Advanced Copper-Catalyzed Synthesis of Halogenated Oxaallylamine Compounds for Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of Halogenated Oxaallylamine Compounds for Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign processes. A significant breakthrough in this domain is detailed in Chinese Patent CN112441934B, which discloses a novel preparation method for halogenated oxaallylamine compounds. These compounds serve as critical building blocks for a wide array of biologically active molecules, particularly beta-adrenoceptor antagonists used in cardiovascular therapy. The patent introduces a copper-catalyzed halogen amination reaction that utilizes easily accessible allenyl ethers and aromatic amines as starting materials. This approach represents a paradigm shift from traditional methodologies, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines. By leveraging mild reaction conditions and abundant raw materials, this technology addresses key pain points in the supply chain of complex amine derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of allylamine scaffolds containing multiple heteroatoms has presented significant challenges to process chemists. Conventional strategies often rely on transition metal-catalyzed direct allylic amination of allyl alcohols or hydroamination reactions, which frequently suffer from limited substrate scope and苛刻 reaction conditions. Many existing protocols require expensive noble metal catalysts, rigorous exclusion of moisture and oxygen, or the use of directing groups that add unnecessary synthetic steps. Furthermore, the synthesis of halogenated variants specifically has been rare, often necessitating the use of electron-withdrawing allenes or specific halogen sources that limit the diversity of the final product. These limitations result in higher production costs, longer lead times, and difficulties in scaling up for commercial manufacturing, creating bottlenecks for procurement managers aiming to secure stable supplies of high-value intermediates.
The Novel Approach
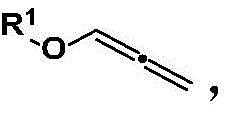
In stark contrast to these legacy methods, the technology described in CN112441934B offers a streamlined and highly efficient alternative. The core innovation lies in the direct reaction of allenyl ethers with aromatic amines in the presence of a copper halide catalyst under an oxygen atmosphere. This method eliminates the need for complex pre-functionalization or harsh reagents, operating effectively at mild temperatures between 35°C and 45°C. The reaction demonstrates exceptional versatility, accommodating a broad range of substituents on both the ether and amine components, including phenyl, naphthyl, and various halogenated groups. This flexibility allows for the rapid generation of diverse chemical libraries, facilitating drug discovery and process optimization. The simplicity of the operational procedure, combined with high step economy, positions this technology as a superior choice for modern pharmaceutical manufacturing.

Mechanistic Insights into Copper-Catalyzed Halogen Amination
The mechanistic pathway of this transformation is both elegant and efficient, relying on the unique reactivity of copper species to activate the allene system. The reaction initiates with the formation of a copper amine intermediate, which subsequently activates the allenyl ether substrate. This activation generates a key alkenyl copper intermediate, a pivotal species that dictates the regioselectivity and efficiency of the process. Following this activation, a free amine molecule performs a nucleophilic attack on the activated complex. The cycle concludes with a reductive elimination step that releases the desired halogenated oxaallylamine product and regenerates the active copper catalyst. This mechanism ensures high atom economy and minimizes the formation of unwanted byproducts, which is crucial for maintaining high purity standards in API intermediate production.
From an impurity control perspective, the mild nature of the reaction conditions plays a vital role in ensuring a clean product profile. The use of an oxygen balloon as the oxidant source avoids the introduction of harsh chemical oxidants that could lead to over-oxidation or degradation of sensitive functional groups. The tolerance for various substituents, such as methyl, methoxy, and halogen groups on the aromatic rings, indicates that the catalytic cycle is robust against electronic and steric variations. This robustness translates to a simplified downstream purification process, often requiring only standard extraction and chromatography techniques. For R&D directors, this means a more predictable impurity profile and easier validation of the manufacturing process, ultimately accelerating the timeline from bench-scale development to commercial production.
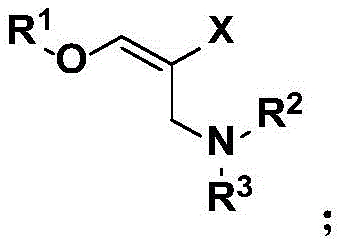
How to Synthesize Halogenated Oxaallylamine Efficiently
The synthesis protocol outlined in the patent is designed for practicality and reproducibility, making it highly suitable for transfer to pilot and production scales. The process begins with the preparation of the allenyl ether substrate, which can be readily synthesized from phenols and propargyl bromide followed by isomerization, ensuring a steady supply of high-quality starting materials. The main reaction involves charging a reactor with copper halide, dissolving it in a common organic solvent like 1,4-dioxane or toluene, and adding the amine and ether substrates. The mixture is then stirred under an oxygen atmosphere at controlled temperatures. Detailed standard operating procedures for this synthesis, including precise stoichiometric ratios and workup parameters, are essential for consistent quality. For a comprehensive guide on executing this synthesis with optimal yield and purity, please refer to the standardized protocol below.
- Charge a reactor with copper halide (CuX) and dissolve in an organic solvent such as 1,4-dioxane or toluene.
- Add the allene ether substrate and aromatic amine reactant to the mixture under an oxygen atmosphere.
- Stir the reaction at 35-45°C for 5-10 hours, then perform extraction and purification via thin layer chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain. By utilizing simple phenols and propargyl bromide as precursors for the allenyl ethers, manufacturers can bypass the need for exotic or proprietary starting materials that are often subject to volatile pricing and supply disruptions. The reliance on copper halides, which are commodity chemicals, further reduces the dependency on expensive noble metals like palladium or rhodium. This shift not only lowers the direct material costs but also mitigates the risk associated with the supply of critical catalysts, ensuring greater continuity of supply for long-term production contracts.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of costly reagents and complex purification steps. Traditional methods often require expensive ligands or stoichiometric amounts of oxidants, whereas this method uses catalytic amounts of inexpensive copper salts and molecular oxygen. The mild reaction temperatures (35-45°C) significantly reduce energy consumption compared to high-temperature reflux processes. Furthermore, the high step economy means fewer unit operations are required to reach the final intermediate, leading to substantial savings in labor, equipment time, and solvent usage. These factors collectively contribute to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Stability and predictability are paramount in the pharmaceutical supply chain, and this technology delivers on both fronts. The substrates involved, such as substituted anilines and phenyl allene ethers, are commercially available or easily synthesized from bulk chemicals, reducing the risk of raw material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, resulting in consistent batch-to-batch performance. This reliability allows supply chain planners to forecast production schedules with greater accuracy and maintain lower safety stock levels, optimizing working capital while ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often introduces unforeseen challenges, but this method is inherently scalable due to its simplicity. The use of standard organic solvents and the absence of hazardous high-pressure reagents facilitate a smooth transition to large-scale reactors. Additionally, the environmental footprint of the process is minimized by the use of oxygen as a green oxidant and the generation of minimal waste streams. The ability to efficiently convert a series of halogenated oxaallylamine compounds also opens avenues for downstream derivatization, such as Grignard reactions, adding further value to the supply chain by enabling the production of diverse polysubstituted allylamine compounds from a common intermediate platform.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthetic method is crucial for stakeholders evaluating its potential for integration into existing manufacturing workflows. The following questions address common inquiries regarding the operational parameters, scope, and downstream utility of this copper-catalyzed process. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Whether you are concerned about reaction safety, substrate compatibility, or purification requirements, the information below aims to clarify the practical aspects of implementing this technology in a commercial setting.
Q: What are the key advantages of this copper-catalyzed method over traditional allylamine synthesis?
A: Unlike conventional methods that often require harsh conditions or expensive transition metal catalysts, this patent utilizes simple copper halides and readily available allenyl ethers. The process operates at mild temperatures (35-45°C) with excellent functional group tolerance, significantly simplifying the purification workflow.
Q: Can this method be scaled for industrial production of beta-blocker intermediates?
A: Yes, the patent explicitly demonstrates high step economy and wide substrate applicability, including naphthalene and substituted phenyl groups common in beta-adrenoceptor antagonists. The use of standard organic solvents and oxygen balloons indicates strong potential for commercial scale-up without specialized high-pressure equipment.
Q: What is the purity profile of the resulting halogenated oxaallylamine compounds?
A: The method yields high-purity products through straightforward workup procedures involving ethyl acetate extraction and column chromatography. The specific examples in the patent report isolated yields ranging from 45% to 86%, with structural confirmation via NMR and HRMS, ensuring the material meets stringent specifications for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Oxaallylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN112441934B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this copper-catalyzed method can be fully realized in an industrial environment. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for your drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this efficient synthesis route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for related intermediates and to discuss route feasibility assessments for your target molecules. Let us help you accelerate your path to market with reliable, high-quality chemical solutions.
