Advanced One-Pot Synthesis of 1,4,6-Tri-substituted 1,2-Dihydro-1,3,5-Triazines for Commercial Scale-up
Introduction to Next-Generation Triazine Scaffold Construction
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies for constructing nitrogen-rich heterocyclic scaffolds, particularly 1,3,5-triazine derivatives, which serve as critical backbones for numerous bioactive agents. Patent CN111533706A discloses a groundbreaking preparation method for 1,4,6-tri-substituted 1,2-dihydro-1,3,5-triazine compounds, addressing the longstanding need for efficient, scalable routes to these valuable intermediates. This technology leverages a novel copper-catalyzed multi-component reaction that merges aromatic amidines with specific amine-based carbon sources in a streamlined one-pot process. The significance of this innovation lies in its ability to bypass complex multi-step sequences traditionally required for triazine ring assembly, thereby offering a direct pathway to structurally diverse libraries essential for drug discovery programs targeting malaria, cancer, and bacterial infections. By utilizing inexpensive copper salts and air-stable conditions, this protocol represents a paradigm shift towards greener, more economically viable manufacturing processes for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted triazine compounds has relied on methodologies that often suffer from significant operational drawbacks and economic inefficiencies. Prior art, such as the method disclosed in CN104262273A, typically necessitates the use of amidine hydrochlorides mixed with alcohols and copper acetate monohydrate in the presence of bases like sodium carbonate. These conventional routes frequently demand elevated reaction temperatures ranging from 110°C to 120°C and extended reaction times spanning 12 to 24 hours to achieve acceptable conversion. Furthermore, many traditional protocols require strict exclusion of moisture or the use of stoichiometric oxidants, which not only increases the complexity of the reaction setup but also generates substantial amounts of chemical waste. The reliance on harsh thermal conditions can also lead to decomposition of sensitive functional groups, limiting the substrate scope and resulting in lower overall yields that are detrimental to cost-effective commercial production.
The Novel Approach
In stark contrast, the methodology presented in CN111533706A introduces a highly efficient one-pot strategy that dramatically simplifies the synthetic landscape for 1,4,6-tri-substituted 1,2-dihydro-1,3,5-triazines. This innovative approach utilizes aromatic amidines and N,N-dimethyl-containing amine reagents, which uniquely serve a dual role as both the carbon source and the reaction solvent. The reaction proceeds smoothly at milder temperatures, optimally between 80°C and 110°C, and remarkably, it can be conducted directly under an air atmosphere without the need for inert gas protection. This tolerance to air and moisture eliminates the need for expensive specialized equipment and rigorous drying procedures, thereby enhancing process safety and operational simplicity. The use of simple copper salts as catalysts ensures high atom economy, as the reaction avoids the generation of excessive by-products associated with stoichiometric oxidants, making it an ideal candidate for sustainable industrial manufacturing.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
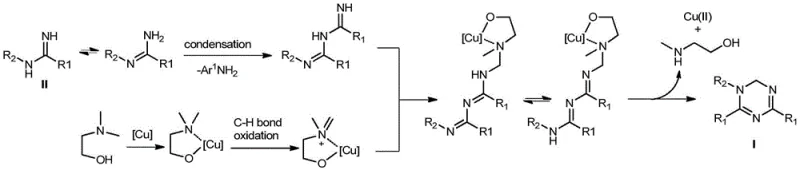
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize process parameters and ensure consistent product quality. The proposed mechanism involves a sophisticated interplay between the copper catalyst and the organic substrates, initiating with the condensation of two molecules of aromatic amidine. This initial step is followed by the coordination and activation of the N,N-dimethyl amine carbon source by the copper species, facilitating the formation of critical carbon-nitrogen bonds. The copper center acts as a redox mediator, promoting the oxidative cyclization necessary to close the triazine ring while regenerating the active catalytic species through interaction with molecular oxygen from the air. This catalytic cycle is highly efficient, minimizing the accumulation of reactive intermediates that could otherwise lead to polymerization or side reactions, thus ensuring a clean reaction profile.
From an impurity control perspective, the mechanism offers distinct advantages by avoiding the use of strong external oxidants that often generate difficult-to-remove inorganic salts or over-oxidized by-products. The mild oxidative conditions provided by the copper-air system allow for precise control over the oxidation state of the final triazine product, preventing the formation of fully aromatic 1,3,5-triazine impurities which might arise under harsher conditions. Furthermore, the chelation of the copper catalyst with the amine solvent helps stabilize the metal species, preventing precipitation or deactivation that could lead to incomplete conversion. This mechanistic robustness translates directly to a narrower impurity profile in the crude reaction mixture, significantly reducing the burden on downstream purification steps and enabling the production of high-purity pharmaceutical intermediates suitable for stringent regulatory standards.
How to Synthesize 1,4,6-Tri-substituted 1,2-Dihydro-1,3,5-Triazines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal profiles to maximize yield and reproducibility. The standard protocol involves dissolving the aromatic amidine starting material and a catalytic amount of copper chloride in N,N-dimethylethanolamine, which serves as both the solvent and the one-carbon donor. The mixture is then heated to approximately 80°C and stirred for a period of 28 to 36 hours under ambient air conditions. Following the reaction completion, the workup procedure is straightforward, involving aqueous quenching followed by extraction with organic solvents like ethyl acetate. The detailed standardized synthesis steps, including specific molar ratios, temperature ramping rates, and purification techniques for various substrate derivatives, are outlined in the guide below.
- Dissolve aromatic amidine compounds and a copper salt catalyst (e.g., CuCl2) in a dual-function solvent/carbon source like N,N-dimethylethanolamine.
- Heat the reaction mixture to 80-110°C under air atmosphere for 28-36 hours to facilitate oxidative cyclization.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials; the key reagents, aromatic amidines and dimethylethanolamine, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of single-source dependency. Moreover, the elimination of sensitive reagents and the ability to run the reaction in air significantly reduces the logistical costs associated with transporting and storing hazardous or air-sensitive materials. This robustness ensures a more reliable supply of critical intermediates, preventing production bottlenecks that can arise from the unavailability of specialized catalysts or solvents required by older technologies.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several cost-saving factors. By utilizing a dual-function solvent that also acts as a carbon source, the process eliminates the need for purchasing and disposing of separate bulk solvents, thereby reducing both material costs and waste treatment expenses. The use of inexpensive copper salts instead of precious metal catalysts like palladium or rhodium further drives down the bill of materials, while the absence of stoichiometric oxidants removes another significant cost center. Additionally, the simplified workup procedure reduces labor hours and energy consumption associated with complex purification trains, leading to substantial overall cost savings in API intermediate manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: The operational resilience of this method directly translates to improved supply chain continuity. Since the reaction is tolerant to moisture and air, it does not require specialized glass-lined reactors equipped with rigorous inert gas systems, allowing for production in a wider range of manufacturing facilities. This flexibility enables faster technology transfer between sites and reduces the lead time for scaling up production to meet market demand. The stability of the catalyst system also means that batch-to-batch variability is minimized, ensuring consistent delivery schedules and reducing the risk of failed batches that could disrupt the downstream supply of finished pharmaceutical products.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with modern green chemistry principles and regulatory expectations. The high atom economy ensures that a maximum proportion of raw materials ends up in the final product, minimizing the generation of hazardous waste streams that require costly disposal. The ability to operate at moderate temperatures reduces the energy footprint of the manufacturing process, contributing to lower carbon emissions. Furthermore, the simplified effluent profile facilitates easier compliance with environmental regulations, making it easier to obtain necessary permits for commercial scale-up and ensuring long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this process for potential integration into their supply chains. Understanding these details is essential for assessing the feasibility of adopting this method for specific project requirements.
Q: What are the key advantages of this copper-catalyzed triazine synthesis over traditional methods?
A: This method utilizes a one-pot strategy with high atom economy, operating under mild conditions (80°C) in air without additional oxidants, unlike prior art requiring higher temperatures and inert atmospheres.
Q: Is the catalyst system sensitive to moisture or oxygen?
A: No, the copper salt catalyst described in patent CN111533706A is resistant to moisture and air, significantly simplifying operational requirements and reducing equipment costs for industrial scale-up.
Q: What is the substrate scope for R1 and R2 groups in this triazine synthesis?
A: The method tolerates a wide range of aryl substituents including phenyl, naphthyl, and various substituted phenyl rings with electron-donating or withdrawing groups like methyl, methoxy, chloro, and fluoro.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,6-Tri-substituted 1,2-Dihydro-1,3,5-Triazines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the copper-catalyzed triazine construction described in CN111533706A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the management of copper residues to meet stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we guarantee that every batch of 1,4,6-tri-substituted 1,2-dihydro-1,3,5-triazines meets the highest industry standards for purity and performance.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective and scalable technology for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your project needs,索取 specific COA data, and obtain comprehensive route feasibility assessments that will demonstrate how our expertise can accelerate your development timelines and optimize your manufacturing costs.
