Advanced Copper-Catalyzed Synthesis of 1,4,6-Tri-substituted 1,2-Dihydro-1,3,5-Triazines for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. Patent CN111533706A introduces a groundbreaking preparation method for 1,4,6-tri-substituted 1,2-dihydro-1,3,5-triazine compounds, addressing the long-standing challenges associated with their efficient synthesis. These triazine derivatives are not merely academic curiosities; they form the structural backbone of significant bioactive molecules, including antimalarial agents and antineoplastic drugs like Altretamine. The disclosed technology leverages a copper-catalyzed multi-component reaction that operates under remarkably mild conditions, utilizing aromatic amidines and N,N-dimethyl amine reagents as carbon sources. This innovation represents a paradigm shift for a reliable pharmaceutical intermediates supplier, offering a pathway to access complex chemical spaces with unprecedented operational simplicity and economic efficiency.
Furthermore, the strategic importance of this patent lies in its ability to facilitate the rapid generation of diverse chemical libraries for structure-activity relationship studies. Traditional approaches often suffer from limited substrate scope or require harsh reaction environments that degrade sensitive functional groups. In contrast, this novel protocol demonstrates exceptional tolerance to various substituents, allowing for the incorporation of halogens, alkoxy groups, and alkyl chains without compromising the integrity of the triazine core. For research and development teams focused on high-purity OLED material or agrochemical intermediate development, this flexibility is invaluable. The method ensures that the resulting compounds maintain high structural fidelity, which is essential for downstream biological evaluation and regulatory filing processes in the competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted triazines has been plagued by significant technical hurdles that impede cost reduction in fine chemical manufacturing. Prior art, such as the methods described in CN104262273A, typically relies on the condensation of amidine hydrochlorides with alcohols in the presence of copper acetate and sodium carbonate. These conventional routes often necessitate elevated temperatures ranging from 110°C to 120°C and extended reaction times spanning 12 to 24 hours. Such rigorous conditions not only consume substantial energy but also increase the risk of thermal decomposition of sensitive intermediates. Moreover, the requirement for specific stoichiometric additives and the potential need for inert atmospheres add layers of complexity to the process control, making scale-up operations risky and expensive for supply chain managers.
Another critical drawback of existing technologies is the poor atom economy and the generation of significant chemical waste. Many traditional protocols require the use of stoichiometric oxidants or protecting group strategies that result in low overall yields and difficult purification processes. The separation of byproducts often involves multiple chromatographic steps or recrystallizations, which drastically reduces the throughput of the manufacturing line. For procurement managers, this translates to higher raw material costs and longer lead times for high-purity intermediates. The inability to conduct these reactions under ambient air conditions further complicates the engineering requirements, demanding specialized reactors that can exclude moisture and oxygen, thereby inflating the capital expenditure required for commercial scale-up of complex polymer additives or pharmaceutical ingredients.
The Novel Approach
The methodology outlined in CN111533706A offers a transformative solution by employing a one-pot multi-component reaction that constructs the triazine skeleton directly from aromatic amidines and dimethyl amine carbon sources. This approach eliminates the need for pre-functionalized starting materials or harsh activating agents, streamlining the synthetic route into a single operational step. The use of a copper salt catalyst, which is resistant to both moisture and air, allows the reaction to proceed efficiently under an open atmosphere. This feature is particularly advantageous for cost reduction in electronic chemical manufacturing, as it removes the necessity for expensive glovebox techniques or rigorous solvent drying procedures. The reaction system is designed to maximize atom economy, ensuring that the majority of the starting mass is incorporated into the final product, thereby minimizing waste disposal costs.

In addition to operational simplicity, this novel approach provides exceptional structural diversity, enabling the synthesis of a wide array of 1,4,6-tri-substituted derivatives. The compatibility with various substituents on the aromatic rings, including electron-withdrawing and electron-donating groups, allows chemists to fine-tune the physicochemical properties of the final compounds. This versatility is crucial for developing high-purity pharmaceutical intermediates where specific substitution patterns are required to optimize biological activity. The reaction conditions are mild, typically operating between 70°C and 130°C, which preserves the stability of thermally labile functional groups. By simplifying the workflow and reducing the number of unit operations, this technology significantly enhances the scalability and environmental compliance of the manufacturing process, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Copper-Catalyzed Cyclization
Understanding the mechanistic pathway of this transformation is essential for R&D directors aiming to optimize the process for specific substrates. The proposed mechanism involves the initial condensation of two molecules of aromatic amidine, followed by coordination with a copper complex derived from the dimethylethanolamine solvent. This coordination facilitates the formation of critical carbon-nitrogen bonds through a sequence of oxidation and cyclization steps. The copper catalyst acts as a Lewis acid to activate the amidine species while simultaneously mediating the oxidative coupling required to close the triazine ring. This dual functionality ensures that the reaction proceeds with high selectivity, minimizing the formation of oligomeric byproducts that often plague multi-component reactions. The stability of the copper catalyst under aerobic conditions suggests a robust catalytic cycle that can tolerate the presence of oxygen without deactivation.
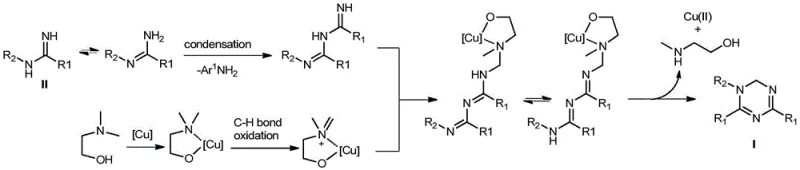
Impurity control is another critical aspect governed by this mechanistic profile. The one-pot nature of the reaction avoids the isolation of unstable intermediates, which are often prone to degradation or side reactions during workup. By maintaining the reaction mixture in a homogeneous phase throughout the transformation, the system ensures consistent exposure of the reactants to the catalytic species. This uniformity leads to a narrower impurity profile, simplifying the downstream purification process. For quality control teams, this means that achieving stringent purity specifications becomes more predictable and less resource-intensive. The mechanism also explains the observed tolerance to water, as the copper complex remains active even in the presence of trace moisture, preventing the hydrolysis of the amidine starting materials. This resilience is a key factor in ensuring batch-to-batch consistency during commercial production.
How to Synthesize 1,4,6-Tri-substituted 1,2-Dihydro-1,3,5-Triazines Efficiently
To implement this synthesis effectively, operators must adhere to precise parameters regarding temperature and reaction time to maximize yield. The protocol dictates dissolving the aromatic amidine compound and the copper catalyst in an organic solvent, preferably N,N-dimethylethanolamine, which serves a dual role as both solvent and carbon source. The mixture is then heated to a temperature range of 80°C to 110°C and stirred for a duration of 28 to 36 hours. Monitoring the reaction progress is vital, as extending the time beyond 36 hours does not significantly improve conversion, while shortening it may lead to incomplete reaction and lower yields. Upon completion, the reaction is quenched with water, and the product is extracted using ethyl acetate. The detailed standardized synthesis steps see the guide below.
- Dissolve aromatic amidine compounds and a copper salt catalyst in an organic solvent such as N,N-dimethylethanolamine.
- Heat the reaction mixture to a temperature range of 80°C to 110°C under air atmosphere for 28 to 36 hours.
- Quench the reaction with water, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly impact the bottom line for procurement and supply chain teams. The elimination of expensive transition metal catalysts that require rigorous removal steps translates into significant cost savings in the manufacturing process. Traditional methods often rely on palladium or other precious metals that leave toxic residues, necessitating costly purification protocols to meet regulatory standards for pharmaceutical ingredients. By utilizing a base copper salt that is both cheap and easily removable, the overall cost of goods sold is drastically reduced. Furthermore, the ability to run the reaction under air without the need for inert gas purging simplifies the reactor setup, allowing for the use of standard glass-lined or stainless steel vessels without specialized modifications.
- Cost Reduction in Manufacturing: The process achieves cost optimization by utilizing inexpensive and readily available raw materials such as aromatic amidines and dimethyl amines. The high atom economy of the reaction ensures that minimal raw material is wasted, directly lowering the input costs per kilogram of product. Additionally, the absence of stoichiometric oxidants or additives reduces the consumption of auxiliary chemicals, further contributing to financial efficiency. The simplified workup procedure, involving basic extraction and chromatography, minimizes labor hours and solvent usage, resulting in a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the robustness of the reaction conditions. Since the catalyst is resistant to moisture and air, the storage and handling of reagents do not require specialized climate-controlled environments. This reduces the risk of raw material degradation during transit and storage, ensuring a consistent supply of high-quality inputs. The shortened reaction timeline compared to traditional multi-step syntheses also reduces the manufacturing cycle time, allowing for faster turnaround on customer orders. This agility is crucial for meeting the dynamic demands of the global pharmaceutical market and reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scalability is inherently supported by the one-pot design, which reduces the number of transfer operations and potential points of failure during scale-up. The reaction generates minimal hazardous waste, aligning with increasingly stringent environmental regulations regarding chemical discharge. The use of recyclable solvents and the potential for solvent recovery systems further enhance the environmental profile of the process. This compliance not only mitigates regulatory risks but also improves the corporate sustainability metrics, making the supply chain more attractive to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation. They are intended to provide clarity on the operational feasibility and strategic value of adopting this method for large-scale production. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of this copper-catalyzed method over traditional triazine synthesis?
A: This method utilizes air-stable copper catalysts and does not require additional oxidants or inert gas protection, significantly simplifying the operational complexity and reducing equipment costs compared to traditional methods requiring strict anhydrous conditions.
Q: Can this synthesis protocol be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the one-pot procedure uses cheap and readily available raw materials with high atom economy. The catalyst's resistance to moisture and air makes it highly suitable for large-scale commercial manufacturing without specialized infrastructure.
Q: What is the typical yield range for the 1,4,6-tri-substituted triazine derivatives?
A: Under optimized conditions of 80°C to 110°C for 28 to 36 hours, the reaction yields typically fall within the 50% to 90% range, depending on the electronic properties of the substituents on the aromatic amidine starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,6-Tri-substituted 1,2-Dihydro-1,3,5-Triazines Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like CN111533706A are successfully translated into industrial reality. Our technical team is adept at optimizing reaction parameters to meet stringent purity specifications required by top-tier pharmaceutical clients. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence ensures that the complex heterocycles produced via this copper-catalyzed route maintain the highest standards of consistency and reliability, supporting your drug development pipelines without interruption.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency. By partnering with us, you gain access to a reliable partner dedicated to delivering high-quality chemical solutions that drive your business forward in a competitive global landscape.
