Revolutionizing Triazole Synthesis: Safe Copper-Catalyzed Process for High-Purity Pharmaceutical Intermediates at Commercial Scale
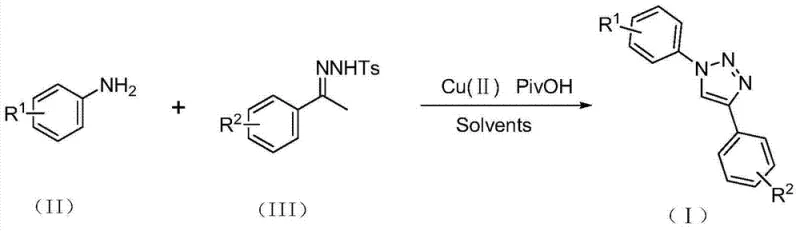
Patent CN103483279A presents a groundbreaking methodology for synthesizing 1,4-disubstituted triazole compounds, which serve as critical building blocks in pharmaceutical development due to their prevalence in bioactive molecules including HIV inhibitors and β3-adrenergic receptor modulators. This innovative approach eliminates the need for hazardous azide reagents that have traditionally been required in triazole synthesis, thereby addressing significant safety concerns in chemical manufacturing processes while maintaining excellent yields across diverse substrate combinations. The method employs readily available copper(II) salts and pivalic acid as catalysts under mild reaction conditions of 100–110°C without requiring anhydrous or oxygen-free environments—a substantial advancement over conventional copper-catalyzed azide-alkyne cycloaddition techniques that depend on toxic and explosive sodium azide. This patent demonstrates how this novel process achieves comparable or superior yields while dramatically improving operational safety and reducing process complexity for industrial-scale production of these valuable heterocyclic compounds essential to modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of 1,4-disubstituted triazoles primarily relies on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions that require handling toxic and potentially explosive azide compounds such as sodium azide or organic azides—a significant operational hazard that necessitates specialized safety protocols, dedicated equipment for anhydrous and oxygen-free conditions, and extensive operator training to prevent catastrophic accidents during manufacturing operations. These stringent environmental controls substantially increase capital expenditure through specialized reactor systems and containment infrastructure while limiting production flexibility in standard chemical facilities due to regulatory restrictions on hazardous material handling. Furthermore, the purification processes for azide-containing reactions often involve additional steps to remove residual heavy metals and toxic byproducts like hydrazoic acid, complicating waste management protocols and increasing the overall environmental footprint through specialized neutralization procedures required before disposal or treatment.
The Novel Approach
The patented methodology described in CN103483279A overcomes these limitations through an innovative copper-catalyzed reaction pathway that utilizes p-toluenesulfonyl hydrazones instead of hazardous azides as the key nitrogen source—eliminating the primary safety concern while maintaining high regioselectivity for the desired 1,4-isomer configuration critical for pharmaceutical applications. This approach operates effectively under standard laboratory conditions without requiring specialized anhydrous or oxygen-free environments, significantly simplifying process setup and reducing capital investment requirements by leveraging existing manufacturing infrastructure without modifications. The reaction employs commercially available copper(II) acetate monohydrate and pivalic acid as catalysts in common organic solvents like toluene at moderate temperatures of 100–110°C for precisely controlled durations of 10–12 hours—conditions easily maintained in standard chemical processing equipment without exotic engineering controls.
Mechanistic Insights into Copper-Catalyzed Triazole Formation
The reaction mechanism begins with copper(II) salts promoting dehydrogenation of p-toluenesulfonyl hydrazones to form diazoalkene intermediates through a redox process where copper facilitates hydrogen abstraction—this critical activation step occurs without generating explosive intermediates that characterize traditional azide-based approaches. These diazoalkenes subsequently undergo N-hetero-Michael addition with aromatic amines at the olefinic double bond, a transformation enabled by the Lewis acidic properties of copper(II) ions that activate the diazoalkene toward nucleophilic attack by the amine nitrogen while preventing undesired side reactions through precise coordination control. The resulting adduct then undergoes copper-catalyzed N-N bond formation followed by aromatization to yield the final 1,4-disubstituted triazole product with excellent regioselectivity—this sequence avoids the formation of potentially explosive intermediates while maintaining high conversion efficiency through careful modulation of the copper catalyst's oxidation state by pivalic acid.
The process demonstrates exceptional impurity control through multiple mechanisms inherent to its innovative design—firstly by eliminating potential formation of toxic hydrazoic acid byproducts that commonly contaminate conventional triazole syntheses through azide decomposition pathways under thermal stress conditions. Secondly, the moderate reaction temperature range (100–110°C) prevents thermal decomposition pathways that could generate unwanted side products such as dimerized or polymerized species that often complicate purification in higher temperature processes. Thirdly, the specific copper(II)/pivalic acid catalyst system promotes clean conversion to the target triazole without significant over-reaction or isomerization byproducts due to its selective activation profile toward the desired reaction pathway—this selectivity is further enhanced by using aromatic amines in excess which ensures complete consumption of the hydrazone substrate while minimizing unreacted starting materials in the final product mixture.
How to Synthesize 1,4-Disubstituted Triazoles Efficiently
This patented methodology offers a practical solution for manufacturing high-purity triazole intermediates through a streamlined process that eliminates hazardous reagents while maintaining excellent yield and selectivity across diverse substrate combinations including various substituted aromatic systems with R1 groups ranging from hydrogen to halogens and R2 groups covering alkyl to aryl moieties. The procedure leverages readily available starting materials and standard laboratory equipment to produce these valuable heterocyclic compounds under safe operating conditions that are fully compatible with existing manufacturing infrastructure without requiring capital-intensive modifications or specialized handling protocols for dangerous chemicals.
- Combine divalent copper salt (e.g., copper acetate), pivalic acid, aromatic amine, and substituted acetophenone p-toluenesulfonyl hydrazone in organic solvent (toluene preferred) at optimized molar ratios
- Heat reaction mixture to 100–110°C under standard atmospheric conditions for 10–12 hours without requiring anhydrous or oxygen-free environment
- Perform post-treatment via filtration followed by silica gel sample mixing and column chromatography purification to obtain high-purity triazole product
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology addresses critical pain points in the supply chain for pharmaceutical intermediates by offering a safer, more reliable production pathway that reduces operational complexities while maintaining high product quality standards required by regulatory agencies—particularly valuable given increasing global scrutiny on chemical manufacturing safety protocols and environmental impact assessments within pharmaceutical supply chains.
- Cost Reduction in Manufacturing: The elimination of toxic azide reagents removes significant costs associated with specialized safety equipment investments, hazardous material handling protocols requiring dedicated personnel training, and extensive waste treatment procedures necessary for neutralizing azide-containing effluents—these savings manifest through reduced capital expenditure on containment systems and lower operational costs from simplified regulatory compliance requirements without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials that do not require special storage conditions significantly improves supply chain flexibility by removing dependency on specialized chemical suppliers who handle hazardous materials—this methodology's compatibility with standard manufacturing equipment enables rapid scale-up using existing production facilities without requiring new infrastructure investments or complex validation procedures typically needed when introducing hazardous reagents into established production lines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory benchtop (as demonstrated in patent examples yielding up to gram-scale quantities) through pilot plant operations to full commercial production volumes due to its straightforward reaction conditions that avoid extreme temperature/pressure requirements—this scalability is further enhanced by simplified waste streams that eliminate hazardous components requiring special disposal protocols, aligning with increasingly stringent global environmental regulations while reducing overall environmental footprint through minimized solvent usage during purification steps.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement specialists and technical teams regarding implementation of this patented triazole synthesis methodology in commercial manufacturing environments—these answers are derived directly from experimental data presented in patent CN103483279A including detailed reaction condition optimization studies across multiple substrate combinations.
Q: How does this method eliminate safety risks compared to conventional triazole synthesis?
A: The patented process replaces hazardous azide reagents with p-toluenesulfonyl hydrazones as nitrogen sources, eliminating risks associated with toxic/explosive sodium azide handling while maintaining high regioselectivity for the desired 1,4-isomer.
Q: What advantages does this method offer for supply chain continuity?
A: By utilizing commercially available starting materials that require no special storage conditions and eliminating regulatory barriers associated with hazardous chemicals, this method significantly enhances supply chain resilience and simplifies global logistics.
Q: How does the process maintain high purity without specialized equipment?
A: The inherent reaction design prevents formation of toxic byproducts through controlled copper catalysis at moderate temperatures (100–110°C), with standard column chromatography achieving pharmaceutical-grade purity without requiring specialized purification infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Disubstituted Triazole Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds including this patented triazole synthesis methodology—our state-of-the-art manufacturing facilities are equipped with rigorous QC labs that ensure stringent purity specifications are consistently met across all production batches through comprehensive analytical testing protocols validated against pharmacopeial standards. As a trusted CDMO partner specializing in challenging heterocyclic chemistry development and manufacturing services, we combine deep technical expertise with flexible production capabilities to support clients from early-stage development through commercial production phases while maintaining full regulatory compliance across global markets.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target compounds—take advantage of our Customized Cost-Saving Analysis service which evaluates how this innovative synthesis approach can optimize your supply chain while maintaining the highest quality standards required for pharmaceutical applications through detailed process economics modeling based on your specific volume requirements.
