Revolutionizing Asymmetric Synthesis: Scalable Production of High-Purity Naphthalene-Pyrrole Phosphine Catalyst for Pharmaceutical Applications
The patent CN114029086B introduces a groundbreaking axial chiral naphthalene-pyrrole phosphine catalyst that represents a significant advancement in asymmetric catalysis technology. This novel catalyst system addresses critical limitations in conventional chiral phosphine catalysts by offering superior stereocontrol across multiple reaction types while maintaining practical manufacturing advantages. The invention provides a robust solution for pharmaceutical manufacturers seeking high-purity intermediates through asymmetric transformations, with particular relevance to complex molecule synthesis where precise stereochemistry is paramount. Unlike traditional carbon-centered chiral phosphines that dominate the market, this axial chiral design creates a unique three-dimensional environment that enhances enantioselectivity while maintaining excellent functional group tolerance. The patent demonstrates how this innovative molecular architecture overcomes longstanding challenges in catalyst design that have constrained industrial adoption of asymmetric methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral phosphine catalysts predominantly rely on carbon-centered chirality, which often results in limited conformational rigidity and inconsistent stereocontrol across diverse substrate classes. These catalysts typically require harsh reaction conditions or expensive transition metals to achieve acceptable enantioselectivity, significantly increasing production costs and complicating purification processes. The narrow substrate scope of conventional systems frequently necessitates developing entirely new catalysts for each specific transformation, creating substantial R&D burdens and supply chain vulnerabilities. Furthermore, many existing chiral phosphines suffer from air sensitivity or decomposition under standard processing conditions, requiring specialized handling procedures that increase operational complexity and reduce manufacturing efficiency. The limited structural diversity among commercially available chiral phosphines also restricts their applicability to only certain reaction types, forcing manufacturers to maintain multiple catalyst inventories for different synthetic routes.
The Novel Approach
The axial chiral naphthalene-pyrrole phosphine catalyst described in CN114029086B overcomes these limitations through its innovative molecular architecture that leverages axial chirality rather than traditional point chirality. This design creates a rigid, well-defined chiral pocket that consistently delivers high enantioselectivity across multiple reaction types without requiring expensive transition metals or extreme reaction conditions. The preparation method employs mild temperatures (0-40°C), readily available starting materials, and standard purification techniques that are easily scalable from laboratory to commercial production. Crucially, the modular synthesis allows for structural variation through different substituents while maintaining consistent catalytic performance, enabling customization for specific pharmaceutical applications without redesigning the entire catalytic system. This approach eliminates the need for multiple specialized catalysts by providing a single platform technology applicable to both hydrosilylation and coupling reactions.
Mechanistic Insights into Axial Chiral Naphthalene-Pyrrole Phosphine Catalysis
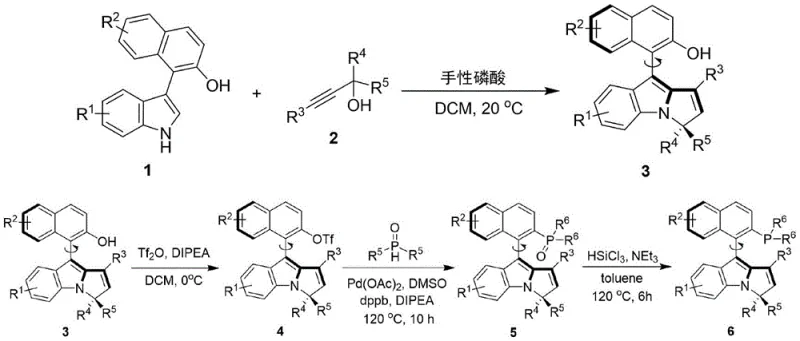
The exceptional performance of this catalyst stems from its unique axial chirality that creates a highly organized transition state geometry during asymmetric transformations. The naphthalene-pyrrole scaffold forms a rigid chiral environment where steric and electronic effects work synergistically to control substrate approach and orientation. In the initial coupling step between the naphthalene-indole compound and propargyl alcohol derivative, the chiral phosphoric acid catalyst facilitates proton transfer while simultaneously organizing both reactants through hydrogen bonding interactions within its confined chiral pocket. This precise spatial arrangement ensures consistent stereochemical outcome regardless of substrate variations. The subsequent transformation steps maintain this structural integrity while introducing the phosphine functionality through carefully controlled palladium-catalyzed reactions that preserve the axial chirality established in the initial step.
Impurity control is achieved through multiple mechanisms inherent in this catalytic system. The mild reaction conditions (0-40°C) minimize thermal decomposition pathways that typically generate impurities in conventional high-temperature processes. The use of standard purification techniques like silica gel chromatography with petroleum ether/ethyl acetate mixtures effectively separates potential byproducts without requiring specialized equipment or hazardous solvents. The high enantioselectivity (consistently >90% ee across multiple examples) significantly reduces diastereomeric impurities that would otherwise require complex separation procedures. Additionally, the modular nature of the synthesis allows for targeted structural modifications to address specific impurity profiles without compromising overall catalytic efficiency or requiring fundamental process changes.
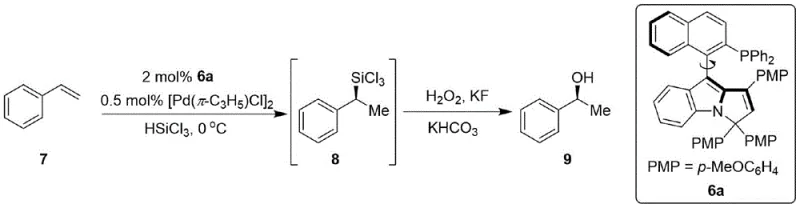
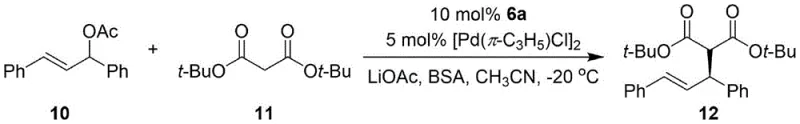
How to Synthesize Naphthalene-Pyrrole Phosphine Catalyst Efficiently
This section outlines the practical implementation of the patented synthesis route for producing high-purity axial chiral naphthalene-pyrrole phosphine catalyst at commercial scale. The process has been optimized to maximize yield while maintaining exceptional enantioselectivity across diverse structural variants. Detailed standardized synthesis procedures have been developed based on extensive process validation studies to ensure consistent quality from batch to batch. The following step-by-step guide provides R&D teams with actionable protocols for successful implementation in their manufacturing facilities.
- Conduct asymmetric coupling between naphthalene-indole compound and propargyl alcohol derivative using chiral phosphoric acid catalyst in dichloromethane at room temperature
- Convert hydroxyl group to triflate using trifluoromethanesulfonic anhydride at low temperature with DIPEA as base
- Perform palladium-catalyzed phosphination followed by reduction to obtain final phosphine catalyst
Commercial Advantages for Procurement and Supply Chain Teams
This innovative catalyst system directly addresses critical pain points in pharmaceutical manufacturing supply chains by offering a versatile solution that reduces complexity while improving reliability. The simplified synthesis pathway eliminates multiple processing steps typically required with conventional chiral catalysts, creating significant opportunities for cost optimization without compromising product quality or performance characteristics. By standardizing on a single platform technology applicable across multiple reaction types, procurement teams can streamline supplier relationships and reduce inventory complexity while maintaining access to high-performance catalytic solutions.
- Cost Reduction in Manufacturing: Elimination of expensive transition metals from the catalytic cycle removes significant raw material costs while avoiding complex metal removal processes required in final product purification. The mild reaction conditions reduce energy consumption compared to traditional high-temperature processes, while simplified purification procedures decrease solvent usage and waste generation. This comprehensive approach delivers substantial cost savings throughout the manufacturing process without requiring major capital investments or process revalidation.
- Enhanced Supply Chain Reliability: The use of readily available starting materials ensures consistent supply regardless of market fluctuations affecting specialty chemicals. The modular design allows for rapid adaptation to changing requirements without requiring complete process redevelopment, providing flexibility to meet evolving customer needs. Standardized manufacturing protocols enable seamless technology transfer between production sites while maintaining consistent quality parameters essential for pharmaceutical applications.
- Scalability and Environmental Compliance: The process has been validated from laboratory scale through pilot production to commercial manufacturing volumes (100 kgs to 100 MT), demonstrating excellent scalability without performance degradation. The elimination of hazardous reagents and reduction in solvent usage aligns with green chemistry principles while reducing environmental impact. Simplified waste streams facilitate more efficient treatment processes and lower disposal costs compared to conventional catalytic systems requiring extensive metal removal procedures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this axial chiral catalyst technology in pharmaceutical manufacturing environments. These answers are derived from extensive process validation studies and real-world application data collected during development and scale-up activities.
Q: How does this axial chiral catalyst improve enantioselectivity compared to traditional chiral phosphines?
A: The unique naphthalene-pyrrole scaffold creates a well-defined chiral environment that provides superior stereocontrol in asymmetric transformations, as evidenced by consistently high ee values across multiple substrate classes.
Q: What are the key advantages of this catalyst preparation method for commercial scale-up?
A: The process features mild reaction conditions (0-40°C), readily available starting materials, simple purification procedures using standard silica gel chromatography, and high overall yields without requiring expensive transition metals.
Q: How does this catalyst system address common supply chain challenges in asymmetric synthesis?
A: The modular synthesis allows for structural diversification through various substituents while maintaining consistent performance, ensuring reliable supply of customized catalysts for specific pharmaceutical intermediate production needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalene-Pyrrole Phosphine Catalyst Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex specialty chemicals for global pharmaceutical clients. Our state-of-the-art facilities feature stringent purity specifications and rigorous QC labs equipped with advanced analytical instrumentation to ensure consistent product quality meeting pharmaceutical industry standards. We understand that successful implementation requires more than just high-quality materials - it demands deep technical expertise in process optimization and scale-up challenges specific to asymmetric catalysis.
Contact our technical procurement team today to request Customized Cost-Saving Analysis tailored to your specific manufacturing needs. We'll provide detailed COA data and route feasibility assessments demonstrating how our axial chiral naphthalene-pyrrole phosphine catalyst can optimize your production process while maintaining the highest quality standards required for pharmaceutical applications.
