.
- Supplement the reaction with additional silicon reagent and photocatalyst, continue irradiation for another 24 hours, then perform standard workup including washing, drying, and column chromatography to isolate the target cyclopropane product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational resilience, this photocatalytic technology offers compelling commercial benefits that extend beyond mere chemical novelty. The shift to a visible-light-mediated process fundamentally alters the cost structure and risk profile of producing heterocyclic cyclopropanes. By relying on abundant and stable starting materials rather than specialized, hazardous reagents, the supply chain becomes more robust and less susceptible to market volatility. The elimination of extreme temperature requirements (cryogenic cooling or high heat) translates directly into reduced energy consumption and lower capital expenditure on specialized reactor equipment. Moreover, the simplified workup and purification procedures reduce solvent usage and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals. These factors collectively contribute to a more agile and cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the use of inexpensive, commercially available raw materials and the avoidance of costly transition metal catalysts often required in traditional cross-coupling or carbene insertion reactions. While photocatalysts like Iridium complexes are used, they are employed in very low loadings (catalytic amounts), and the overall process efficiency minimizes material loss. The mild conditions also reduce the wear and tear on manufacturing equipment, lowering maintenance costs over time. Additionally, the high selectivity of the reaction reduces the burden on purification teams, saving significant time and resources associated with removing complex impurities, thereby lowering the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing reliability is a critical metric for procurement managers, and this synthesis excels by utilizing commodity chemicals. The heterocyclic substrates and silicon reagents are widely available from multiple global suppliers, reducing the risk of single-source dependency. The stability of these reagents allows for long-term storage without significant degradation, enabling manufacturers to maintain strategic stockpiles without fear of spoilage. Furthermore, the operational simplicity of the reaction means it can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without requiring extensive re-validation or specialized training, ensuring consistent supply continuity even in the face of logistical disruptions.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but advancements in flow chemistry and LED technology have made this highly feasible. The patent describes conditions that are inherently scalable, operating at ambient pressure and temperature, which simplifies the engineering requirements for large-scale reactors. From an environmental standpoint, the process is greener, generating less hazardous waste compared to carbene-based methods. The use of common solvents that can be readily recycled further enhances the sustainability profile. This alignment with green chemistry principles not only mitigates regulatory risks but also appeals to end-clients who prioritize environmentally responsible sourcing in their supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for making informed procurement and development decisions. The following questions address common inquiries regarding the scope, safety, and scalability of the technology described in CN112898202B. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical stakeholders evaluating this pathway for their own pipelines.
Q: What are the advantages of this photocatalytic method over traditional carbene routes?
A: Traditional methods often require harsh conditions and unstable carbene intermediates. This patented photocatalytic approach operates at room temperature under visible light, utilizing commercially available starting materials for a safer and more efficient one-step synthesis.
Q: Which photocatalysts are most effective for this transformation?
A: The patent highlights several effective photocatalysts, including Iridium complexes like Ir(dFCF3ppy)2(dtbbpy)PF6 and organic catalysts like 4CzIPN, which provide high yields under mild visible light irradiation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for industrialization. It uses common organic solvents, operates at ambient temperatures, and avoids expensive or hazardous reagents, making it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in modern drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112898202B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with the necessary photochemical reactors and rigorous QC labs to handle complex heterocyclic syntheses with stringent purity specifications. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry, leveraging our technical expertise to optimize yield and minimize impurities at every stage of production.
We invite you to explore how this cutting-edge synthesis method can enhance your project's efficiency and cost-effectiveness. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Whether you need rapid gram-scale quantities for preclinical studies or multi-ton support for commercial launch, we encourage you to contact us to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your journey from molecule to medicine with reliable, scalable, and economically superior chemical solutions.
