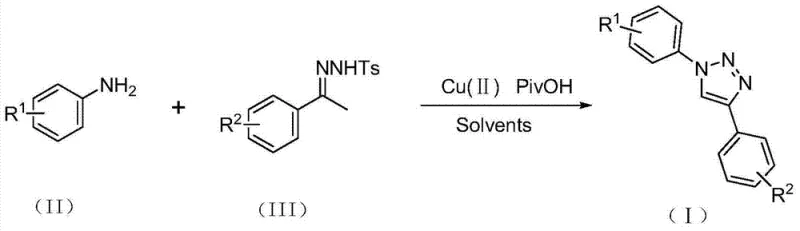
Advanced Copper-Catalyzed Synthesis of 1,4-Disubstituted Triazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies that balance high purity with operational safety, particularly for nitrogen-containing heterocycles which serve as privileged scaffolds in drug design. Patent CN103483279A introduces a transformative preparation method for 1,4-disubstituted triazole compounds, addressing critical bottlenecks in traditional heterocyclic synthesis. This technology leverages a copper-catalyzed coupling of aromatic amines and substituted acetophenone p-toluenesulfonyl hydrazones, bypassing the notorious hazards associated with azide chemistry. For R&D directors and procurement specialists, this represents a paradigm shift towards safer, more scalable manufacturing of high-purity pharmaceutical intermediates. The method's ability to operate under ambient atmospheric conditions without the need for rigorous exclusion of moisture or oxygen drastically reduces the complexity of reactor setup, making it an ideal candidate for cost reduction in API manufacturing where safety and throughput are paramount concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-disubstituted triazoles has been dominated by the Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC) reaction, famously discovered by Sharpless and Meldal. While chemically elegant, this conventional approach suffers from severe practical limitations when translated to industrial scale. The primary dependency on sodium azide or organic azides introduces significant safety liabilities, as these reagents are inherently toxic and possess a high risk of explosive decomposition, especially upon heating or shock. Furthermore, the requirement for strictly anhydrous and oxygen-free environments necessitates expensive specialized equipment, such as gloveboxes or extensive nitrogen purging systems, which inflates capital expenditure and operational costs. These factors collectively create a fragile supply chain for high-purity OLED material or API precursors, where any deviation in inert gas quality can lead to batch failure.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a benign catalytic system comprising a divalent copper salt and pivalic acid to drive the cyclization of readily available aromatic amines and tosylhydrazones. This innovative route completely eliminates the need for hazardous azide reagents, replacing them with stable, commercially accessible starting materials that pose minimal handling risks. The reaction proceeds efficiently in common organic solvents like toluene or DMF at moderate temperatures of 100-110°C, and critically, it tolerates the presence of air and moisture. This robustness simplifies the post-treatment process to basic filtration and column chromatography, removing the need for complex quenching procedures required for reactive azides. By shifting the synthetic logic from cycloaddition to an oxidative coupling mechanism, this approach offers a reliable pharmaceutical intermediate supplier pathway that is both economically and environmentally superior.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation is distinct from the classic [3+2] cycloaddition, proceeding instead through a copper-mediated oxidative sequence that ensures high regioselectivity for the 1,4-isomer. The divalent copper salt, typically copper acetate, serves a dual function as both an oxidant and a Lewis acid catalyst. Initially, the copper species promotes the dehydrogenation of the substituted acetophenone p-toluenesulfonyl hydrazone, generating a reactive diazoalkene intermediate in situ. This electrophilic species is then attacked by the nucleophilic aromatic amine in an N-hetero-Michael addition fashion. The presence of pivalic acid is mechanistically vital, acting as a proton shuttle and ligand that stabilizes the copper center, thereby facilitating the subsequent intramolecular N-N bond formation. This step is followed by a final aromatization driven by the copper catalyst, yielding the stable 1,4-disubstituted triazole core. Understanding this mechanism allows process chemists to fine-tune the molar ratios, specifically maintaining the copper to pivalic acid ratio between 1:1.5 and 1:2.5 to maximize conversion efficiency.
From an impurity control perspective, this mechanism offers significant advantages over radical-based or high-energy azide pathways. Because the reaction avoids the generation of free azide ions or highly energetic nitrene intermediates, the formation of side products such as polymeric tars or explosive byproducts is minimized. The selectivity is further enhanced by the specific coordination environment created by the pivalate ligands, which directs the attack of the amine to the correct position on the diazo species. This intrinsic selectivity reduces the burden on downstream purification, meaning that commercial scale-up of complex polymer additives or drug candidates can be achieved with fewer chromatographic cycles. For quality control teams, this translates to a cleaner crude profile and a more consistent impurity spectrum, which is essential for meeting the stringent purity specifications required in regulated pharmaceutical markets.
How to Synthesize 1,4-Disubstituted Triazoles Efficiently
Implementing this synthesis protocol requires precise attention to reagent stoichiometry and thermal management to ensure optimal yields. The process begins by charging a reaction vessel with the divalent copper salt, pivalic acid, the chosen aromatic amine, and the substituted acetophenone p-toluenesulfonyl hydrazone in a suitable aprotic solvent. The mixture is then heated to a controlled temperature range of 100-110°C and maintained for a duration of 10 to 12 hours to ensure complete consumption of the starting materials. Unlike sensitive organometallic reactions, this step does not require inert gas blanketing, allowing for simpler reactor operation. Following the reaction period, the detailed standardized synthesis steps involve a straightforward workup procedure where the mixture is filtered to remove inorganic salts, mixed with silica gel, and subjected to column chromatography to isolate the pure target compound.
- Combine divalent copper salt, pivalic acid, aromatic amine, and substituted acetophenone p-toluenesulfonyl hydrazone in an organic solvent.
- Heat the reaction mixture to 100-110°C and maintain for 10-12 hours without requiring inert atmosphere protection.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on risk mitigation and cost efficiency. The elimination of explosive azides removes a major regulatory and safety hurdle, significantly lowering insurance premiums and storage costs associated with hazardous materials. Furthermore, the reliance on commodity chemicals like anilines and acetophenones, which are produced in massive volumes globally, ensures a stable and resilient supply base that is less susceptible to market volatility compared to specialized alkyne or azide building blocks. This stability is crucial for maintaining continuous production schedules and reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations without the fear of raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of operational requirements and the use of low-cost catalysts. By removing the necessity for expensive inert atmosphere equipment and the rigorous drying of solvents, the capital and operational expenditures for the manufacturing facility are drastically reduced. Additionally, the use of inexpensive copper acetate and pivalic acid replaces costly noble metal catalysts or specialized ligands often required in alternative cross-coupling reactions. The simplified workup procedure, which avoids complex aqueous quenches for hazardous reagents, further lowers waste disposal costs and labor time, resulting in substantial cost savings across the entire production lifecycle.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions directly translates to improved supply chain reliability and predictability. Since the reaction tolerates ambient conditions, the risk of batch failure due to minor leaks in reactor seals or moisture ingress is virtually eliminated, ensuring consistent output quality. The starting materials, aromatic amines and tosylhydrazones, are widely available from multiple global suppliers, preventing single-source bottlenecks. This diversification of the supply base enhances the security of supply for critical intermediates, ensuring that production timelines are met reliably even during periods of global logistical disruption or raw material scarcity.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous azides, which often impose strict limits on batch sizes due to thermal runaway risks. The ability to run reactions at 100-110°C in standard glass-lined or stainless steel reactors without special explosion-proofing allows for seamless technology transfer. Moreover, the reduced generation of toxic waste streams aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. This environmental compatibility not only reduces compliance costs but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B chemical procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety protocols, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios for fine chemical intermediates.
Q: Why is this copper-catalyzed method safer than traditional CuAAC reactions?
A: Traditional Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC) relies on sodium azide or organic azides, which are highly toxic and prone to explosive decomposition. This novel method utilizes stable tosylhydrazones and aromatic amines, eliminating the safety risks associated with handling explosive azide reagents while maintaining high regioselectivity.
Q: What are the optimal reaction conditions for scaling up this synthesis?
A: The process operates efficiently at temperatures between 100°C and 110°C using common aprotic solvents like toluene, DMF, or acetonitrile. Crucially, the reaction does not require stringent anhydrous or oxygen-free conditions, significantly simplifying the engineering requirements for large-scale commercial production compared to sensitive organometallic processes.
Q: How does the use of pivalic acid impact the reaction efficiency?
A: Pivalic acid acts as a crucial promoter in this catalytic system. It facilitates the dehydrogenation of the p-toluenesulfonylhydrazone to form the reactive diazoalkene intermediate and assists in the subsequent N-N bond formation and aromatization steps, leading to higher yields and cleaner reaction profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Disubstituted Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global fine chemicals market. Our team of expert process chemists has extensively evaluated the copper-catalyzed route described in CN103483279A and possesses the technical capability to optimize this methodology for your specific project needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,4-disubstituted triazole meets the highest standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this safer, more cost-effective synthetic route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this azide-free methodology for your specific molecule. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our catalog compounds, and receive comprehensive route feasibility assessments tailored to your production goals. Let us help you secure a reliable supply of high-quality intermediates while optimizing your manufacturing costs and safety profile.
