Advanced Copper-Catalyzed Synthesis of Halogenated Oxaallylamines for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for constructing complex heteroatom-rich scaffolds, particularly those serving as critical intermediates for cardiovascular medications. Patent CN112441934B discloses a groundbreaking methodology for the preparation of halogenated oxaallylamine compounds, a structural motif prevalent in potent beta-adrenoceptor antagonists. As illustrated in the background art, compounds such as Oxprenolol and Propranolol rely on similar allylamine architectures to achieve high affinity binding to beta-AR receptors, with Ki values in the nanomolar range.  . This patent introduces a novel copper-catalyzed strategy that bypasses the limitations of conventional synthesis, utilizing easily accessible allenyl ethers and aromatic amines to generate these valuable intermediates with exceptional efficiency and functional group tolerance.
. This patent introduces a novel copper-catalyzed strategy that bypasses the limitations of conventional synthesis, utilizing easily accessible allenyl ethers and aromatic amines to generate these valuable intermediates with exceptional efficiency and functional group tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of allylamine compounds containing multiple heteroatoms has been fraught with synthetic challenges that hinder efficient commercial manufacturing. Traditional strategies often rely on transition metal-catalyzed direct allylic amination of allyl alcohols or hydroamination reactions, which frequently suffer from narrow substrate scopes and stringent reaction requirements. Furthermore, existing methods for introducing halogen atoms into these frameworks typically involve the reaction of electron-withdrawing allenes with halogen sources or require specific guiding groups like p-toluenesulfonamide to direct the amination. These legacy approaches limit the versatility of the synthesis, making it difficult to access diverse libraries of halogenated oxaallylamines without extensive optimization or protecting group manipulations, thereby increasing both time and material costs for process development teams.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the disclosed invention presents a streamlined copper-catalyzed halogen amination reaction that directly couples divinyl ethers with aromatic amines. This innovative route operates under remarkably mild conditions, typically between 35°C and 45°C, utilizing simple copper halides as catalysts in common organic solvents like 1,4-dioxane.  . The reaction proceeds under an oxygen atmosphere, eliminating the need for inert gas handling in many contexts, and delivers the target halogenated oxaallylamine compounds in a single step with high atom economy. This method significantly expands the chemical space accessible to medicinal chemists by tolerating a wide array of substituents on both the ether and amine components, including various phenyl, naphthyl, and halogenated derivatives.
. The reaction proceeds under an oxygen atmosphere, eliminating the need for inert gas handling in many contexts, and delivers the target halogenated oxaallylamine compounds in a single step with high atom economy. This method significantly expands the chemical space accessible to medicinal chemists by tolerating a wide array of substituents on both the ether and amine components, including various phenyl, naphthyl, and halogenated derivatives.
Mechanistic Insights into Copper-Catalyzed Halogen Amination
The mechanistic pathway of this transformation is elegantly simple yet highly effective, driven by the unique reactivity of the copper-amine complex. The reaction initiates when the copper halide interacts with the aromatic amine to form a reactive copper-amine intermediate, which subsequently activates the allenyl ether substrate. This activation generates a transient alkenyl copper species that is poised for nucleophilic attack. The free amine then attacks this activated intermediate, facilitating the formation of the carbon-nitrogen bond essential for the allylamine backbone. Finally, a reductive elimination step releases the final halogenated oxaallylamine product and regenerates the copper catalyst, completing the catalytic cycle. This mechanism avoids the formation of stable byproducts often seen in radical pathways, ensuring a cleaner reaction profile that simplifies downstream processing.
From an impurity control perspective, the mild thermal conditions (35-45°C) play a crucial role in maintaining product integrity. Higher temperatures often promote the polymerization of allene ethers or the degradation of sensitive halogenated intermediates, leading to complex impurity profiles that are difficult to separate. By maintaining the reaction within this narrow temperature window, the process minimizes side reactions such as over-halogenation or ether cleavage. Additionally, the use of stoichiometric oxygen acts as a mild oxidant to facilitate the catalytic turnover without introducing harsh oxidative stress that could damage the electron-rich aromatic rings. This precise control over reaction parameters ensures that the resulting crude product contains high levels of the desired isomer, reducing the burden on purification units and enhancing the overall yield of high-purity material suitable for subsequent Grignard reactions or direct API synthesis.
How to Synthesize Halogenated Oxaallylamine Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it highly attractive for pilot plant and commercial scale operations. The process begins with the preparation of the key allenyl ether starting material, which can be synthesized from readily available phenols and propargyl bromide followed by isomerization, as depicted in the preparatory schemes. 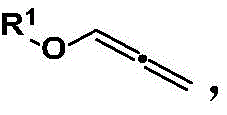 . Once the substrates are prepared, the core coupling reaction requires only the mixing of the copper catalyst, solvent, and reactants in a standard reactor equipped with an oxygen source. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures optimized for maximum recovery, are outlined in the technical guide below.
. Once the substrates are prepared, the core coupling reaction requires only the mixing of the copper catalyst, solvent, and reactants in a standard reactor equipped with an oxygen source. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures optimized for maximum recovery, are outlined in the technical guide below.
- Prepare the reaction system by adding copper halide (CuX) into a reactor, followed by the addition of allenyl ether and aromatic amine substrates dissolved in an organic solvent such as 1,4-dioxane.
- Stir the reaction mixture under an oxygen atmosphere (e.g., oxygen balloon) at a controlled temperature range of 35-45°C for a duration of 5 to 10 hours to ensure complete conversion.
- Upon completion, cool the reaction liquid, extract with ethyl acetate, dry the organic phase over anhydrous sodium sulfate, and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers substantial strategic benefits regarding cost structure and supply reliability. The primary advantage lies in the drastic simplification of the raw material portfolio; the process utilizes commodity chemicals such as phenols, propargyl bromide, and simple aromatic amines, which are globally available from multiple suppliers. This diversity in sourcing mitigates the risk of supply chain disruptions that often plague processes dependent on exotic or single-source reagents. Furthermore, the elimination of complex multi-step sequences required by older methods translates directly into reduced manufacturing lead times and lower inventory holding costs, allowing for a more agile response to market demand fluctuations for cardiovascular intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of expensive transition metal catalysts and specialized ligands often required in palladium or rhodium-catalyzed alternatives. By utilizing inexpensive copper halides and avoiding the need for cryogenic conditions or high-pressure equipment, the operational expenditure (OPEX) for utility consumption is significantly lowered. Additionally, the high step economy means fewer isolation and purification stages are required, which reduces solvent usage and waste disposal costs, contributing to a leaner and more cost-effective manufacturing process for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, a critical factor for maintaining uninterrupted API production schedules. The tolerance for various functional groups means that the same core process can be adapted to produce a wide range of analogues without requalifying the entire manufacturing line. This flexibility allows suppliers to consolidate production campaigns and optimize reactor utilization rates, thereby shortening the delivery windows for custom synthesis projects and ensuring a steady flow of materials to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process aligns well with green chemistry principles by operating at near-ambient temperatures and generating minimal hazardous waste. The absence of toxic heavy metals simplifies the effluent treatment process, reducing the environmental compliance burden on manufacturing sites. Moreover, the demonstrated stability of the reaction conditions suggests excellent scalability from laboratory gram-scale to multi-ton commercial production, enabling manufacturers to confidently commit to long-term supply agreements for complex polymer additives or specialty chemical intermediates derived from this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenated oxaallylamine synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential licensees or contract manufacturing partners.
Q: What are the primary advantages of this copper-catalyzed method over traditional transition metal catalysis?
A: This method utilizes simple and readily available allenyl ethers and aromatic amines, operating under mild conditions (35-45°C) with high step economy. Unlike prior art which often requires harsh conditions or specific guiding groups, this approach offers wide substrate applicability and excellent functional group tolerance.
Q: Can this synthesis method be scaled for industrial production of beta-blocker intermediates?
A: Yes, the patent highlights the potential application value in industry through large-scale experiments. The use of common solvents like 1,4-dioxane and standard copper halides suggests a pathway that is amenable to commercial scale-up for producing polysubstituted allylamine compounds.
Q: What is the typical purification process for these halogenated oxaallylamine compounds?
A: The purification involves a standard workup procedure including extraction with ethyl acetate, drying with anhydrous sodium sulfate, and removal of solvent under reduced pressure. Final purification is achieved using thin layer chromatography or column chromatography with a petroleum ether and ethyl acetate mixed solvent system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Oxaallylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in streamlining the production of next-generation cardiovascular therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of halogenated oxaallylamine intermediate meets the exacting standards required for global regulatory filings.
We invite you to leverage our technical expertise to optimize your supply chain for these critical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this patented method can drive value and efficiency in your API manufacturing operations.
