Advanced FeCl₃-Catalyzed Manufacturing Process for High-Purity Trifluoromethyl Triazole Derivatives in Pharmaceutical Intermediate Production
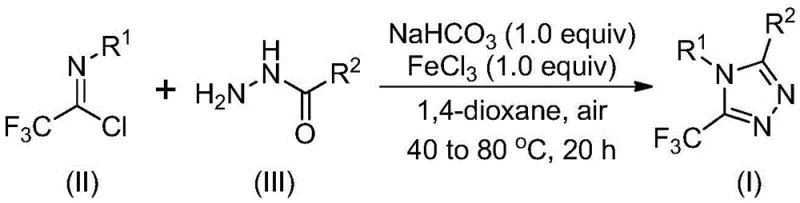
The present analysis examines patent CN111978265B detailing an innovative synthetic route to trifluoromethyl-substituted triazole derivatives—a critical class of compounds with demonstrated applications in pharmaceutical development as evidenced by their presence in marketed drugs such as maraviroc and sitagliptin. This patented methodology represents a significant advancement over conventional approaches by eliminating stringent environmental requirements while maintaining high substrate flexibility and operational simplicity. The process leverages iron-based catalysis to achieve efficient cyclization under ambient conditions, thereby addressing key industry pain points related to cost-intensive infrastructure and complex handling protocols previously required for similar heterocyclic syntheses. By utilizing commercially accessible starting materials including aryl imidoyl chlorides and hydrazides, this method establishes a robust foundation for scalable production of high-value intermediates essential to modern drug discovery pipelines without compromising on quality or consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for trifluoromethylated triazoles have been severely constrained by multiple operational deficiencies that hinder commercial viability. The prevalent methods involving oxadiazole condensation or hydrazone cyclization typically demand rigorously anhydrous conditions and inert atmospheres that necessitate specialized equipment investment and complex procedural controls beyond standard manufacturing capabilities. These approaches further suffer from narrow substrate tolerance that restricts structural diversity while requiring extended reaction times under elevated temperatures that increase energy consumption and decomposition risks. Critically, the limited functional group compatibility observed in existing protocols creates significant barriers when synthesizing complex derivatives needed for advanced pharmaceutical applications. The cumulative effect of these constraints manifests as inconsistent yields below industry standards and prohibitive production costs that undermine supply chain reliability for time-sensitive drug development programs requiring consistent intermediate availability.
The Novel Approach
The patented methodology introduces a paradigm shift through its strategic implementation of iron(III) chloride catalysis that operates effectively under ambient atmospheric conditions without requiring moisture-sensitive handling protocols. This innovation enables direct utilization of cost-effective starting materials including readily available aryl imidoyl chlorides and hydrazides through a two-stage temperature-controlled process that first establishes carbon-nitrogen bonds at moderate temperatures followed by cyclization at elevated conditions optimized for dehydration kinetics. The elimination of transition metal catalysts not only reduces raw material expenses but also removes downstream purification challenges associated with heavy metal residues that previously compromised product quality specifications. Crucially, the expanded substrate scope accommodates diverse functional groups including alkyl chains and halogenated aromatics while maintaining consistent conversion rates across varied molecular architectures—thereby enabling tailored synthesis of complex derivatives previously inaccessible through conventional routes while significantly improving overall process efficiency.
Mechanistic Insights into FeCl₃-Catalyzed Cyclization
The reaction mechanism proceeds through a well-defined sequence initiated by base-promoted nucleophilic attack where sodium bicarbonate facilitates deprotonation of the hydrazide nitrogen to form an active nucleophile that attacks the electrophilic carbon of the imidoyl chloride precursor. This intermolecular carbon-nitrogen bond formation generates a key trifluoroacetamidine intermediate that subsequently undergoes Lewis acid-mediated intramolecular cyclization where ferric chloride coordinates with nitrogen atoms to activate the system toward dehydration condensation. The iron catalyst lowers the energy barrier for ring closure through selective orbital interactions that promote proton transfer while suppressing competing side reactions that typically generate impurities in conventional syntheses. This dual-stage process operates within a precisely controlled temperature window—first at moderate conditions to form stable intermediates followed by elevated temperatures that drive cyclization kinetics—thereby ensuring high regioselectivity toward the desired triazole core structure while maintaining excellent functional group tolerance across diverse substrates.
Impurity profile management is inherently addressed through the reaction's mechanistic design where the mild catalytic system minimizes decomposition pathways while the two-stage temperature protocol prevents accumulation of reactive intermediates that could lead to side products. The absence of transition metals eliminates metal-induced degradation pathways that commonly generate trace impurities requiring extensive purification in alternative methods. Furthermore, the process demonstrates exceptional selectivity toward forming the desired regioisomer without requiring additional separation steps—thereby directly contributing to higher final product purity levels essential for pharmaceutical applications where even minor impurities can compromise drug safety profiles. This inherent selectivity extends across various substrate combinations as evidenced by consistent yields across diverse functional groups including halogenated aromatics and alkyl chains without significant byproduct formation.
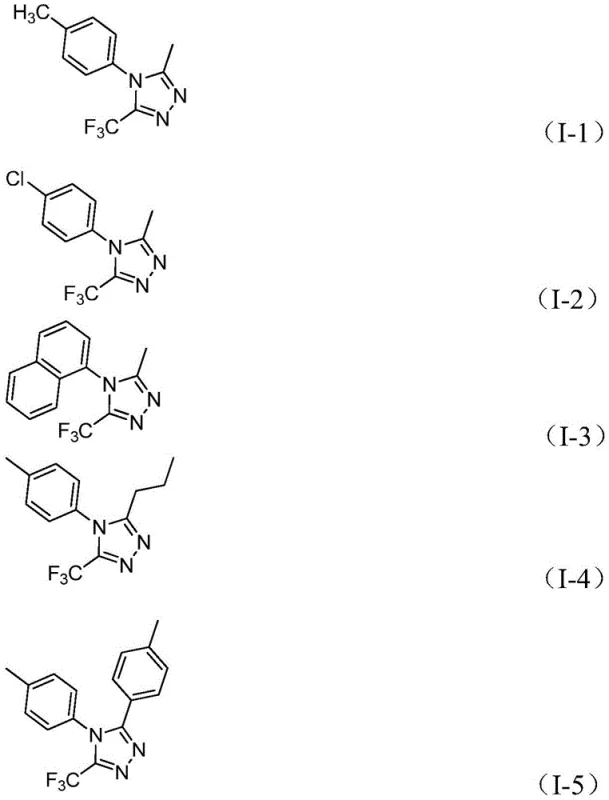
How to Synthesize Trifluoromethyl Triazole Derivatives Efficiently
This patented synthetic route provides a streamlined pathway for producing high-purity trifluoromethyl triazole derivatives through a carefully optimized sequence that balances reactivity with operational simplicity. The methodology eliminates traditional barriers by operating under ambient atmospheric conditions while maintaining excellent yield consistency across diverse substrate combinations—making it particularly suitable for industrial implementation where process robustness is paramount. Detailed standardized procedures have been developed based on extensive experimental validation to ensure reproducible results from laboratory scale through commercial production volumes. The following section outlines the precise operational parameters required to achieve optimal results when implementing this innovative manufacturing approach.
- Combine sodium bicarbonate (1.0 equiv), trifluoroethyl imidoyl chloride precursor (II), and hydrazide compound (III) in anhydrous dioxane solvent under ambient atmosphere at controlled temperature between 40°C and 80°C for initial carbon-nitrogen bond formation.
- Introduce ferric chloride catalyst (1.0 equiv) to the reaction mixture and elevate temperature to optimize cyclization kinetics while maintaining oxygen tolerance during the dehydration condensation phase.
- Execute purification through filtration to remove inorganic residues followed by silica gel mixing and column chromatography to achieve stringent purity specifications required for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
This manufacturing innovation delivers substantial value across procurement and supply chain functions by addressing critical pain points inherent in traditional intermediate sourcing strategies. The elimination of specialized infrastructure requirements directly translates to reduced capital expenditure while the simplified operational profile enables faster technology transfer between production sites—thereby enhancing overall supply chain resilience against market volatility and demand fluctuations. Furthermore, the use of commercially available starting materials establishes multiple sourcing options that mitigate single-supplier dependencies while maintaining consistent quality standards required for pharmaceutical applications.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization through elimination of expensive transition metal catalysts and specialized handling equipment required by conventional methods. By operating under standard atmospheric conditions without moisture-sensitive protocols, manufacturers avoid substantial infrastructure investments while reducing energy consumption through moderate temperature profiles. The simplified purification workflow further contributes to cost savings by minimizing solvent usage and processing time compared to traditional multi-step purification sequences required when using alternative synthetic routes.
- Enhanced Supply Chain Reliability: The methodology's robustness against environmental variables ensures consistent production output regardless of regional facility capabilities while maintaining high yield stability across different raw material batches. This operational flexibility enables seamless transfer between manufacturing sites without revalidation requirements—thereby providing procurement teams with multiple qualified production options that enhance supply continuity during market disruptions or capacity constraints.
- Scalability and Environmental Compliance: The absence of hazardous reagents and transition metals simplifies waste stream management while meeting increasingly stringent environmental regulations without requiring additional treatment steps. The process demonstrates exceptional scalability from laboratory validation directly to commercial production volumes due to its inherent operational simplicity—enabling rapid response to changing demand patterns while maintaining consistent quality metrics essential for regulatory compliance in pharmaceutical manufacturing environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented manufacturing process based on extensive experimental validation data from multiple production scales.
Q: How does this synthesis overcome conventional limitations in triazole derivative manufacturing?
A: The patented method eliminates harsh reaction conditions and narrow substrate scope by utilizing mild FeCl₃ catalysis under air atmosphere with readily available starting materials. Unlike traditional approaches requiring anhydrous environments or expensive catalysts, this process operates efficiently at moderate temperatures without inert gas protection while accommodating diverse functional groups.
Q: What scalability advantages does this process offer for commercial production?
A: The methodology demonstrates exceptional scalability from laboratory to industrial scale due to its simplified operational requirements and elimination of specialized equipment. The absence of moisture-sensitive steps enables straightforward transition from gram-scale validation to multi-kilogram production without re-engineering reaction parameters or purification protocols.
Q: How does the process ensure high purity levels required for pharmaceutical intermediates?
A: The catalytic system minimizes side reactions through selective activation pathways while the two-stage temperature profile controls intermediate stability. Combined with standard chromatographic purification that removes trace metal residues without additional processing steps, this approach consistently delivers products meeting stringent pharmaceutical quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Derivative Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. Our dedicated technical teams apply rigorous QC labs and process analytical technologies to ensure consistent product quality across all production volumes—providing pharmaceutical clients with reliable access to high-purity intermediates essential for drug development timelines. By leveraging this patented methodology within our integrated manufacturing platform, we deliver superior process reliability that directly supports clients' critical path activities through robust supply chain management and continuous quality improvement initiatives.
Request a Customized Cost-Saving Analysis from our technical procurement team today to evaluate how this innovative synthesis can optimize your intermediate sourcing strategy. We provide comprehensive support including specific COA data and route feasibility assessments tailored to your unique manufacturing requirements—enabling informed decision-making that drives both quality assurance and operational efficiency across your supply chain.
