Industrial Scale-Up of Apalutamide: A Safer, Cyanide-Free Synthetic Route for Global Supply Chains
The pharmaceutical landscape for treating castration-resistant prostate cancer has been significantly advanced by the development of Apalutamide, a potent androgen receptor inhibitor. However, the commercial viability of this critical medication heavily depends on the efficiency and safety of its synthetic manufacturing process. Patent CN108069869B introduces a groundbreaking preparation method that addresses long-standing safety and scalability issues found in earlier synthetic routes. This innovation focuses on a novel condensation strategy that constructs the core spiro-cyclic structure without relying on hazardous cyanide reagents or energy-intensive microwave irradiation. For global supply chain leaders, this represents a pivotal shift towards more sustainable and compliant API intermediate production. The structural integrity of the final molecule, as depicted in the chemical diagram below, relies on precise control over the spiro-cyclization step to ensure therapeutic efficacy.
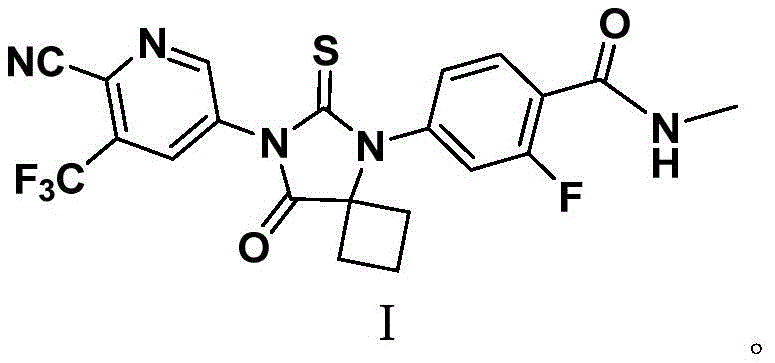
As a reliable Apalutamide intermediate supplier, understanding the nuances of this patent is essential for securing a stable supply of high-purity active pharmaceutical ingredients. The method described not only enhances operator safety but also streamlines the purification process, which is a critical factor in reducing overall manufacturing costs. By transitioning away from toxic reagents, manufacturers can mitigate regulatory hurdles associated with hazardous material transport and disposal. This technical insight report delves deep into the mechanistic advantages and commercial implications of this new route, providing R&D directors and procurement managers with the data needed to optimize their sourcing strategies. The ability to produce complex heterocyclic intermediates under mild conditions is a key differentiator in the competitive fine chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for Apalutamide, such as those disclosed in CN101454002A, have historically relied on the use of sodium cyanide to introduce the cyano group essential for the pyridine ring structure. This reliance on highly toxic cyanide salts presents severe challenges for industrial scale-up, including stringent safety protocols, expensive waste treatment facilities, and significant environmental compliance burdens. Furthermore, some existing methods require microwave irradiation to drive the reaction to completion, a condition that is notoriously difficult to replicate efficiently in large-scale batch reactors. The combination of toxic reagents and specialized energy inputs creates a bottleneck for cost reduction in API manufacturing, limiting the ability of suppliers to meet growing global demand. Additionally, the multi-step nature of older routes often leads to cumulative yield losses and increased impurity profiles, complicating the downstream purification process.
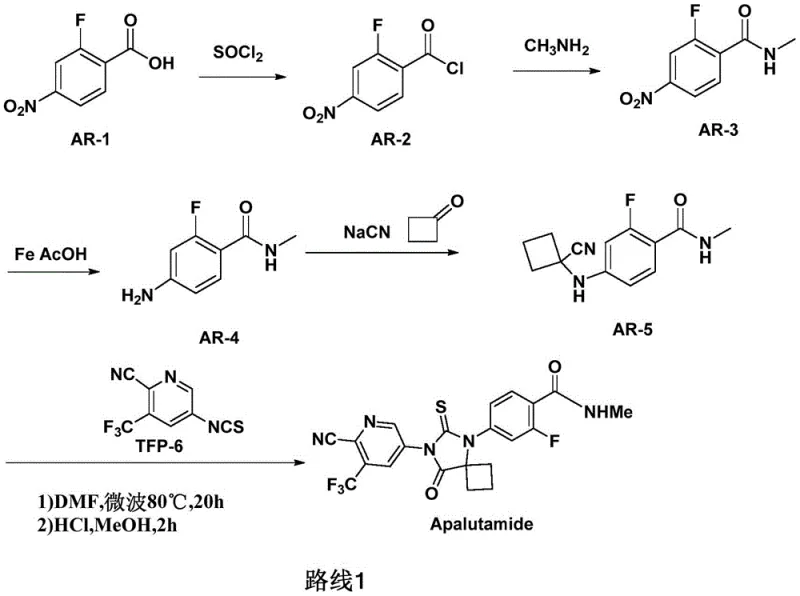
The Novel Approach
The novel approach detailed in the patent data fundamentally reimagines the construction of the Apalutamide scaffold by utilizing a direct condensation reaction between a fluorinated benzamide derivative and an aminocyclobutane carboxylic acid. This strategy elegantly bypasses the need for cyanide substitution, thereby eliminating the associated toxicity and regulatory risks from the production line. The reaction conditions are remarkably mild, utilizing common polar aprotic solvents like DMSO or DMF and standard inorganic bases such as potassium carbonate. This shift allows for the use of conventional heating and stirring equipment, facilitating a seamless transition from laboratory benchtop to commercial tonnage production. By simplifying the synthetic sequence and removing hazardous steps, the new method significantly enhances the commercial scale-up of complex pharmaceutical intermediates. The result is a more robust process that delivers consistent quality while minimizing the operational risks that often plague traditional synthetic pathways.
Mechanistic Insights into the Cyanide-Free Condensation Strategy
The core of this innovative synthesis lies in the nucleophilic substitution and subsequent cyclization mechanisms that form the critical spiro-hydantoin ring system. In the first stage, the amino group of the cyclobutane derivative attacks the electrophilic center of the fluorinated benzamide precursor, facilitated by the presence of a base that scavenges the resulting acid byproduct. This condensation step is carefully controlled to prevent over-reaction or degradation of the sensitive cyclobutane ring, which is prone to ring-opening under harsh acidic or basic conditions. The choice of solvent plays a pivotal role in stabilizing the transition state, with DMSO providing excellent solubility for both organic and inorganic components. This precise control over reaction kinetics ensures that the intermediate Formula II compound is generated with high selectivity, minimizing the formation of regioisomers that could be difficult to separate later. The mechanistic pathway avoids the generation of free cyanide ions, which are known to cause side reactions and metal contamination in the final product.
Following the formation of the intermediate, the final cyclization with the isothiocyanate derivative (TFP-6) proceeds through a thiourea intermediate that spontaneously closes to form the hydantoin ring. This step is crucial for establishing the correct stereochemistry and structural rigidity required for androgen receptor binding. The absence of cyanide in this stage means that the reaction mixture is cleaner, allowing for simpler workup procedures such as direct precipitation or extraction. Impurity control is significantly enhanced because the potential for cyanide-related byproducts is completely eradicated from the process stream. For R&D teams, this means a more predictable impurity profile that simplifies method validation and regulatory filing. The overall mechanism demonstrates how strategic bond disconnection can lead to safer and more efficient synthetic designs without compromising the structural complexity of the target molecule.
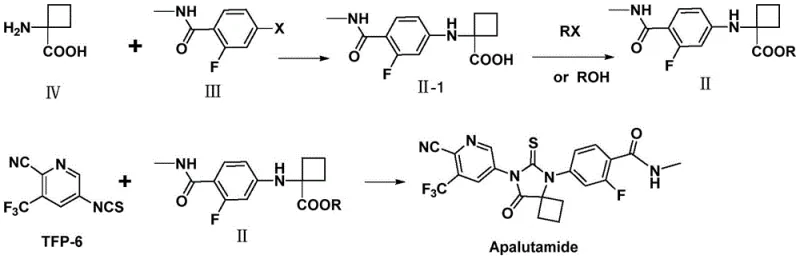
How to Synthesize Apalutamide Efficiently
The synthesis of Apalutamide intermediates via this patented route involves a streamlined sequence that prioritizes safety and yield. The process begins with the coupling of readily available starting materials under controlled thermal conditions to form the key carboxylic acid intermediate. Detailed standard operating procedures for temperature ramping, reagent addition rates, and quenching protocols are essential to maintain batch-to-batch consistency. The subsequent esterification or direct cyclization steps are optimized to maximize conversion while minimizing solvent usage. For process chemists looking to implement this technology, the detailed standardized synthesis steps see the guide below.
- Condense 1-aminocyclobutanecarboxylic acid derivatives with fluorinated benzamide precursors using a base like potassium carbonate in DMF or DMSO.
- Isolate the intermediate Formula II compound through aqueous workup and pH adjustment to ensure high purity before the final cyclization.
- React the purified intermediate with TFP-6 isothiocyanate derivative in a polar aprotic solvent to form the final Apalutamide spiro-cycle structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this cyanide-free synthetic route offers substantial cost savings and supply chain resilience. The elimination of sodium cyanide removes the need for specialized hazardous material handling, storage, and disposal services, which are often cost-prohibitive and logistically complex. This simplification of the raw material portfolio allows procurement managers to source chemicals from a broader range of vendors, reducing dependency on single-source suppliers of toxic reagents. Furthermore, the use of standard solvents and bases ensures that supply disruptions are less likely to impact production schedules. The overall reduction in process complexity translates to lower operational expenditures, making the final API more cost-competitive in the global market. These factors collectively contribute to a more agile and responsive supply chain capable of adapting to fluctuating market demands.
- Cost Reduction in Manufacturing: The removal of toxic cyanide reagents significantly lowers the costs associated with environmental compliance and waste treatment facilities. By avoiding the need for specialized destruction processes for cyanide-containing waste, manufacturers can allocate resources more efficiently towards production capacity expansion. Additionally, the mild reaction conditions reduce energy consumption compared to microwave-assisted methods, leading to lower utility costs per kilogram of product. The simplified purification process also reduces solvent consumption and loss, further driving down the variable costs of production. These cumulative efficiencies result in a more economically viable manufacturing process that supports long-term price stability for customers.
- Enhanced Supply Chain Reliability: Sourcing non-hazardous raw materials significantly reduces lead time for high-purity pharmaceutical intermediates by avoiding strict transportation regulations. Standard chemicals like potassium carbonate and DMF are widely available from multiple global suppliers, ensuring continuity of supply even during regional disruptions. The robustness of the synthetic route means that production can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility is crucial for maintaining inventory levels and meeting just-in-time delivery requirements for downstream drug product manufacturers. A reliable supply of key intermediates ensures that the final drug product can reach patients without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing equipment that is standard in most fine chemical manufacturing plants. The absence of microwave technology removes a significant barrier to increasing batch sizes, allowing for true tonnage production capabilities. Environmental compliance is greatly simplified as the process generates no cyanide-containing effluent, reducing the burden on wastewater treatment systems. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturer, appealing to environmentally conscious partners. The ability to scale safely and cleanly ensures that production can grow in tandem with market demand for this critical oncology medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is key to evaluating the feasibility of integrating this technology into existing production lines. The responses highlight the safety, efficiency, and scalability advantages that define this new method.
Q: How does this new method improve safety compared to prior art routes?
A: The patented method eliminates the use of sodium cyanide and microwave irradiation, which are significant safety hazards in traditional synthesis. By avoiding highly toxic cyanide reagents, the process reduces environmental pollution risks and simplifies hazardous waste management, making it far more suitable for large-scale industrial production.
Q: What are the key reaction conditions for the condensation step?
A: The condensation reaction typically proceeds in solvents such as DMF or DMSO at temperatures ranging from 80°C to 120°C. Common bases like potassium carbonate or triethylamine are used to facilitate the coupling of the aminocyclobutane derivative with the fluorinated benzamide, ensuring mild conditions that protect sensitive functional groups.
Q: Is this synthetic route scalable for commercial API manufacturing?
A: Yes, the route is specifically designed for industrial scale-up. Unlike prior art methods that rely on microwave conditions which are difficult to scale, this process uses standard heating and stirring equipment. The avoidance of exotic reagents and the use of commercially available solvents ensure a robust and continuous supply chain capability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apalutamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic technologies like the one described in CN108069869B to deliver superior pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Apalutamide intermediate meets the highest global standards. Our commitment to safety and quality makes us an ideal partner for long-term supply agreements in the oncology sector. We understand the critical nature of API supply chains and are dedicated to providing uninterrupted service.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this cyanide-free method. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the quality and viability of our production capabilities. Our team is ready to support your R&D and commercialization goals with tailored solutions. Let us collaborate to enhance the efficiency and safety of your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
