Advanced Photocatalytic Synthesis of Heterocyclic Cyclopropanes for Commercial Scale-Up
Advanced Photocatalytic Synthesis of Heterocyclic Cyclopropanes for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct strained ring systems, particularly cyclopropanes fused to heterocycles, due to their prevalence in bioactive molecules. Patent CN112898202B introduces a groundbreaking approach to synthesizing these valuable scaffolds through a visible-light mediated photoredox catalysis strategy. This technology represents a significant departure from classical synthetic routes, offering a pathway that is not only operationally simple but also environmentally benign and highly efficient. By leveraging the power of modern photocatalysis, this method enables the direct functionalization of readily available indole derivatives with specialized silicon reagents, bypassing the need for hazardous precursors. For R&D directors and procurement managers alike, understanding the nuances of this patent is critical for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity compounds with consistent quality.
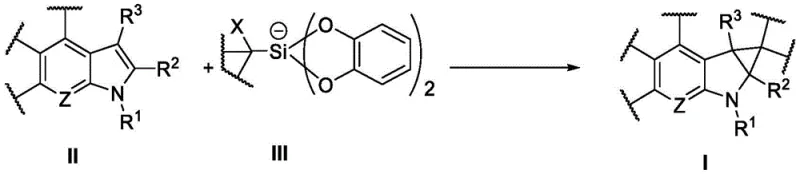
This innovation addresses a long-standing challenge in organic synthesis: the efficient construction of the cyclopropane ring on a heterocyclic core without compromising sensitive functional groups. The core transformation involves the reaction of a compound containing the structural fragment shown in Formula II with a cationic and anionic silicon reagent depicted in Formula III. Under the influence of a photocatalyst and visible light, these precursors undergo a sophisticated radical cascade to yield the target heterocyclic cyclopropane compound of Formula I. The versatility of this reaction is underscored by the broad scope of substituents tolerated, ranging from esters and ketones to sulfonamides, making it a versatile tool for the cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of heterocyclic cyclopropanes has relied heavily on carbene chemistry, which presents substantial hurdles for industrial application. Traditional protocols often necessitate the generation of highly reactive carbene intermediates, typically requiring harsh reaction conditions such as strong bases, elevated temperatures, or the use of toxic heavy metal catalysts in stoichiometric amounts. These aggressive conditions frequently lead to poor chemoselectivity, resulting in complex impurity profiles that are difficult and costly to remove during downstream processing. Furthermore, the instability of carbene species often limits the substrate scope, preventing the incorporation of sensitive functional groups that are common in advanced drug candidates. For supply chain heads, these factors translate into longer lead times, lower overall yields, and increased safety risks associated with handling hazardous reagents on a commercial scale.
The Novel Approach
In stark contrast, the methodology disclosed in CN112898202B utilizes a mild, visible-light driven photoredox catalytic cycle that operates effectively at room temperature. This novel approach eliminates the need for unstable carbene intermediates, instead generating reactive radical species through the single-electron transfer (SET) capabilities of the photocatalyst. The use of commercially available heterocyclic alkyl raw materials and stable silicon-based reagents ensures a safer and more predictable reaction profile. The conditions are remarkably gentle, typically employing blue or white LED light sources and common organic solvents like DMSO or acetonitrile mixtures. This shift not only simplifies the operational complexity but also significantly enhances the purity of the crude product, thereby reducing the burden on purification teams and facilitating the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Visible-Light Photoredox Catalysis
The success of this transformation lies in the intricate interplay between the photocatalyst, the silicon reagent, and the heterocyclic substrate. Upon irradiation with visible light, the photocatalyst—such as the iridium complex Ir(dFCF3ppy)2(dtbbpy)PF6 or the organic dye 4CzIPN—is excited to a high-energy state. This excited species acts as a potent oxidant or reductant, initiating the cleavage of the carbon-silicon bond in the reagent to generate a carbon-centered radical. This radical subsequently adds to the electron-rich double bond of the indole system, triggering a cyclization event that forms the strained cyclopropane ring. The catalytic cycle is closed through a subsequent electron transfer and proton loss, regenerating the ground-state catalyst for further turnover. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substrates.
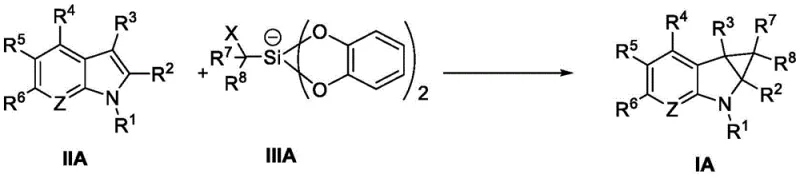
Impurity control is inherently built into this mechanistic pathway. Unlike thermal reactions that might promote non-selective background decomposition, the photochemical activation is highly specific to the catalyst-substrate interaction. The patent data indicates that the reaction proceeds with high regioselectivity, favoring the formation of the desired fused cyclopropane structure over potential oligomerization or polymerization byproducts. Additionally, the ability to tune the redox potential by selecting different photocatalysts allows for precise control over the reaction kinetics. For instance, the use of mixed solvent systems like DMSO and acetonitrile in specific ratios (e.g., 9:1) further stabilizes the ionic intermediates and enhances solubility, ensuring a clean reaction profile that meets stringent purity specifications required for API synthesis.
How to Synthesize Heterocyclic Cyclopropane Efficiently
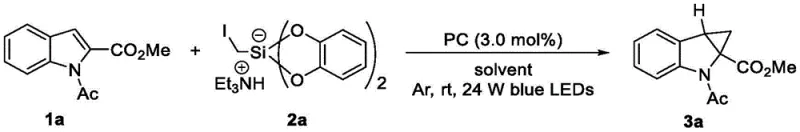
Implementing this synthesis requires careful attention to the addition sequence and light source intensity to maximize yield and reproducibility. The patent outlines a protocol where reagents are added in portions to maintain optimal concentration gradients throughout the reaction timeline. Typically, the heterocyclic derivative and the silicon reagent are dissolved in the solvent system under an inert atmosphere, followed by the addition of the photocatalyst. The reaction vessel is then subjected to irradiation, often using 24W blue LEDs, for a defined period before a second charge of reagents is introduced to drive the conversion to completion. This controlled addition strategy minimizes side reactions and ensures high conversion rates.
- Combine the heterocyclic substrate (e.g., N-acetyl indole ester), the silicon reagent (iodonium silicate salt), and the photocatalyst (e.g., Ir complex) in a sealed tube with DMSO/MeCN solvent.
- Degas the mixture under vacuum/inert gas and irradiate with blue LEDs at room temperature for 24 hours.
- Add a second portion of reagents, continue irradiation, then perform standard aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology offers compelling strategic advantages beyond mere chemical elegance. The primary benefit lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for cryogenic conditions or expensive, air-sensitive reagents typical of carbene chemistry, the facility requirements for production are significantly lowered. This accessibility allows for manufacturing in standard glass-lined reactors equipped with LED arrays, rather than specialized high-pressure or low-temperature vessels, leading to substantial cost savings in capital investment and energy consumption.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the high atom economy and the use of earth-abundant or low-loading catalysts. Traditional methods often suffer from low yields due to decomposition, necessitating larger batches of starting materials to achieve the same output. In contrast, this photocatalytic route demonstrates high yields across a broad range of substrates, meaning less raw material waste and lower cost of goods sold (COGS). Furthermore, the simplified workup procedure, involving standard aqueous washes and column chromatography, reduces solvent usage and labor hours, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on commercially available starting materials. The indole derivatives and silicon reagents specified in the patent are either commodity chemicals or easily synthesized from bulk precursors, mitigating the risk of supply bottlenecks associated with exotic reagents. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining steady inventory levels and meeting the just-in-time delivery demands of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles. The absence of heavy metal waste streams (common in stoichiometric carbene methods) and the use of visible light as the energy source significantly reduce the environmental footprint of the synthesis. Scalability is facilitated by the homogeneous nature of the reaction and the ease of heat management at room temperature. As regulatory pressures regarding waste disposal and energy efficiency intensify, adopting such an environmentally friendly process positions the supply chain for long-term compliance and sustainability, avoiding future liabilities associated with hazardous waste treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction scope, catalyst selection, and process optimization. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: What are the advantages of this photocatalytic method over traditional carbene routes?
A: Traditional methods often require harsh conditions and unstable carbene intermediates. This patent describes a mild, visible-light driven process that operates at room temperature with high yields and easier purification.
Q: Which photocatalysts are most effective for this transformation?
A: The patent highlights Iridium complexes such as Ir(dFCF3ppy)2(dtbbpy)PF6 and organic catalysts like 4CzIPN as highly effective options for driving the redox cycle.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method uses commercially available raw materials, mild conditions, and simple workup procedures, making it highly suitable for industrial scale-up and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Cyclopropane Supplier
As the demand for complex heterocyclic scaffolds continues to rise in drug discovery, partnering with a technically proficient manufacturer is essential. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the nuances of photoredox catalysis and can adeptly navigate the optimization of reaction parameters to ensure stringent purity specifications are met for every batch. With our rigorous QC labs and state-of-the-art pilot facilities, we are uniquely positioned to translate this patented laboratory methodology into a robust industrial process.
We invite you to collaborate with us to leverage this advanced synthesis for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality.
